Advanced Transketalization Technology for Commercial Scale-Up of Complex Steroid Intermediates
Introduction to Advanced Steroid Synthesis via Patent CN1473164A
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of high-value glucocorticoid intermediates, which serve as critical building blocks for anti-inflammatory and immunosuppressive therapies. Patent CN1473164A introduces a transformative process for the preparation of 16,17-[(cyclohexylmethylene)bis(oxy)]-11,21-dihydroxy-pregna-1,4-diene-3,20-dione and its derivatives through a sophisticated transketalization reaction. This technology addresses long-standing challenges in steroid chemistry, particularly regarding stereochemical control at the C-22 position, which is pivotal for the biological activity and safety profile of the final Active Pharmaceutical Ingredient (API). By leveraging specific acid-catalyzed conditions, this method achieves exceptional purity levels of the desired R-epimer, streamlining the downstream purification processes that typically burden manufacturing costs. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for evaluating potential supply chain optimizations and ensuring the consistent quality of raw materials entering the production line.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating 16,17-protected glucocorticoid derivatives often rely on direct esterification or the manipulation of 16,17-dihydroxy compounds, which can be fraught with inefficiencies and selectivity issues. Conventional methods frequently result in complex mixtures of epimers at the C-22 position, necessitating rigorous and costly separation techniques such as preparative high-performance liquid chromatography (HPLC) to isolate the biologically active isomer. Furthermore, the use of sensitive protecting groups in older methodologies often requires harsh deprotection conditions that can degrade the steroid nucleus or lead to unwanted side reactions, thereby reducing overall yield and increasing the impurity load. These limitations not only escalate the cost of goods sold (COGS) due to excessive solvent and resin consumption but also introduce significant variability in batch-to-batch consistency, posing risks for regulatory compliance and supply chain reliability in the competitive generic drug market.
The Novel Approach
In stark contrast, the novel approach detailed in CN1473164A utilizes a transketalization strategy starting from stable 16,17-ketal precursors, such as acetonide derivatives, reacting directly with cyclohexanecarbaldehyde. This method elegantly bypasses the need for unstable diester intermediates, offering a more direct and atom-economical pathway to the target molecule. The reaction is driven by thermodynamic control under mild acidic conditions, which inherently favors the formation of the sterically less hindered and thermodynamically stable R-epimer. 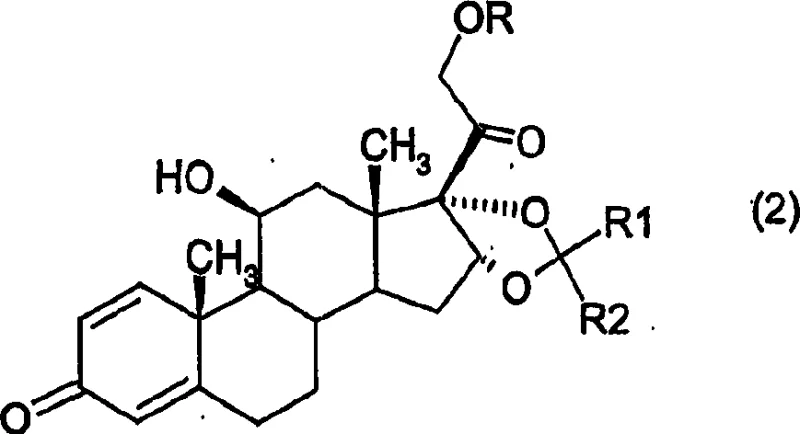 As illustrated in the reaction scheme, the transformation preserves the integrity of the sensitive steroid backbone while efficiently installing the bulky cyclohexylmethylene protecting group. This strategic shift in synthetic design allows manufacturers to achieve high conversion rates with minimal byproduct formation, significantly simplifying the isolation procedure to basic filtration and neutralization steps, thus representing a substantial leap forward in process chemistry efficiency.
As illustrated in the reaction scheme, the transformation preserves the integrity of the sensitive steroid backbone while efficiently installing the bulky cyclohexylmethylene protecting group. This strategic shift in synthetic design allows manufacturers to achieve high conversion rates with minimal byproduct formation, significantly simplifying the isolation procedure to basic filtration and neutralization steps, thus representing a substantial leap forward in process chemistry efficiency.
