Advanced Pressurized Ammonolysis for High-Purity Glycyl-L-glutamine Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical amino acid derivatives, particularly those serving as active ingredients in parenteral nutrition formulations. A pivotal advancement in this domain is documented in patent CN103694313A, which discloses a highly efficient preparation method for Glycyl-L-glutamine. This dipeptide is a vital component in compound amino acid injections, necessitating stringent purity standards to ensure patient safety. The core innovation lies in replacing traditional, salt-heavy ammonolysis protocols with a pressurized ammonia gas technique. By utilizing chloroacetyl-L-glutamine as the starting material and reacting it directly with ammonia gas under controlled pressure (0.2~0.4 Mpa) and moderate temperature (42~48°C), the process achieves a reaction completion time of merely 5 hours. This stands in stark contrast to legacy methods that often require days or involve cumbersome downstream processing. The structural integrity and identity of the target molecule, Glycyl-L-glutamine monohydrate, are central to this optimization effort.
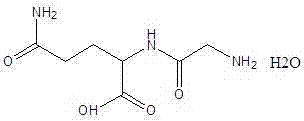
Furthermore, the absence of introduced ammonium salts during the reaction phase represents a paradigm shift in process chemistry for this intermediate. Conventional wisdom often dictated the use of excessive ammonium bicarbonate or concentrated aqueous ammonia to drive equilibrium, but this patent demonstrates that physical pressure can achieve superior results without the chemical baggage. The resulting crude product undergoes a refined purification sequence involving activated carbon treatment and methanol-induced crystallization at sub-zero temperatures (-4~-1°C). This meticulous control over the crystallization environment ensures that the final purity of the Glycyl-L-glutamine is not less than 99.3%, meeting the rigorous specifications required for a reliable pharmaceutical intermediate supplier catering to global injectable drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Glycyl-L-glutamine have been plagued by significant operational inefficiencies and chemical complexities that hinder industrial scalability. For instance, earlier patents such as CN1532204A described a multi-step pathway involving amino protection of glycine, activation to an ester, coupling with L-glutamine, and finally, the removal of protecting groups via palladium-carbon reduction. This sequence is inherently fragile; the reliance on precious metal catalysts introduces not only high raw material costs but also the regulatory burden of validating heavy metal clearance, which complicates the quality control workflow. Another prior art reference, CN1740188A, utilized glutamic acid as a starting point but suffered from excessively long reaction cycles, requiring the reaction mixture to stand in a light bulb bottle for up to one week. Such temporal inefficiencies create bottlenecks in production scheduling and occupy valuable reactor capacity for extended periods, drastically reducing overall plant throughput and increasing the cost reduction in API manufacturing.
Perhaps the most critical drawback of previous methodologies, exemplified by EP0678501A1, was the reliance on massive stoichiometric excesses of ammonium salts. This method required 20 equivalents of ammonia water and 10 equivalents of ammonium bicarbonate relative to the substrate. The introduction of such vast quantities of inorganic salts creates a downstream nightmare: the reaction mixture contains nearly 11.14 times the volume of water that must be evaporated to isolate the product. During concentration, the volatilized ammonium salts tend to sublime and redeposit in the condenser systems, leading to frequent blockages that halt production and require manual intervention. This not only poses safety risks due to pressure buildup but also results in inconsistent batch quality and extended downtime for equipment cleaning, making these conventional routes unsuitable for modern, continuous commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology outlined in CN103694313A effectively dismantles these barriers by re-engineering the ammonolysis step to operate in a cleaner, anhydrous-favorable environment. Instead of flooding the reactor with aqueous ammonia and bicarbonate salts, the novel approach introduces high-purity ammonia gas directly into an aqueous solution of chloroacetyl-L-glutamine. By maintaining a system pressure of 0.2~0.4 Mpa, the solubility of ammonia in the reaction medium is significantly enhanced without the need for additional water or salt carriers. This physical modification of the reaction conditions allows the nucleophilic substitution to proceed rapidly, completing the transformation in just 5 hours at a mild temperature range of 42~48°C. The elimination of ammonium salts means that the volume of solvent requiring evaporation is minimized to less than 5 times the raw material weight, a drastic reduction compared to the 11-fold evaporation required by older methods. Consequently, the concentration step is faster, energy consumption is lowered, and the risk of condenser fouling is entirely eradicated, facilitating a smoother transition from laboratory bench to industrial production.
