Advanced One-Pot Synthesis of p-Phthaloquinone Compounds for Industrial Scale-Up
Advanced One-Pot Synthesis of p-Phthaloquinone Compounds for Industrial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, more efficient synthetic routes that do not compromise on yield or purity. A significant breakthrough in this domain is detailed in patent CN107365243B, which discloses a novel one-pot method for synthesizing p-phthaloquinone compounds. These compounds serve as critical building blocks in the production of essential vitamins such as Vitamin K and E, as well as in the formulation of dyes, photochemical agents, and antioxidants for the rubber industry. The traditional reliance on harsh oxidants and multi-step processes has long been a bottleneck for reliable pharmaceutical intermediates supplier networks seeking to optimize their supply chains. This new methodology leverages an innovative catalytic system involving iodide salts and potassium hydrogen persulfate (Oxone) to achieve transformation under remarkably mild conditions, setting a new benchmark for cost reduction in fine chemical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of phenols or hydroquinones to generate p-phthaloquinones has relied heavily on stoichiometric amounts of aggressive oxidizing agents. Literature and prior art frequently cite the use of Fremy's salt, dichromates, silver oxides, ceric salts, and hypervalent iodine compounds. While chemically effective, these reagents present severe drawbacks for modern industrial application. Firstly, the requirement for greater than equivalent amounts of these oxidants drives up raw material costs exponentially. Secondly, the environmental footprint is substantial; heavy metal residues from chromium or silver necessitate complex and expensive waste treatment protocols to meet stringent environmental regulations. Furthermore, alternative methods utilizing air or oxygen often require heavy metal halide catalysts and high-boiling solvents like heptanol or decanol. The separation of products from these high-boiling solvents often demands heating and reduced pressure, which poses a risk of product loss due to the sublimation tendency of p-phthaloquinone compounds, thereby reducing overall isolation yields.
The Novel Approach
In stark contrast, the methodology outlined in CN107365243B introduces a paradigm shift by employing a one-pot strategy that generates the active catalytic species in situ. Instead of purchasing expensive pre-formed catalysts or oxidants, the process utilizes simple aromatic compounds and iodide salts which react with Oxone to form the active iodobenzene catalyst within the reaction vessel. This approach not only simplifies the operational workflow but also drastically reduces the inventory of hazardous chemicals required on-site. The reaction proceeds in a mixed solvent system of water and inert organic solvents like acetonitrile, which are far easier to recover and recycle compared to the long-chain alcohols used in older methods. By operating at temperatures between 0°C and 60°C, the process mitigates thermal risks associated with exothermic oxidations, ensuring a safer working environment while maintaining high reaction efficiency. This represents a significant leap forward for any organization aiming to become a reliable fine chemical intermediates supplier in a regulated market.
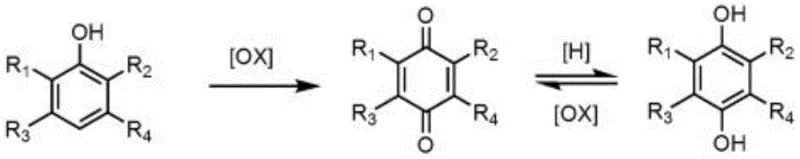
Mechanistic Insights into Oxone-Mediated Iodide Catalysis
The core innovation of this technology lies in the elegant interplay between the iodide salt and the terminal oxidant, Oxone. Mechanistically, the process begins with the oxidation of the iodide salt by Oxone in the presence of an aromatic compound (such as benzene or anisole) to generate an iodobenzene derivative. This species acts as a redox mediator, shuttling electrons between the terminal oxidant and the phenolic substrate. The mild nature of this catalytic cycle is crucial for impurity control. In traditional strong oxidations, over-oxidation can lead to ring cleavage or the formation of polymeric tars, which are notoriously difficult to remove and can severely impact the color and stability of the final API intermediate. By maintaining the reaction temperature strictly between 0°C and 60°C during the phenol addition phase, the kinetic energy of the system is controlled, favoring the selective formation of the quinone carbonyl groups without degrading the aromatic ring structure.
Furthermore, the workup procedure described in the patent is specifically designed to maximize purity. After the reaction reaches completion, monitored by GC chromatography, the mixture is cooled and filtered to remove inorganic salts. The subsequent liquid-liquid extraction involves washing the organic phase with an alkaline solution until the pH exceeds 7. This step is critical for neutralizing any acidic byproducts or residual Oxone, ensuring that the final organic layer contains minimal ionic contaminants. The use of a second solvent like ethyl acetate for dissolution and final drying allows for the crystallization of the product in high purity forms, often exceeding 98% GC purity as demonstrated in the examples. This level of control over the impurity profile is essential for high-purity pharmaceutical intermediates destined for sensitive downstream applications like vitamin synthesis.
