Advanced Fluorinated Phenyldiacetylene Derivatives: Scalable Synthesis for Next-Gen LCD Applications
The rapid evolution of flat panel display technology demands liquid crystal materials with increasingly sophisticated physicochemical profiles. Patent CN102557896A introduces a novel class of (poly)fluorine-substituted phenyldiacetylene (bi)phenyl derivatives designed to address these critical performance gaps. These compounds are engineered to possess large optical anisotropy (Δn) and suitable dielectric anisotropy (Δε), essential parameters for high-resolution TFT-LCD and STN-LCD applications. The structural versatility allows for precise tuning of the nematic phase temperature range and driving voltage. As a reliable electronic chemical supplier, understanding the underlying molecular architecture is key to leveraging these materials for next-generation display formulations.
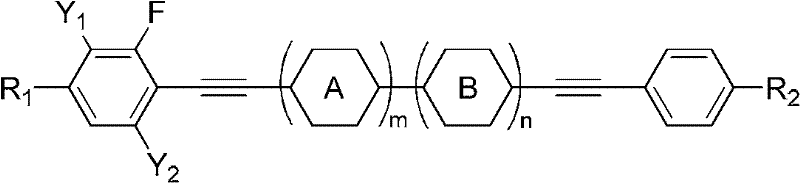
The core innovation lies in the strategic placement of fluorine atoms and the extension of the conjugated system via diacetylene linkages. Unlike conventional liquid crystal monomers that may struggle to balance viscosity and birefringence, this patented architecture utilizes the rigid rod-like structure of the diacetylene bridge to maximize Δn while lateral fluorination ensures low rotational viscosity. This dual optimization is critical for achieving fast response times in modern displays without sacrificing thermal stability. The general formula encompasses a wide range of substituents, allowing manufacturers to customize mixtures for specific electro-optical requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for high-birefringence liquid crystals often rely on complex multi-step sequences that suffer from poor atom economy and difficult purification challenges. Conventional methods for introducing acetylenic linkages frequently encounter issues with homocoupling side reactions, leading to impurities that degrade the voltage holding ratio (VHR) of the final mixture. Furthermore, achieving specific lateral fluorination patterns on highly conjugated backbones can be synthetically demanding, often requiring harsh conditions that compromise the integrity of sensitive functional groups. These limitations result in lower overall yields and increased production costs, hindering the widespread adoption of high-performance monomers in cost-sensitive consumer electronics markets.
The Novel Approach
The methodology described in CN102557896A overcomes these hurdles through a modular, palladium-catalyzed cross-coupling strategy. By breaking the synthesis into distinct, high-yielding coupling events, the process minimizes side reactions and simplifies downstream purification. The use of protected alkyne intermediates, such as 3-methyl-1-butyn-3-ol, allows for controlled chain extension before revealing the reactive terminal alkyne functionality. This stepwise construction ensures that the delicate diacetylene core is formed under mild conditions, preserving the optical purity of the molecule. The approach is inherently scalable, utilizing robust catalytic systems that are well-understood in industrial organic synthesis, thereby facilitating cost reduction in display material manufacturing.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling
The synthetic pathway relies heavily on the efficiency of the Sonogashira coupling reaction, mediated by tetrakis(triphenylphosphine)palladium(0). This catalytic cycle facilitates the formation of carbon-carbon bonds between aryl halides and terminal alkynes with remarkable selectivity. The mechanism involves oxidative addition of the aryl iodide or bromide to the Pd(0) center, followed by transmetallation with the copper-acetylide species generated in situ. Reductive elimination then releases the coupled product and regenerates the active catalyst. In the context of this patent, the reaction conditions are optimized to tolerate the presence of multiple fluorine atoms, which can sometimes deactivate catalytic centers or lead to defluorination side products if not carefully managed.
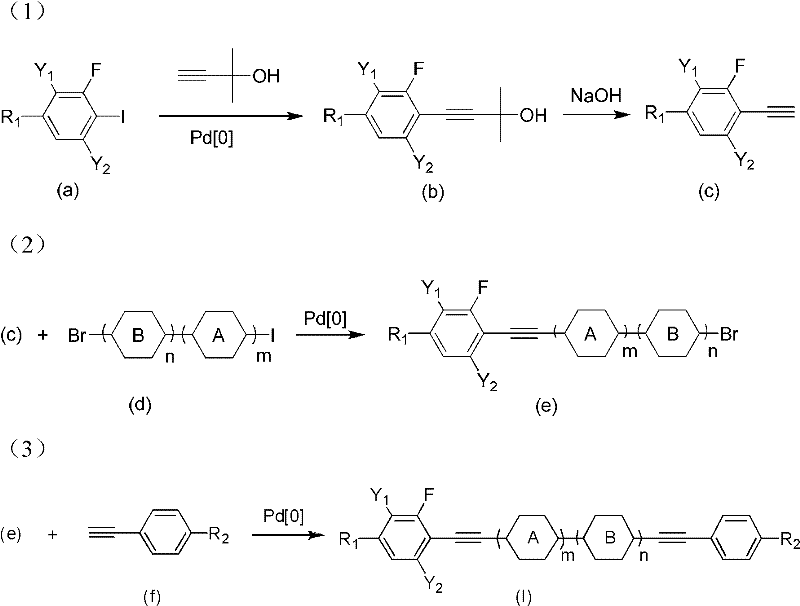
Impurity control is paramount in liquid crystal synthesis, as trace contaminants can severely impact the resistivity and reliability of the display panel. The patented process incorporates specific purification steps, including column chromatography and recrystallization from solvents like n-hexane or ethanol, to remove residual palladium and unreacted starting materials. The use of inert atmospheres, specifically high-purity argon, throughout the reaction sequence prevents oxidative degradation of the alkyne moieties. Additionally, the base-mediated elimination step to generate the terminal alkyne is conducted at elevated temperatures (100-110°C) to ensure complete conversion, minimizing the carryover of protected intermediates that could act as impurities in the final coupling stage.
