Revolutionizing Bibenzyl Production: A Green Photocatalytic One-Step Method for Commercial Scale-up
The chemical industry is currently witnessing a paradigm shift towards sustainable manufacturing, driven by stringent environmental regulations and the urgent need for cost-effective processes. Patent CN109896919B introduces a groundbreaking methodology for the synthesis of bibenzyl compounds, specifically targeting the production of 1,2-diphenylethane and its derivatives through a photocatalytic one-step method. This innovation represents a significant departure from legacy technologies, leveraging green light energy to drive dehydrogenative coupling reactions that were previously energy-intensive or chemically hazardous. By utilizing abundant toluene derivatives as feedstocks and employing a robust solid photocatalyst system, this technology addresses critical pain points in the supply chain of fine chemical intermediates. For R&D directors and procurement managers alike, the implications are profound, offering a pathway to high-purity products while drastically simplifying the downstream purification workflow. The ability to operate at room temperature without the need for aggressive reagents positions this patent as a cornerstone for the next generation of eco-friendly pharmaceutical and agrochemical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of bibenzyl compounds has relied heavily on Friedel-Crafts alkylation, a process fraught with significant operational and environmental challenges. As illustrated in the traditional reaction pathway below, the coupling of benzene with 1,2-dichloroethane requires stoichiometric amounts of anhydrous aluminum chloride (AlCl3), a highly corrosive Lewis acid that demands specialized reactor materials and rigorous safety protocols.

The generation of substantial quantities of hydrochloric acid as a byproduct necessitates complex waste treatment systems to meet modern emission standards, thereby inflating the overall cost of production. Furthermore, alternative routes involving ethylene oxide or benzyl chloride introduce additional risks, including the potential for explosive reactions and the use of expensive active metals like zinc or magnesium for reductive coupling. These conventional methods not only suffer from low atom economy but also create a heavy burden on supply chain logistics due to the handling and disposal of hazardous chemicals. The reliance on such toxic and corrosive reagents limits the scalability of these processes and increases the lead time for high-purity fine chemical intermediates, making them increasingly unviable in a market that prioritizes green chemistry principles.
The Novel Approach
In stark contrast to these archaic methods, the photocatalytic strategy outlined in CN109896919B offers a streamlined, environmentally benign alternative that transforms simple toluene derivatives directly into valuable bibenzyl structures. The core of this innovation lies in the use of visible light to drive the endothermic dehydrogenation and subsequent C-C bond formation, eliminating the need for thermal activation or harsh chemical oxidants. The general reaction scheme demonstrates the versatility of this approach, accommodating a wide range of substrates including substituted toluenes and heterocyclic derivatives.
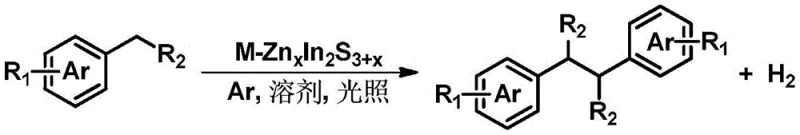
This novel approach operates under mild conditions, typically at room temperature and ambient pressure, which significantly reduces energy consumption and equipment stress. The use of a solid photocatalyst, such as the doped ternary sulfide system described in the patent, allows for easy separation from the reaction mixture via simple filtration or centrifugation, enabling catalyst recycling and minimizing waste generation. For procurement teams, this translates to a reliable bibenzyl compound supplier capability that is not constrained by the volatility of hazardous reagent markets. The process inherently produces hydrogen gas as a benign byproduct, which can potentially be captured and utilized for onsite heating, further enhancing the overall energy efficiency of the manufacturing facility. This shift from batch-wise corrosive chemistry to continuous flow photocatalysis marks a pivotal advancement in the commercial scale-up of complex polymer additives and pharmaceutical precursors.
