Advanced Biocatalytic Production of Chiral Statin Intermediates via KRED102 Technology
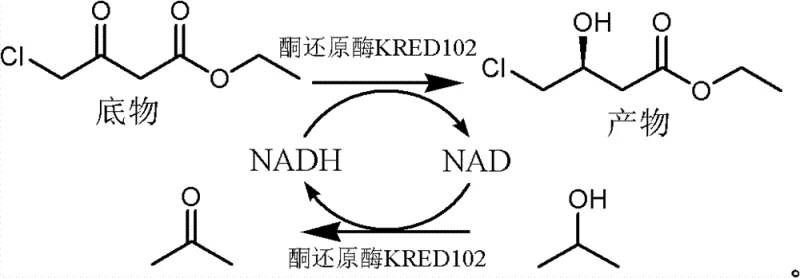
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and cost-effective routes for chiral building blocks has led to significant advancements in biocatalysis, particularly for the synthesis of statin intermediates. Patent CN102605011B introduces a groundbreaking biological preparation method for (S)-4-chloro-3-hydroxyl ethyl butyrate, a pivotal chiral precursor in the manufacturing of HMG-CoA reductase inhibitors. This technology leverages the specific catalytic power of ketoreductase KRED102 to perform an asymmetric reduction of 4-chloro-3-carbonyl ethyl butyrate under mild, aqueous conditions. Unlike traditional chemical reduction methods that often struggle with stereoselectivity and environmental hazards, or earlier enzymatic processes burdened by expensive cofactor requirements, this innovation utilizes a robust system incorporating isopropanol as a hydrogen donor, polyethylene glycol 400 as a dispersant, and divalent metal ions as activity enhancers. The result is a highly scalable process capable of achieving high substrate concentrations and exceptional conversion rates, positioning it as a superior choice for reliable pharmaceutical intermediates supplier networks seeking to optimize their supply chains for cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral chlorohydrins like (S)-4-chloro-3-hydroxyl ethyl butyrate has been fraught with technical and economic challenges that hinder large-scale commercialization. Conventional chemical synthesis routes typically rely on harsh reducing agents such as borohydrides, which often yield racemic mixtures requiring costly and yield-loss-inducing resolution steps to isolate the desired (S)-enantiomer. Furthermore, these chemical processes frequently necessitate the use of volatile organic solvents and generate significant hazardous waste, creating substantial environmental compliance burdens and disposal costs for manufacturing facilities. In the realm of biocatalysis, earlier generations of enzymatic methods often depended on NADP-dependent reductases coupled with glucose dehydrogenase systems. While effective, these systems impose a heavy financial burden due to the high cost of NADP cofactors and the requirement for stoichiometric amounts of glucose, which can lead to reaction mixture emulsification and complicate the downstream extraction and purification of the target product. Additionally, many prior art methods required two-phase solvent systems with high organic content to solubilize the hydrophobic substrate, thereby negating the green chemistry benefits of using enzymes and increasing the overall process complexity and safety risks.