How to Synthesize Fluorinated Phenyldiacetylene Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these high-value intermediates. It begins with the coupling of a fluorinated iodobenzene with a protected alkyne, followed by deprotection to yield a reactive terminal alkyne. This intermediate is then sequentially coupled with dihalogenated benzene cores and finally capped with a substituted phenylacetylene. The detailed standardized synthesis steps below outline the specific molar ratios, temperatures, and workup procedures required to achieve the reported high purity levels.
- Perform initial Sonogashira coupling of fluoro-iodobenzene with 3-methyl-1-butyn-3-ol, followed by base-mediated elimination to form the terminal alkyne intermediate.
- Execute a second cross-coupling reaction between the terminal alkyne and a bromo-iodobenzene derivative to extend the molecular core.
- Finalize the synthesis via a third coupling step with a substituted phenylacetylene to achieve the target diacetylene structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical performance. The reliance on commodity chemicals such as substituted iodobenzenes and phenylacetylenes ensures a stable supply chain with reduced risk of raw material bottlenecks. The modularity of the synthesis allows for flexible production scheduling, where different variants of the final liquid crystal can be produced using common intermediates, thereby optimizing inventory management and reducing warehousing costs associated with diverse SKU portfolios.
- Cost Reduction in Manufacturing: The streamlined three-step sequence significantly reduces the number of unit operations compared to traditional linear syntheses. By eliminating the need for exotic reagents and utilizing standard palladium catalysis, the process lowers the overall cost of goods sold. The high experimental yields reported in the patent examples, such as 86% in the second coupling step, indicate a material-efficient process that minimizes waste generation. This efficiency translates directly into improved margins for large-scale production runs without compromising on the stringent quality standards required for electronic grade chemicals.
- Enhanced Supply Chain Reliability: The use of robust reaction conditions, such as moderate temperatures ranging from 50°C to 110°C and common solvents like toluene and THF, ensures that the process can be easily transferred to multiple manufacturing sites. This geographic flexibility mitigates supply chain risks associated with single-source dependencies. Furthermore, the stability of the intermediates allows for potential stockpiling, enabling manufacturers to respond rapidly to fluctuating market demands for specific liquid crystal mixtures used in TV, monitor, and mobile display panels.
- Scalability and Environmental Compliance: The synthesis is designed with industrial scalability in mind, avoiding cryogenic conditions or high-pressure equipment that would limit batch sizes. The waste profile is manageable, primarily consisting of aqueous washes and spent organic solvents which can be recovered and recycled. This aligns with increasingly strict environmental regulations in the chemical sector. The ability to produce high-purity electronic chemical intermediates with a lower environmental footprint enhances the sustainability credentials of the final display products, a key consideration for major electronics brands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated diacetylene derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for R&D and procurement decision-making.
Q: What are the key optical properties of these fluorinated derivatives?
A: According to patent data, these compounds exhibit large optical anisotropy (Δn), suitable dielectric anisotropy (Δε), and high clearing points, making them ideal for tuning LCD mixtures.
Q: Is the synthesis route suitable for industrial scale-up?
A: Yes, the process utilizes standard Pd(0) catalysis and commercially available raw materials, with reported experimental yields reaching up to 86% in intermediate steps.
Q: How does fluorine substitution impact liquid crystal performance?
A: Lateral fluorine substitution increases molecular polarity and reduces assembly density, leading to lower viscosity and improved voltage holding ratios in display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Phenyldiacetylene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to mass market deployment is seamless. We understand the critical nature of stringent purity specifications in the display industry and operate rigorous QC labs equipped to detect trace impurities that could compromise device performance. Our commitment to quality assurance guarantees that every batch meets the exacting standards required for high-end LCD applications.
We invite you to collaborate with our technical procurement team to explore how these innovative liquid crystal intermediates can enhance your product portfolio. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to support your development timelines and secure your supply chain for the future of display technology.