Mechanistic Insights into Pressurized Gas Ammonolysis
The success of this synthesis hinges on the thermodynamic and kinetic advantages provided by pressurized gas-liquid reactions. In the context of nucleophilic acyl substitution, the chloroacetyl group on the starting material is highly susceptible to attack by ammonia. However, in aqueous environments, the concentration of free ammonia is limited by its equilibrium with ammonium hydroxide. By applying external pressure (0.2~0.4 Mpa), the system forces a higher concentration of molecular ammonia into the liquid phase, effectively driving the reaction forward according to Le Chatelier's principle. This high local concentration of the nucleophile ensures that the primary ammonolysis reaction occurs swiftly and selectively. Crucially, the patent data indicates that maintaining this specific pressure window is vital; comparative examples show that lower pressures or atmospheric conditions lead to incomplete reactions or the formation of secondary ammonolysis by-products, where the ammonia attacks other susceptible sites on the molecule. The pressurized environment suppresses these side reactions by ensuring the primary substitution is kinetically dominant, thereby preserving the structural fidelity of the dipeptide backbone.
Impurity control is further managed through the strategic exclusion of inorganic salts. In traditional salt-based methods, the presence of ammonium bicarbonate can lead to complex ion-pairing interactions that trap impurities within the crystal lattice of the product or form difficult-to-separate co-crystals. By avoiding the introduction of these extraneous ions, the new process yields a crude product with a liquid phase purity of not less than 97.8% even before final recrystallization. The subsequent purification steps leverage this cleanliness; the use of activated carbon at 52~56°C effectively adsorbs trace organic colored impurities and residual starting materials without interference from high salt loads. Finally, the addition of methanol as an anti-solvent at low temperatures (-4~-1°C) exploits the differential solubility of the target dipeptide versus potential by-products. This precise thermal and solvent control ensures that the final crystalline form is highly ordered, resulting in the reported HPLC purity of 99.3% to 99.5%, which is essential for reducing lead time for high-purity pharmaceutical intermediates in regulatory filings.
How to Synthesize Glycyl-L-glutamine Efficiently
The operational protocol derived from this patent offers a clear pathway for manufacturing teams to implement this superior chemistry. The process is designed to be robust, utilizing standard lined reactors that are commonly available in fine chemical facilities, thus avoiding the need for exotic metallurgy. The key to success lies in the precise control of the ammonia feed rate and the maintenance of the pressure-temperature profile during the initial 5-hour reaction window. Following the reaction, the workup procedure is streamlined to minimize hold times, moving quickly from concentration to decolorization and finally to crystallization. Detailed standardized operating procedures regarding the specific addition rates of methanol and the cooling ramps are critical to maximizing yield and crystal morphology. For a comprehensive breakdown of the exact parameters and safety considerations required for execution, please refer to the technical guide below.
- Dissolve chloroacetyl-L-glutamine in water (5: 1 ratio) and introduce ammonia gas to 0.2-0.4 Mpa, reacting at 42-48°C for 5 hours.
- Concentrate under reduced pressure to near dryness, redissolve in water, treat with activated carbon, and filter to remove impurities.
- Add methanol to the filtrate, cool to -4 to -1°C to induce crystallization, then centrifuge and dry to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this pressurized ammonolysis technology offers profound advantages that extend beyond mere chemical yield. The primary value driver is the significant simplification of the utility load and waste management profile. By eliminating the need to evaporate massive volumes of water associated with ammonium salt by-products, the facility drastically reduces its steam consumption and wastewater generation. This translates directly into substantial cost savings in manufacturing operations, as energy represents a major portion of the variable cost in bulk pharmaceutical synthesis. Furthermore, the removal of palladium catalysts and complex protecting group chemistries simplifies the raw material supply chain. Procurement managers no longer need to source expensive noble metals or manage the regulatory documentation associated with heavy metal residues, thereby reducing administrative overhead and mitigating supply risk for critical reagents.