The Novel Approach
The methodology disclosed in patent CN102605011B represents a paradigm shift by engineering a single-phase aqueous reaction system that maximizes efficiency while minimizing operational complexity. By employing ketoreductase KRED102, the process achieves high stereospecificity directly, eliminating the need for chiral resolution. A critical innovation in this approach is the substitution of expensive glucose/NADP systems with an isopropanol-driven cofactor regeneration cycle, which not only reduces raw material costs but also produces acetone as a benign byproduct that is easily removed. The strategic addition of polyethylene glycol 400 serves as a non-toxic dispersant, allowing for significantly higher substrate loading (up to 30% w/v) in water without the need for large volumes of organic co-solvents. This enhancement effectively overcomes the mass transfer limitations typical of aqueous enzymatic reactions involving hydrophobic substrates. Furthermore, the inclusion of divalent metal ions acts as a potent enzymatic activity intensifier, accelerating the reaction kinetics and ensuring that transformation efficiencies reach between 80% and 100% within practical timeframes, making this route exceptionally viable for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into KRED102-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the precise mechanistic interplay between the biocatalyst KRED102, the cofactor recycling system, and the specialized reaction additives. The enzyme KRED102 facilitates the hydride transfer from the reduced cofactor NADH to the prochiral ketone carbonyl of the substrate, 4-chloro-3-carbonyl ethyl butyrate, with absolute stereocontrol to yield the (S)-configured alcohol. To sustain this catalytic cycle economically, the system utilizes isopropanol as a sacrificial hydrogen donor; as the enzyme reduces the substrate, it oxidizes isopropanol to acetone, simultaneously regenerating NADH from NAD+. This coupling ensures that only catalytic amounts of the expensive nicotinamide cofactor are required, driving the equilibrium towards product formation through the removal or volatility of the acetone byproduct. The presence of polyethylene glycol 400 is mechanistically vital as it modifies the microenvironment of the enzyme, likely forming micellar structures or acting as a phase-transfer agent that solubilizes the lipophilic ester substrate within the aqueous buffer, thereby increasing the effective concentration of substrate available at the enzyme's active site without denaturing the protein.
Beyond the primary catalytic cycle, the role of divalent metal ions such as calcium (Ca2+) or magnesium (Mg2+) cannot be overstated in terms of process robustness and impurity control. These ions function as enzymatic activity tougheners, likely stabilizing the tertiary structure of the KRED102 protein against thermal denaturation or conformational shifts that could occur during prolonged agitation at industrial scales. From an impurity profile perspective, this aqueous, metal-enhanced system minimizes the formation of side products often associated with chemical reduction, such as over-reduced alkanes or elimination products (chloro-butenoates), because the enzyme's active site provides a highly specific steric environment. The mild pH range of 5.0 to 9.0 further ensures that the sensitive ester moiety of the substrate remains intact, preventing hydrolysis which is a common degradation pathway in strongly acidic or basic chemical conditions. Consequently, the resulting crude product possesses a much cleaner impurity spectrum, reducing the burden on downstream purification units and facilitating the production of high-purity pharmaceutical intermediates that meet stringent regulatory specifications for statin synthesis.
How to Synthesize (S)-4-chloro-3-hydroxyl ethyl butyrate Efficiently
Implementing this biocatalytic route requires careful attention to the formulation of the reaction medium to balance substrate solubility with enzyme stability. The process begins with the preparation of a buffered aqueous solution, typically phosphate or Tris-HCl, adjusted to a pH between 5.0 and 9.0 to match the optimal activity window of KRED102. Into this buffer, polyethylene glycol 400 is added at a concentration ranging from 10% to 70% v/v, serving as the critical dispersant that enables the high-concentration feeding of the hydrophobic substrate. The substrate, 4-chloro-3-carbonyl ethyl butyrate, is introduced alongside isopropanol (1.2 to 1.5 equivalents) and the divalent metal salt, such as calcium chloride, which activates the enzyme. Once the biocatalyst KRED102 is added, the mixture is stirred at moderate temperatures (20°C to 40°C) to maintain reaction velocity without compromising enzyme integrity. Detailed standard operating procedures and specific step-by-step synthesis instructions are provided below.
- Prepare an aqueous buffered solution (pH 5.0-9.0) containing polyethylene glycol 400 as a dispersant and divalent metal ions such as calcium chloride.
- Introduce the substrate 4-chloro-3-carbonyl ethyl butyrate and the hydrogen donor isopropanol into the reaction vessel along with the biocatalyst KRED102.