Mechanistic Insights into Acid-Catalyzed Transketalization
The core of this technological breakthrough lies in the precise mechanistic control of the acid-catalyzed exchange of ketal groups. The reaction initiates with the protonation of the oxygen atoms in the starting 16,17-ketal by a strong acid catalyst, such as perchloric acid or methanesulfonic acid, rendering the carbon center highly electrophilic. Subsequent nucleophilic attack by cyclohexanecarbaldehyde, followed by the elimination of the original ketal moiety (e.g., acetone), drives the equilibrium toward the formation of the new cyclohexylmethylene ketal. The choice of solvent plays a critical role in this mechanism; polar aprotic solvents like nitromethane or 1-nitropropane stabilize the charged intermediates and transition states, facilitating the reaction kinetics without participating in side reactions. This delicate balance of acidity and solvation ensures that the reaction proceeds smoothly at temperatures ranging from 0°C to 60°C, preventing thermal degradation of the sensitive enone system present in the A-ring of the steroid structure.
Furthermore, the stereoselectivity observed in this process is a result of the specific conformational preferences imposed by the bulky cyclohexyl ring during the ketalization event. 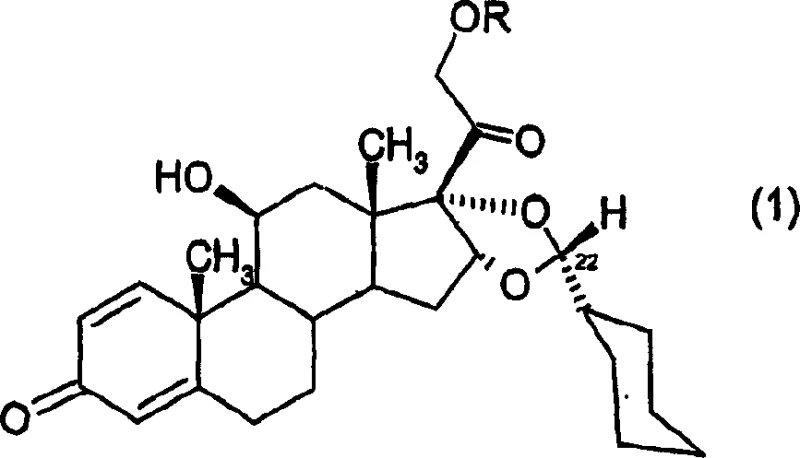 As depicted in the product structure, the configuration at the C-22 position is strictly controlled to favor the R-epimer, which is the desired stereochemistry for subsequent biological activity. The patent data indicates that under optimized conditions, the ratio of the R-epimer to the S-epimer can exceed 97:3, demonstrating an extraordinary level of diastereocontrol. This high selectivity minimizes the presence of the undesired S-epimer, which acts as a difficult-to-remove impurity in conventional syntheses. By effectively suppressing the formation of this isomer through kinetic and thermodynamic tuning of the reaction parameters, the process ensures that the final crude product meets stringent purity specifications, drastically reducing the burden on downstream purification units and enhancing the overall throughput of the manufacturing facility.
As depicted in the product structure, the configuration at the C-22 position is strictly controlled to favor the R-epimer, which is the desired stereochemistry for subsequent biological activity. The patent data indicates that under optimized conditions, the ratio of the R-epimer to the S-epimer can exceed 97:3, demonstrating an extraordinary level of diastereocontrol. This high selectivity minimizes the presence of the undesired S-epimer, which acts as a difficult-to-remove impurity in conventional syntheses. By effectively suppressing the formation of this isomer through kinetic and thermodynamic tuning of the reaction parameters, the process ensures that the final crude product meets stringent purity specifications, drastically reducing the burden on downstream purification units and enhancing the overall throughput of the manufacturing facility.
How to Synthesize 16,17-Cyclohexylmethylene Ketal Steroids Efficiently
Implementing this synthesis requires careful attention to reagent quality and reaction parameters to replicate the high success rates reported in the patent literature. The process begins by suspending the 16-hydroxyprednisolone ketal starting material in a selected solvent, with 1-nitropropane being the preferred medium due to its ability to solubilize both the steroid substrate and the aldehyde reagent effectively. The addition of the acid catalyst must be performed under controlled cooling, typically using an ice bath, to manage the exotherm associated with the protonation step and to prevent localized overheating that could lead to decomposition. Following the addition of cyclohexanecarbaldehyde, the reaction mixture is allowed to stir at ambient temperature, often overnight, to ensure complete conversion of the starting material. The workup involves a straightforward neutralization with a bicarbonate solution, precipitating the product which is then collected by filtration, washed, and dried to yield the high-purity target compound ready for the next synthetic step.
