Advanced Enzymatic Production of S-Allyl-L-Cysteine for Commercial Scale-Up
Advanced Enzymatic Production of S-Allyl-L-Cysteine for Commercial Scale-Up
The pharmaceutical and nutraceutical industries have long sought reliable methods to produce bioactive sulfur compounds with consistent quality and high purity. A significant breakthrough in this domain is detailed in patent CN103451249A, which discloses a novel preparation method for S-Allyl-L-Cysteine (SAC). This water-soluble non-protein amino acid, naturally found in garlic, possesses potent antioxidant, antibacterial, and neuroprotective properties, making it a high-value target for therapeutic applications. However, traditional extraction methods from aged garlic suffer from inherently low yields and instability. The patented technology overcomes these historical bottlenecks by utilizing a precise enzymatic conversion of gamma-glutamyl-S-allyl-L-cysteine (GSAC). By shifting the paradigm from uncontrolled plant extraction to controlled biocatalysis, this process offers a robust pathway for manufacturing high-purity SAC, addressing critical supply chain vulnerabilities associated with agricultural raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of S-Allyl-L-Cysteine has been inextricably linked to the processing of garlic bulbs, specifically through the production of black garlic or aged garlic extracts. This conventional approach is fraught with biochemical inefficiencies that severely impact commercial viability. In natural garlic metabolic processes, SAC is an intermediate that is rapidly oxidized by endogenous alliinase enzymes into Alliin, preventing any significant accumulation of the desired compound. Even in aged garlic, where some conversion occurs, the content of SAC remains notoriously low, often requiring massive quantities of raw biomass to isolate minute amounts of the active ingredient. Furthermore, the extraction process involves complex solvent systems and lengthy fermentation periods, leading to inconsistent batch-to-batch quality and difficulties in meeting the stringent impurity profiles required for pharmaceutical grade intermediates. The reliance on agricultural cycles also introduces volatility in pricing and availability, making it a risky strategy for large-scale manufacturing.
The Novel Approach
The methodology presented in patent CN103451249A fundamentally reengineers the production landscape by decoupling SAC synthesis from direct garlic extraction. Instead of fighting against natural enzymatic degradation, this process leverages a specific biosynthetic precursor, gamma-glutamyl-S-allyl-L-cysteine (GSAC), which is available in higher concentrations in fresh garlic bulbs. The core innovation lies in the use of gamma-glutamyl transpeptidase (GGT) as a highly selective biocatalyst. This enzyme cleanly cleaves the gamma-glutamyl bond from the GSAC substrate under mild aqueous conditions, directly yielding S-Allyl-L-Cysteine without triggering the oxidative cascade that leads to Alliin formation. This targeted enzymatic hydrolysis not only drastically simplifies the downstream purification requirements but also ensures that the stereochemical integrity of the L-cysteine moiety is preserved. By controlling the reaction environment, manufacturers can achieve yields that are substantially superior to extraction methods, transforming SAC from a scarce natural extract into a reliably manufacturable fine chemical.
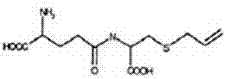
Mechanistic Insights into Gamma-Glutamyl Transpeptidase Catalysis
The efficacy of this synthesis route hinges on the precise mechanistic action of gamma-glutamyl transpeptidase (GGT) within a carefully buffered system. The reaction operates optimally within a pH range of 7.0 to 10.5, typically utilizing an ammonia-ammonium chloride buffer system to maintain stability. Mechanistically, the GGT enzyme facilitates the transfer of the gamma-glutamyl group from the donor substrate (GSAC) to an acceptor molecule, which in this hydrolytic context is water. This nucleophilic attack results in the liberation of glutamic acid and the formation of the free amine on the cysteine derivative, effectively generating S-Allyl-L-Cysteine. A critical aspect of this mechanism is the enzyme's specificity; it recognizes the gamma-peptide bond exclusively, leaving the sensitive allyl-thioether linkage and the carboxylic acid groups untouched. This chemoselectivity is paramount for minimizing side reactions and ensuring that the final product profile is clean, thereby reducing the burden on subsequent purification steps such as chromatography.
Furthermore, the patent highlights the strategic advantage of using immobilized enzyme preparations to enhance process control and product stability. By embedding the purified GGT into insoluble carriers such as chitosan cross-linked with glutaraldehyde or alginate matrices, the biocatalyst becomes heterogeneous. This physical state change prevents the enzyme from contaminating the final product stream and allows for its recovery and reuse across multiple reaction cycles. From a mechanistic standpoint, immobilization often enhances the thermal and operational stability of the enzyme, allowing the reaction to proceed efficiently at temperatures around 37°C for extended periods without significant loss of activity. This stability is crucial for maintaining consistent reaction kinetics in large-scale reactors, ensuring that the conversion of GSAC to SAC proceeds to completion with minimal formation of degradation byproducts, ultimately securing the high purity levels observed in the final crystalline product.
