Advanced Synthesis of 17-Position Steroid Carboxylates for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for glucocorticoids, a class of steroidal drugs essential for treating inflammatory conditions, allergies, and autoimmune disorders. Patent CN102964414A introduces a transformative synthesis method for 17-position steroid carboxylates, addressing a critical bottleneck in the production of high-purity corticosteroid intermediates. This technology focuses on the selective protection and subsequent deprotection of the 17-hydroxyl group in 17,21-dihydroxy corticosteroids, a structural motif found in blockbuster drugs like fluticasone propionate and methylprednisolone. The core innovation lies in the hydrolysis of cyclic orthoester intermediates using specific strong acid weak base salt catalysts. This approach fundamentally alters the reaction landscape by minimizing the formation of unwanted 21-position esterified by-products, which have historically plagued manufacturers with low yields and complex purification challenges. By leveraging this patented methodology, producers can achieve superior impurity profiles and streamlined processing, positioning this technique as a vital asset for any reliable pharmaceutical intermediate supplier aiming to optimize their API manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating 17-position esterified steroids typically involve a two-step sequence: formation of a cyclic orthoester followed by acid-catalyzed ring opening. However, the second step, hydrolysis, presents severe chemoselectivity issues. Under standard strong acid catalysis conditions, the reaction lacks the precision to distinguish effectively between the 17-hydroxyl and 21-hydroxyl groups. Consequently, a significant proportion of the starting material converts into the undesired 21-position esterified isomer rather than the target 17-position product. Patent data indicates that in conventional processes, the ratio of this specific by-product can range drastically from 20% to even 100% relative to the desired product. This high level of impurity generation necessitates rigorous and often yield-depleting purification steps, such as multiple recrystallizations, because the solubility profiles of the isomers are frequently too similar for simple separation. Furthermore, conventional methods demand stringent reaction monitoring to halt the process immediately upon reaching the endpoint, as prolonged exposure to harsh acidic conditions exacerbates by-product formation, creating operational risks and batch-to-batch variability that supply chain managers find unacceptable.
The Novel Approach
The methodology described in CN102964414A offers a sophisticated solution by replacing traditional strong mineral acids with strong acid weak base salts as catalysts. This subtle yet powerful change in the catalytic system dramatically shifts the selectivity profile of the hydrolysis reaction. Instead of generating overwhelming amounts of the 21-esterified impurity, the new process reduces this by-product ratio to less than 5%, thereby significantly enhancing the crude purity of the 17-position steroid carboxylate. The reaction operates efficiently in a biphasic system comprising a Class A solvent (like dichloromethane or ethyl acetate) and a Class B co-solvent (such as methanol or tetrahydrofuran) with added water. This setup not only facilitates the hydrolysis but also simplifies the post-reaction workup; since the catalysts are highly water-soluble salts, they can be effortlessly removed via a simple aqueous wash, eliminating the need for complex neutralization or extraction procedures associated with liquid acids. This robustness allows for more flexible reaction windows where extending time or slightly adjusting temperature does not compromise product integrity, representing a major leap forward in cost reduction in pharmaceutical intermediate manufacturing.
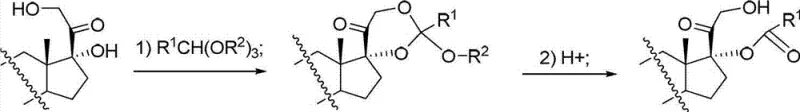
Mechanistic Insights into Salt-Catalyzed Selective Hydrolysis
The mechanistic advantage of using strong acid weak base salts, such as sodium dihydrogen phosphate (NaH2PO4) or ammonium chloride (NH4Cl), stems from their ability to provide a buffered acidic environment. Unlike free strong acids which create a high concentration of protons that indiscriminately attack various nucleophilic sites, these salts offer a controlled proton source that favors the specific cleavage of the cyclic orthoester bond leading to the 17-ester. The steric and electronic environment of the steroid backbone, particularly around the C17 and C21 positions, is finely tuned by this catalytic system. The cyclic orthoester intermediate, formed initially from the reaction of the 17,21-dihydroxy steroid with an orthoester reagent (e.g., triethyl orthopropionate), possesses a specific conformation. The salt catalyst promotes the hydrolytic opening of this ring in a manner that kinetically favors the retention of the ester group at the C17 position while releasing the C21 hydroxyl group as a free alcohol. This selectivity is crucial because the 21-hydroxyl is often required for subsequent biological activity or further derivatization in the drug synthesis pathway. By suppressing the migration of the acyl group to the C21 oxygen, the process ensures that the molecular architecture remains aligned with the requirements for high-purity API production.
Furthermore, the impurity control mechanism is inherently linked to the solubility and phase behavior of the reaction components. The use of a biphasic solvent system creates a microenvironment where the hydrolysis occurs at the interface or within the organic phase, modulated by the partitioning of the catalyst ions. This physical arrangement helps to sequester the generated alcohol by-products and prevents reverse reactions or secondary esterifications that could lead to the 21-isomer. The patent highlights that even with extended reaction times, the formation of further hydrolysis by-products is negligible, suggesting that the reaction reaches a thermodynamic or kinetic plateau favorable to the target molecule. This stability is a key differentiator for R&D directors evaluating process robustness, as it reduces the risk of batch failure due to minor deviations in process parameters. The result is a cleaner reaction profile where the primary impurity (the 21-ester) is minimized to levels that often allow for direct isolation of the product via concentration and precipitation, bypassing the need for chromatographic purification or extensive recrystallization campaigns.