- Cost Reduction in Manufacturing: The elimination of ammonium salts and the reduction in solvent evaporation volume fundamentally alter the cost structure of production. Traditional methods require extensive energy input to boil off over 11 times the solvent volume, whereas this new method requires evaporating less than 5 times the volume. This reduction in thermal load leads to significantly lower utility costs per kilogram of product. Additionally, the avoidance of palladium-on-carbon catalysts removes a high-cost line item from the bill of materials. The streamlined process also reduces labor hours associated with cleaning clogged condensers and managing complex filtration steps, contributing to a more lean and cost-effective manufacturing operation that enhances overall margin potential for the final API.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to greater predictability in production scheduling. Older methods that relied on week-long standing times or were prone to equipment fouling introduced high variability into lead times. In contrast, the 5-hour reaction cycle of this novel approach allows for rapid batch turnover, enabling manufacturers to respond more agilely to market demand fluctuations. The use of commodity chemicals like ammonia gas and methanol, rather than specialized active esters or protected amino acids, ensures that raw material availability is stable and less susceptible to niche supply disruptions. This reliability is crucial for maintaining continuous supply lines to downstream pharmaceutical customers who operate on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard plastic-lined or enamel reactors that are widely available in the industry. The absence of heavy metal catalysts simplifies the environmental compliance landscape, as there is no need for specialized hazardous waste disposal streams for spent palladium. Moreover, the reduction in wastewater volume and the elimination of ammonium salt discharge align with increasingly stringent environmental regulations regarding nitrogen content in effluent. This 'green chemistry' aspect not only future-proofs the manufacturing site against regulatory tightening but also appeals to multinational corporations with strict sustainability mandates, positioning the supplier as a partner capable of delivering high-purity OLED material or pharma intermediates with a reduced environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific comparative data and beneficial effects highlighted in the patent documentation, providing clarity on why this method represents a superior choice for industrial application. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios.
Q: Why is pressurized ammonia gas preferred over aqueous ammonia in this synthesis?
A: Using pressurized ammonia gas eliminates the introduction of massive amounts of water and ammonium salts (like ammonium bicarbonate) required in conventional aqueous methods. This significantly reduces the volume of solvent that must be evaporated during concentration, preventing condenser clogging and shortening cycle times.
Q: How does this method control secondary ammonolysis by-products?
A: By maintaining a high concentration of ammonia through pressurization (0.2-0.4 Mpa) rather than relying on stoichiometric salt additives, the reaction kinetics favor the primary substitution. This suppresses the formation of difficult-to-remove secondary ammonolysis products, ensuring final purity exceeds 99.3%.
Q: What are the scalability advantages of this route compared to Pd/C reduction methods?
A: Unlike methods requiring palladium-carbon reduction and amino protection groups, this direct ammonolysis route avoids expensive noble metal catalysts and complex protection/deprotection steps. This simplifies the supply chain, removes heavy metal clearance requirements, and allows for easier scale-up in standard lined reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycyl-L-glutamine Supplier
The technical advancements detailed in patent CN103694313A underscore the potential for producing Glycyl-L-glutamine with exceptional efficiency and purity. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facility is equipped with the necessary high-pressure reactor infrastructure and stringent purity specifications required to execute this pressurized ammonolysis process safely and consistently. With our rigorous QC labs, we guarantee that every batch meets the critical quality attributes necessary for parenteral applications, providing our partners with a secure and compliant source of this vital pharmaceutical intermediate.
We invite procurement leaders and technical directors to engage with us to explore how this optimized route can benefit your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the competitive landscape of amino acid therapeutics.