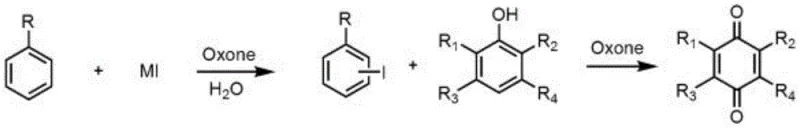
How to Synthesize p-Phthaloquinone Compounds Efficiently
Implementing this synthesis route requires precise control over reagent addition and temperature profiling to ensure the in-situ generation of the catalyst precedes the oxidation of the phenol. The protocol dictates a sequential addition strategy where the aromatic precursor and iodide source are activated first, creating a robust catalytic environment before the introduction of the valuable phenolic substrate. This minimizes side reactions and ensures that the oxidant power is directed efficiently towards the desired transformation. For process chemists looking to adopt this technology, the following guide outlines the standardized operational parameters derived from the patent data to facilitate successful scale-up.
- Prepare the reaction vessel by adding the first solvent (water and inert organic solvent mixture), aromatic compound, iodide salt, and potassium hydrogen persulfate (Oxone).
- Stir the mixture at 0-100°C until the aromatic compound is completely converted to the iodobenzene intermediate, monitored via GC chromatography.
- Add the phenolic compound at 0-60°C, stir until complete conversion, then cool, filter, separate layers, wash with alkali, dry, and concentrate to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this one-pot synthesis method offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the drastic simplification of the raw material portfolio. By replacing exotic, high-cost oxidants like Fremy's salt or silver carbonate with commodity chemicals like sodium iodide and Oxone, the direct material cost is significantly reduced. Moreover, the elimination of heavy metals from the process stream removes the need for specialized scavenging resins or complex wastewater treatment phases dedicated to heavy metal removal. This qualitative shift in process chemistry translates directly into lower operational expenditures and a reduced regulatory burden, facilitating smoother audits and faster time-to-market for new product launches.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the use of catalytic rather than stoichiometric amounts of iodine species, generated from inexpensive salts. Traditional methods often suffer from poor atom economy due to the heavy molecular weight of oxidants like dichromates relative to the oxygen atoms transferred. In this new route, the oxidant (Oxone) is cheap and highly efficient, and the solvent system (acetonitrile/water) allows for easier recovery and recycling compared to high-boiling alcohols. The avoidance of high-temperature reflux conditions also results in substantial energy savings, as the reaction can proceed effectively at moderate temperatures, reducing the load on heating and cooling infrastructure.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability of starting materials. The reagents required for this synthesis—benzene, toluene, anisole, sodium iodide, and Oxone—are bulk commodities produced globally in massive quantities. This stands in contrast to specialized oxidants which may have limited suppliers and long lead times. By anchoring the production process to widely available feedstocks, manufacturers can mitigate the risk of supply disruptions caused by geopolitical issues or single-source vendor failures. This ensures a consistent flow of high-purity pharmaceutical intermediates to downstream customers, reinforcing trust and long-term partnership stability.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden hazards, particularly with exothermic oxidations. This method's ability to operate safely at temperatures as low as 0-60°C makes it inherently safer for large-scale reactors where heat dissipation is a challenge. The absence of explosive high-concentration hydrogen peroxide requirements further de-risks the operation. From an environmental standpoint, the aqueous workup and the ability to treat waste streams with simple reducing agents like sodium bisulfite ensure that effluent meets discharge standards with minimal processing. This aligns perfectly with global trends towards green chemistry and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their production schedules. Understanding these nuances is key to making informed decisions about process adoption and capacity planning.
Q: What are the primary advantages of this one-pot method over traditional oxidation routes?
A: This method eliminates the need for expensive and toxic stoichiometric oxidants like Fremy's salt or heavy metal salts (chromium, silver). It utilizes cheap Oxone and catalytic iodide, operates under mild conditions (0-60°C), and achieves high yields (≥92%) with purity >98%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids hazardous high-concentration hydrogen peroxide usage at reflux temperatures and uses common solvents like acetonitrile and ethyl acetate, making waste treatment and safety management significantly easier for commercial plants.
Q: What types of substrates can be used in this catalytic system?
A: The system is versatile, accommodating various aromatic compounds (benzene, toluene, anisole) to generate the catalyst in situ, and a wide range of phenolic compounds with different substituents (alkyl, alkoxy, halogen) to produce diverse p-phthaloquinone derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Phthaloquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one described in CN107365243B requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facilities are equipped to handle the specific solvent systems and temperature controls required for this Oxone-mediated oxidation, guaranteeing that every batch meets stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity pharmaceutical intermediates that empower your R&D and production teams to innovate without supply constraints.
We invite you to explore how this optimized synthesis route can transform your cost structure and supply reliability. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to secure your supply chain with a solution that combines cutting-edge chemistry with commercial pragmatism, ensuring you stay ahead in the competitive landscape of fine chemical manufacturing.