Mechanistic Insights into Photocatalytic Dehydrogenative Coupling
The success of this synthetic route hinges on the sophisticated design of the photocatalyst, specifically the M-ZnxIn2S3+x system where M represents a transition metal dopant such as Ruthenium, Palladium, or Nickel. The mechanism involves the absorption of photons by the semiconductor catalyst, which generates electron-hole pairs capable of driving the oxidation of the methyl group on the toluene derivative. This photo-induced oxidation facilitates the removal of hydrogen atoms, creating reactive radical intermediates that subsequently undergo homocoupling to form the stable bibenzyl backbone. The precise tuning of the bandgap through metal doping is critical, as it ensures that the catalyst can effectively utilize visible light sources, such as LEDs or xenon lamps, rather than requiring high-energy UV radiation. This optimization not only improves the quantum yield of the reaction but also extends the operational lifetime of the catalyst by preventing rapid deactivation. Understanding these mechanistic nuances is essential for R&D directors aiming to replicate or adapt this technology for specific API intermediate applications, as the electronic properties of the catalyst directly influence the selectivity and conversion rates.
Furthermore, the control of impurities in this photocatalytic system is achieved through the careful selection of reaction parameters, including light intensity, solvent polarity, and substrate concentration. The patent data indicates that while the reaction is a series process where over-reaction can occur, maintaining optimal illumination times and using non-polar solvents like cyclohexane can significantly enhance the selectivity towards the desired bibenzyl product. The solid nature of the catalyst prevents the leaching of metal ions into the product stream, a common issue in homogeneous catalysis that often necessitates expensive purification steps to meet stringent pharmaceutical purity specifications. By avoiding the formation of chlorinated byproducts associated with Friedel-Crafts chemistry, this method inherently produces a cleaner crude product profile, simplifying the crystallization process. This level of impurity control is paramount for manufacturers supplying the electronics or healthcare sectors, where trace contaminants can compromise the performance of the final application. The robustness of the catalyst against deactivation ensures consistent batch-to-batch quality, a key metric for supply chain reliability.
How to Synthesize 1,2-Diphenylethane Efficiently
Implementing this photocatalytic protocol requires a systematic approach to reactor setup and parameter optimization to maximize yield and throughput. The process begins with the preparation of the doped sulfide catalyst, followed by the formulation of the reaction mixture in a quartz vessel that allows for efficient light transmission. Operators must ensure an inert atmosphere is maintained throughout the reaction to prevent oxidative side reactions that could degrade the product or the catalyst. While the fundamental chemistry is straightforward, scaling this process from laboratory glassware to industrial photoreactors involves engineering considerations regarding light penetration and mixing efficiency. The following guide outlines the standardized procedure derived from the patent examples, providing a foundational framework for process engineers to adapt this technology for commercial production.
- Mix toluene derivatives with a solid photocatalyst (e.g., Ru-ZnxIn2S3+x) and a solvent like acetonitrile or cyclohexane in a quartz vessel.
- Replace the atmosphere with inert gas (Argon or Nitrogen) to ensure an oxygen-free environment essential for dehydrogenative coupling.