- Maintain the reaction at 20°C to 40°C with stirring until conversion reaches 80-100%, followed by extraction with ethyl acetate to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented biocatalytic process offers transformative advantages that directly impact the bottom line and operational resilience. The shift from complex two-phase chemical systems or expensive glucose-dependent enzymatic routes to this streamlined aqueous process eliminates several cost centers simultaneously. By removing the dependency on costly NADP cofactors and glucose dehydrogenase enzymes, the raw material bill of materials is significantly reduced, while the use of commodity-grade isopropanol as a hydrogen donor further drives down variable costs. The ability to operate at high substrate concentrations in a single aqueous phase means that reactor volume utilization is maximized, allowing manufacturers to produce more product per batch without expanding capital infrastructure. This efficiency translates into substantial cost savings in manufacturing overheads, energy consumption, and waste treatment, as the process avoids the generation of heavy metal waste streams and reduces the volume of organic solvents required for extraction compared to traditional methods.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the elimination of expensive chiral catalysts and resolving agents required in chemical synthesis. By utilizing a robust enzyme system that recycles its own cofactor using cheap isopropanol, the process removes the need for stoichiometric reducing agents like sodium borohydride, which carry both high material costs and safety handling expenses. Furthermore, the simplified work-up procedure, which avoids complex phase separations and extensive solvent swaps, reduces the consumption of utilities such as steam for distillation and electricity for agitation. This holistic reduction in process complexity ensures that the cost of goods sold (COGS) for the chiral intermediate is drastically lowered, providing a competitive pricing advantage in the global market for statin precursors.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available, commodity-grade raw materials such as isopropanol, calcium chloride, and polyethylene glycol, which are not subject to the same supply constraints as specialized fine chemicals or precious metal catalysts. The robustness of the aqueous system also means that the process is less sensitive to minor fluctuations in raw material quality, reducing the risk of batch failures that can disrupt production schedules. Additionally, the high conversion rates and selectivity minimize the need for reprocessing or reworking off-spec material, ensuring a consistent and predictable output of finished goods. This reliability allows supply chain planners to maintain leaner inventory levels while confidently meeting the demanding delivery timelines of downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is inherently safer and more straightforward due to the absence of pyrophoric reagents and the use of water as the primary reaction medium. The reduction in organic solvent usage aligns perfectly with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste disposal, future-proofing the manufacturing site against regulatory tightening. The high substrate loading capability demonstrated in the patent indicates that the process is ready for ton-scale production without the need for massive reactor expansions, facilitating rapid commercial scale-up of complex pharmaceutical intermediates. This combination of safety, scalability, and environmental stewardship makes the technology an ideal candidate for long-term strategic sourcing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this KRED102-mediated synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process parameters and performance expectations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this biocatalytic step into existing production lines or for procurement specialists assessing the quality and consistency of the supply source.
Q: What is the specific role of Polyethylene Glycol 400 in this enzymatic reaction?
A: Polyethylene glycol 400 acts as a crucial dispersant that allows for higher substrate concentrations in the aqueous phase without the need for toxic organic co-solvents, significantly improving reaction efficiency and simplifying downstream processing.
Q: Why is KRED102 preferred over other ketoreductases for this specific statin intermediate?
A: KRED102 demonstrates superior stereoselectivity and activity when coupled with inexpensive isopropanol as a hydrogen donor, avoiding the high costs associated with NADP-dependent systems that require glucose and glucose dehydrogenase.
Q: How does the addition of divalent metal ions impact the process yield?
A: The inclusion of divalent metal ions like calcium or magnesium serves as an enzymatic activity intensifier, stabilizing the enzyme structure and accelerating the reduction rate to achieve near-quantitative conversion within shorter timeframes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-chloro-3-hydroxyl ethyl butyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the synthesis of life-saving statin medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. By leveraging advanced biocatalytic technologies like the one described in CN102605011B, we offer a supply solution that balances technical excellence with commercial viability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for your upcoming clinical or commercial batches, our experts are ready to provide the support you need. Contact us today to secure a reliable supply of high-purity pharmaceutical intermediates that will drive the success of your drug development programs.