- Suspend the 16,17-ketal starting material (e.g., acetonide derivative) in a suitable solvent such as 1-nitropropane or dichloromethane under cooling conditions.
- Add cyclohexanecarbaldehyde and a catalytic amount of strong acid catalyst, specifically perchloric acid or methanesulfonic acid, while maintaining temperature control between 0°C and 60°C.
- Stir the reaction mixture to completion, followed by filtration and neutralization workup to isolate the pure R-epimer product with high stereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transketalization technology offers compelling economic and operational benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification workflow; because the reaction inherently produces the desired R-epimer with high selectivity, the need for expensive and time-consuming chromatographic separations is virtually eliminated. This reduction in processing complexity translates directly into lower operational expenditures, as it decreases the consumption of high-grade solvents, silica gel, and specialized equipment maintenance. Moreover, the use of robust and commercially available reagents like cyclohexanecarbaldehyde ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized intermediates that often plague specialty chemical manufacturing. The ability to produce high-purity intermediates consistently enhances supply chain reliability, allowing for tighter inventory management and reduced safety stock requirements.
- Cost Reduction in Manufacturing: The elimination of complex purification steps such as preparative HPLC results in substantial cost savings by reducing solvent usage and processing time. The high yield and selectivity mean that less starting material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final intermediate. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further contributing to a leaner manufacturing cost structure that improves margin potential for generic drug producers.
- Enhanced Supply Chain Reliability: By utilizing common industrial solvents and catalysts, the process reduces dependency on single-source suppliers for specialized reagents. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor variations in temperature or reagent grade compared to more fragile enzymatic or organometallic routes. This resilience ensures continuous production capability, minimizing the risk of stockouts and enabling manufacturers to respond more agilely to fluctuations in market demand for glucocorticoid APIs.
- Scalability and Environmental Compliance: The straightforward workup procedure involving filtration and aqueous washes generates less hazardous waste compared to processes requiring extensive organic extractions or column chromatography. This aligns with modern green chemistry principles and simplifies environmental compliance, reducing the costs associated with waste disposal and treatment. The process is inherently scalable from laboratory to multi-ton production without significant re-engineering, facilitating rapid technology transfer and commercial scale-up of complex pharmaceutical intermediates to meet global healthcare needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transketalization process, derived directly from the patent specifications and practical manufacturing considerations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The answers provided reflect the specific advantages of the acid-catalyzed method over traditional alternatives, focusing on purity, scalability, and operational safety.
Q: What is the primary advantage of using transketalization over direct esterification for this steroid intermediate?
A: The transketalization approach described in CN1473164A offers significantly higher stereoselectivity for the desired R-epimer at the C-22 position, often exceeding 95% purity without extensive chromatographic purification, thereby reducing processing time and solvent waste compared to traditional esterification routes.
Q: Which catalysts are preferred for maximizing the yield of the R-epimer?
A: The patent specifies that strong mineral acids such as perchloric acid (35-70% concentration) or sulfonic acids like methanesulfonic acid are highly effective. Perchloric acid at concentrations around 60-70% combined with nitroalkane solvents provides optimal conditions for driving the equilibrium toward the thermodynamically stable R-epimer.
Q: Is this process scalable for industrial manufacturing of glucocorticoid APIs?
A: Yes, the process utilizes readily available reagents like cyclohexanecarbaldehyde and common industrial solvents such as dichloromethane or nitropropane. The mild temperature range (0°C to 60°C) and simple workup procedures involving filtration and neutralization make it highly amenable to large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16,17-[(cyclohexylmethylene)bis(oxy)]-11,21-dihydroxy-pregna-1,4-diene-3,20-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development and commercialization of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated transketalization chemistry described in CN1473164A can be executed with precision and consistency on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the stereochemical integrity and impurity profile of every batch, guaranteeing that our clients receive materials that meet the highest global regulatory standards.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their glucocorticoid projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market with confidence.