- Irradiate the mixture with a light source (LED or Xenon lamp) at room temperature for 1 to 120 hours, then separate the solid catalyst and purify the product via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers transformative benefits that extend far beyond simple chemical synthesis. The primary advantage lies in the drastic reduction of raw material costs, as the process utilizes toluene, a commodity chemical with a stable and abundant global supply, rather than expensive and regulated precursors like benzyl chloride. This shift in feedstock strategy mitigates the risk of supply disruptions and price volatility, ensuring a more predictable cost structure for long-term contracts. Additionally, the elimination of corrosive catalysts and hazardous waste streams significantly lowers the operational expenditure associated with waste disposal and equipment maintenance. Facilities can operate with standard stainless steel or glass-lined reactors without the need for exotic alloys resistant to hydrochloric acid, resulting in substantial capital expenditure savings. The simplicity of the workup procedure, which relies on distillation and crystallization rather than complex aqueous extractions, further accelerates the production cycle, reducing lead time for high-purity fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of low-cost starting materials and the recyclability of the solid photocatalyst. Unlike traditional methods that consume stoichiometric amounts of expensive Lewis acids or active metals, this system allows the catalyst to be recovered and reused multiple times without significant loss of activity. This circular approach to catalysis minimizes the consumption of precious metals and reduces the overall material cost per kilogram of product. Furthermore, the avoidance of hazardous waste treatment fees and the potential utilization of byproduct hydrogen for energy recovery contribute to a leaner manufacturing model. By streamlining the synthesis into a single step under mild conditions, the process reduces energy consumption related to heating and cooling, leading to significant cost reduction in fine chemical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The reliance on toluene derivatives as feedstocks ensures a robust supply chain, as these materials are produced on a massive scale for various industries and are readily available from multiple vendors. This diversification of supply sources reduces dependency on single suppliers of niche reagents, thereby enhancing the resilience of the procurement strategy. The mild reaction conditions also mean that production is less susceptible to interruptions caused by utility failures or safety incidents associated with high-pressure or high-temperature operations. The ability to store the solid catalyst for extended periods without degradation adds another layer of security to the inventory management process. Consequently, manufacturers can guarantee consistent delivery schedules and maintain buffer stocks more effectively, serving as a reliable agrochemical intermediate supplier or pharma partner.
- Scalability and Environmental Compliance: Scaling this technology to industrial levels is facilitated by the modular nature of photocatalytic reactors, which can be arranged in parallel arrays to increase capacity without compromising reaction efficiency. The absence of toxic emissions and corrosive effluents simplifies the permitting process and ensures compliance with increasingly strict environmental regulations. This green credential is not only a regulatory necessity but also a competitive advantage in markets where end-users prioritize sustainability in their sourcing decisions. The ease of separating the solid catalyst from the liquid product stream allows for continuous processing configurations, which are ideal for large-volume production runs. This scalability ensures that the technology can meet the growing demand for bibenzyl-based flame retardants and pharmaceutical building blocks without the bottlenecks typical of batch-wise corrosive chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios. The answers highlight the practical aspects of catalyst handling, reaction optimization, and product isolation, ensuring that both technical and non-technical decision-makers have a comprehensive understanding of the process capabilities.
Q: What are the primary advantages of this photocatalytic method over traditional Friedel-Crafts alkylation?
A: Unlike traditional methods requiring corrosive AlCl3 and generating hazardous waste acid, this photocatalytic route operates at room temperature with a recyclable solid catalyst, significantly reducing environmental impact and equipment corrosion costs.
Q: Which catalyst system is utilized for the dehydrogenative coupling of toluene derivatives?
A: The process utilizes a ternary sulfide photocatalyst doped with transition metals, specifically M-ZnxIn2S3+x (where M can be Ru, Pd, Pt, Au, or Ni), which facilitates efficient hydrogen evolution and C-C bond formation under visible light.
Q: Is this synthesis method suitable for large-scale industrial production of flame retardant intermediates?
A: Yes, the method is highly scalable due to its mild reaction conditions (room temperature and pressure) and the ease of separating the solid photocatalyst from the liquid reaction mixture, allowing for continuous operation and reduced downtime.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bibenzyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis route described in CN109896919B and are fully equipped to bring this technology to commercial fruition. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand that the successful deployment of photocatalytic processes requires specialized engineering expertise, particularly in the design of illuminated reactor systems that maximize photon efficiency. Our state-of-the-art facilities are designed to handle sensitive photochemical reactions with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. Whether you require 1,2-diphenylethane for flame retardant applications or substituted bibenzyls for pharmaceutical synthesis, our CDMO capabilities are tailored to meet your specific volume and purity requirements.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing footprint through this innovative green chemistry solution. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching from traditional Friedel-Crafts alkylation to this photocatalytic method. We encourage potential partners to contact us to request specific COA data and route feasibility assessments for your target molecules. By leveraging our expertise in process development and scale-up, you can secure a sustainable and cost-effective supply of critical intermediates, positioning your organization at the forefront of the green chemical revolution.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