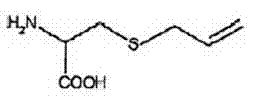
How to Synthesize S-Allyl-L-Cysteine Efficiently
The implementation of this enzymatic route requires a disciplined approach to reaction engineering and downstream processing to maximize yield and purity. The process begins with the dissolution of the GSAC substrate in a buffered solution, followed by the introduction of the biocatalyst under controlled temperature conditions. Once the bioconversion is complete, the reaction mixture undergoes a rigorous purification sequence involving ion exchange chromatography to separate the zwitterionic product from unreacted substrates and glutamic acid byproducts. The eluate is then concentrated and subjected to a crystallization protocol using ethanol, which serves both as an anti-solvent and a washing agent to remove residual impurities. For a detailed breakdown of the specific operational parameters, including flow rates for column chromatography and solvent ratios for recrystallization, please refer to the standardized synthesis guide below.
- Dissolve gamma-glutamyl-S-allyl-L-cysteine (GSAC) in a buffered solution with pH maintained between 7.0 and 10.5 to prepare the substrate mixture.
- Introduce gamma-glutamyl transpeptidase (either free or immobilized) as the biocatalyst to initiate the hydrolysis reaction, converting GSAC into S-Allyl-L-Cysteine.
- Purify the resulting reaction liquid through ion exchange chromatography, eluting with ammonia solution, followed by concentration and ethanol recrystallization to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from agricultural extraction to enzymatic synthesis represents a significant de-risking of the supply chain for S-Allyl-L-Cysteine. Traditional methods are beholden to crop yields, seasonal variations, and the complex logistics of processing perishable biomass, all of which contribute to price volatility and potential supply interruptions. In contrast, this biocatalytic process utilizes defined chemical substrates and reusable enzymes, creating a manufacturing workflow that is independent of harvest cycles. The ability to produce SAC on demand in a controlled facility setting ensures a consistent flow of material, eliminating the bottlenecks associated with sourcing high-quality aged garlic. Moreover, the simplification of the process flow—removing the need for extensive solid-liquid extraction and filtration of plant matter—translates directly into reduced operational expenditures and a smaller physical footprint for production facilities.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the efficiency of the biocatalyst and the simplicity of the purification train. By employing immobilized enzymes, the costly biocatalyst can be recovered and utilized repeatedly, amortizing the initial enzyme cost over a vast production volume and effectively lowering the cost per kilogram of the final API intermediate. Additionally, the avoidance of complex solvent extraction systems typical of botanical processing reduces solvent consumption and waste disposal costs. The high selectivity of the enzymatic reaction minimizes the formation of difficult-to-remove impurities, which in turn reduces the number of purification passes required, leading to substantial savings in energy and consumables while maintaining a competitive cost structure for high-purity pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Reliability is further bolstered by the robustness of the synthetic route against biological variability. Unlike garlic extracts, where the concentration of active ingredients can fluctuate wildly depending on the growing region and storage conditions of the bulb, the enzymatic process starts with a defined quantity of GSAC. This input consistency guarantees a predictable output, allowing for accurate production planning and inventory management. The use of standard unit operations such as packed-bed reactors and ion exchange columns means that the technology can be easily replicated or scaled across different manufacturing sites, providing redundancy and security of supply for global pharmaceutical customers who require uninterrupted access to critical intermediates for their drug formulations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this green chemistry approach offers distinct advantages over traditional extraction. The process operates in aqueous media at moderate temperatures, significantly reducing the energy intensity compared to the high-heat drying and extraction steps required for black garlic production. The use of ethanol for crystallization is preferable to more hazardous organic solvents, aligning with modern sustainability goals and simplifying regulatory compliance regarding solvent residues. The scalability is inherent in the design; the transition from bench-scale flasks to industrial enzyme columns is straightforward, allowing manufacturers to ramp up production from pilot batches to multi-ton annual capacities without encountering the nonlinear scale-up issues often seen in fermentation or extraction processes.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the process mechanics and product specifications. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a transparent view of the method's capabilities. Understanding these details is essential for R&D directors assessing route selection and quality assurance teams defining acceptance criteria for incoming materials. The following section addresses key concerns regarding enzyme stability, impurity profiles, and the specific advantages of the immobilization technique.
Q: How does this enzymatic method prevent the formation of Alliin?
A: Unlike traditional garlic extraction where endogenous alliinase enzymes convert S-Allyl-L-Cysteine into Alliin, this patented method uses specific gamma-glutamyl transpeptidase in a controlled environment, bypassing the oxidative pathways entirely.
Q: Can the biocatalyst be reused for industrial production?
A: Yes, the patent specifically describes the use of immobilized enzymes (e.g., embedded in chitosan or alginate), which allows for easy separation from the reaction mixture and repeated use, significantly enhancing process economics.
Q: What is the expected purity of the final crystalline product?
A: Through the combination of ion exchange chromatography and dual-step recrystallization with ethanol, the process achieves product purity levels exceeding 96%, with some embodiments reaching up to 98.86%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Allyl-L-Cysteine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the enzymatic synthesis of S-Allyl-L-Cysteine can be successfully translated from the laboratory to the manufacturing floor. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity, assay, and impurity levels. Our dedication to quality assurance ensures that every batch of SAC we supply is consistent, safe, and fully compliant with international regulatory standards, providing our partners with the confidence they need to advance their clinical and commercial programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis development or bulk supply of high-purity intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall cost of goods sold.