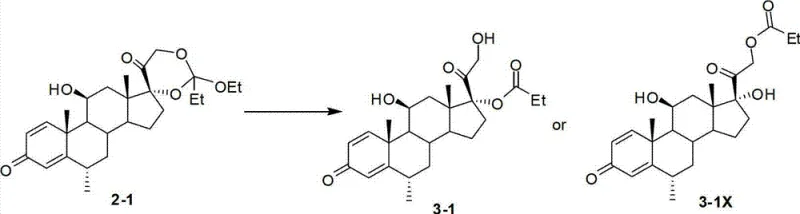
How to Synthesize 17-Position Steroid Carboxylates Efficiently
The synthesis protocol outlined in the patent provides a clear and scalable pathway for converting cyclic orthoester intermediates into the desired 17-position carboxylates. The process begins by dissolving the intermediate in a suitable organic solvent, followed by the addition of water and a co-solvent to ensure homogeneity and facilitate mass transfer. The choice of catalyst is critical, with options ranging from phosphates to ammonium salts, allowing flexibility based on availability and specific substrate requirements. The reaction proceeds under mild thermal conditions, typically between -10°C and 25°C, making it energy-efficient and safe for large-scale operations. Once the hydrolysis is complete, as monitored by TLC or HPLC, the workup is remarkably straightforward: the phases are separated, and the organic layer is washed with water to remove the spent catalyst. The product is then isolated by concentrating the organic phase and inducing precipitation with a non-solvent like petroleum ether. For a detailed breakdown of the specific molar ratios, solvent volumes, and step-by-step operational parameters required to replicate this high-yield process, please refer to the standardized guide below.
- Dissolve the cyclic orthoester intermediate in a Class A solvent such as dichloromethane or ethyl acetate.
- Add water and a Class B co-solvent like methanol or tetrahydrofuran to create a biphasic reaction system.
- Introduce a strong acid weak base salt catalyst such as sodium dihydrogen phosphate and react between -10°C and 25°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible operational efficiencies and risk mitigation. The primary economic driver is the drastic reduction in by-product formation, which directly correlates to higher overall yields and reduced raw material consumption. In conventional processing, the need to discard or reprocess batches contaminated with high levels of 21-esterified impurities represents a significant hidden cost. By minimizing these impurities to less than 5%, the new method ensures that a greater proportion of the expensive steroid starting material is converted into saleable product. Additionally, the simplification of the purification process eliminates the need for resource-intensive recrystallization steps or chromatography, which are often bottlenecks in manufacturing facilities. This streamlining not only accelerates the production cycle but also reduces the consumption of solvents and energy, contributing to a lower carbon footprint and compliance with increasingly strict environmental regulations. The use of inexpensive, commercially available salt catalysts further enhances the cost-effectiveness of the route, avoiding the need for specialized or hazardous reagents that complicate logistics and storage.
- Cost Reduction in Manufacturing: The elimination of complex purification steps such as repeated recrystallization significantly lowers processing costs. Since the by-product ratio is suppressed to minimal levels, the crude product often meets purity specifications after simple washing and precipitation, saving substantial amounts of solvent and labor hours associated with polishing steps. This efficiency gain allows for a more competitive pricing structure for the final intermediate without compromising margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reaction time, reducing the likelihood of batch failures. The catalysts used are stable, non-hazardous salts that are easy to source globally, ensuring that supply chain disruptions due to reagent scarcity are minimized. This reliability is crucial for maintaining consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The biphasic nature of the reaction and the water-solubility of the catalyst facilitate easy scale-up from laboratory to commercial production. The aqueous waste stream containing the catalyst is easier to treat compared to waste streams laden with strong mineral acids or organic bases. This aligns with green chemistry principles, making the process more sustainable and easier to permit in regions with stringent environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 17-position steroid carboxylate synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of catalyst selection, impurity profiles, and operational flexibility is essential for teams evaluating this route for integration into their existing manufacturing portfolios. The answers below clarify how this method outperforms legacy technologies in terms of selectivity and ease of handling.
Q: How does this method improve selectivity compared to conventional acid catalysis?
A: Conventional strong acid catalysis often leads to significant 21-position esterification by-products (20-100%). This novel method uses strong acid weak base salts which significantly reduce this ratio to below 5%, enhancing purity.
Q: What catalysts are suitable for this hydrolysis reaction?
A: The process utilizes water-soluble strong acid weak base salts including sodium dihydrogen phosphate, potassium dihydrogen phosphate, ammonium chloride, and ammonium sulfate, which are easily removed via aqueous washing.
Q: Does this process require strict temperature control to prevent side reactions?
A: Unlike conventional methods requiring immediate stopping at the endpoint, this process is robust; extending reaction time or moderately increasing temperature does not generate further hydrolysis by-products, simplifying operational control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17-Position Steroid Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain. Our technical team has extensively analyzed advanced synthetic routes like the one described in CN102964414A to ensure we can deliver superior materials. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel catalytic hydrolysis method can be realized at an industrial scale. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities, ensuring that every batch of 17-position steroid carboxylate meets stringent purity specifications required by global regulatory bodies. We are committed to bridging the gap between innovative patent chemistry and commercial reality, offering our partners a secure and efficient source for complex steroid intermediates.
We invite procurement leaders and R&D directors to engage with us to explore how this technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this selective hydrolysis process can enhance your production efficiency and reduce overall manufacturing costs.
