Advanced Decarboxylation Technology for 5-Methylpyrazine-2-Carboxylic Acid Manufacturing
Advanced Decarboxylation Technology for 5-Methylpyrazine-2-Carboxylic Acid Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign pathways for synthesizing critical heterocyclic intermediates. Patent CN101519379B introduces a groundbreaking methodology for the preparation of 5-methylpyrazine-2-carboxylic acid, a valuable building block in medicinal chemistry. This innovation leverages a unique aqueous decarboxylation principle conducted within a saturated saline environment, marking a significant departure from traditional high-energy processes. By utilizing 5-methyl-2,3-pyrazinedicarboxylic acid as the primary feedstock, the method achieves effective decarboxylation at remarkably mild temperatures, eliminating the need for harsh organic solvents during the reaction phase. This approach not only simplifies the operational workflow but also aligns with modern green chemistry mandates by minimizing waste generation. The visual representation of the starting material highlights the structural complexity that this mild method successfully manages without degradation. 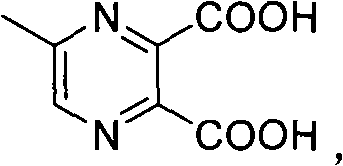
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the decarboxylation of pyrazine derivatives has been plagued by significant technical and economic hurdles that hinder scalable production. Traditional vacuum decarboxylation methods necessitate specialized equipment capable of maintaining high vacuum levels to remove generated carbon dioxide, resulting in substantial capital expenditure and maintenance costs for manufacturing facilities. Alternatively, solvent-based decarboxylation typically employs high-boiling organic solvents such as xylene or trimethylbenzene, requiring reaction temperatures ranging from 140°C to 180°C. These extreme thermal conditions often induce severe carbonization and generate a complex mixture of by-products that are notoriously difficult to separate, thereby compromising the overall yield and purity of the final API intermediate. Furthermore, earlier aqueous methods reported in literature, such as those in patent EP0194671, demand rigorous pH control between 0.7 and 2.2 and temperatures exceeding 155°C, creating a hazardous and energy-intensive environment that is ill-suited for large-scale commercial operations.
The Novel Approach
In stark contrast to these legacy techniques, the novel methodology described in CN101519379B utilizes a saturated saline solution to facilitate the decarboxylation process under significantly milder conditions. By operating within a temperature window of 105°C to 115°C, this process drastically reduces thermal stress on the molecular structure, thereby preserving the integrity of the pyrazine ring and minimizing the formation of degradation impurities. The absence of organic solvents during the core reaction phase eliminates the risks associated with solvent recovery and disposal, while the use of common salt as a reaction medium ensures that raw material costs remain exceptionally low. The reaction is conducted under light-protected conditions to prevent photolytic degradation, ensuring consistent quality across batches. This streamlined approach transforms a previously complex, multi-step purification challenge into a straightforward, high-selectivity transformation that is inherently safer and more cost-effective for industrial application.
Mechanistic Insights into Saline-Mediated Decarboxylation
The core innovation of this technology lies in the manipulation of solubility and ionic strength within the reaction medium to drive the decarboxylation equilibrium forward without extreme heat. In a saturated saline environment, the solubility characteristics of the dicarboxylic acid starting material and the monocarboxylic acid product differ significantly, which aids in shifting the reaction balance. The high ionic strength of the brine likely stabilizes the transition state of the decarboxylation, lowering the activation energy required compared to pure water systems. This allows the reaction to proceed efficiently at temperatures as low as 105°C, whereas pure water systems often fail to achieve comparable conversion rates without exceeding 150°C. The mechanism avoids the use of transition metal catalysts, which removes the risk of heavy metal contamination in the final product, a critical parameter for pharmaceutical grade intermediates. This catalyst-free nature simplifies the downstream purification process, as there is no need for expensive metal scavenging resins or complex filtration steps to meet regulatory limits.
Impurity control is another critical aspect where this saline-mediated mechanism excels, particularly regarding the suppression of isomeric by-products. The high selectivity of the reaction ensures that isomer impurities are tightly constrained within a range of 0.5% to 3%, which is a substantial improvement over the broad impurity profiles seen in high-temperature solvent methods. The mild conditions prevent the random thermal decomposition of the pyrazine ring, which is a common source of tarry by-products in conventional decarboxylation. By maintaining a closed reflux system in the dark, the process further mitigates the risk of oxidative side reactions that could compromise the color and purity of the final crystalline product. This level of control over the impurity profile directly translates to reduced processing time during the work-up phase, as fewer recrystallization cycles are needed to achieve the target specification of 98% HPLC purity.
How to Synthesize 5-Methylpyrazine-2-Carboxylic Acid Efficiently
The operational protocol for this synthesis is designed for robustness and ease of execution in a standard chemical manufacturing plant. The process begins with the preparation of a saturated saline solution, into which the 5-methyl-2,3-pyrazinedicarboxylic acid is introduced. The mixture is then heated to a reflux temperature between 105°C and 115°C and maintained for a duration of 8 to 15 hours, depending on the specific batch scale and conversion monitoring. Reaction progress is typically tracked via HPLC to ensure the starting material is fully consumed before proceeding to the work-up stage. Once the reaction is complete, the mixture is cooled to room temperature, and the product is isolated through a combination of filtration and solvent extraction using butanone. The detailed standardized synthesis steps, including precise stoichiometric ratios and safety precautions, are outlined in the technical guide below.
- Dissolve 5-methyl-2,3-pyrazinedicarboxylic acid in saturated saline solution and heat to 105-115°C.
- Maintain reflux conditions in the dark for 8-15 hours until the starting material is consumed.
- Cool the mixture, filter, extract the filtrate with butanone, and crystallize the product from an ice-water bath.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this saline-based decarboxylation technology offers profound strategic advantages in terms of cost structure and operational reliability. The elimination of high-boiling organic solvents from the reaction step removes a major variable cost driver, as there is no longer a need to purchase, recover, or dispose of large volumes of xylene or similar hydrocarbons. This shift significantly reduces the overall cost of goods sold (COGS) for the intermediate, allowing for more competitive pricing in the global market. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to a lower carbon footprint and aligning with corporate sustainability goals. The simplicity of the equipment requirements means that production can be scaled up rapidly without the need for specialized high-pressure reactors or complex vacuum systems, ensuring a more resilient and flexible supply chain.
- Cost Reduction in Manufacturing: The removal of organic solvents from the primary reaction phase leads to substantial cost savings by eliminating solvent procurement and recovery expenses. Additionally, the absence of transition metal catalysts removes the need for costly metal scavenging processes, further optimizing the production budget. The high selectivity of the reaction minimizes raw material waste, ensuring that the maximum amount of feedstock is converted into valuable product. These factors combine to create a highly efficient economic model that supports long-term price stability for downstream customers.
- Enhanced Supply Chain Reliability: The use of common salt and water as the primary reaction medium ensures that raw material availability is never a bottleneck, as these commodities are globally accessible and inexpensive. The robustness of the process against minor fluctuations in operating conditions means that batch-to-batch consistency is high, reducing the risk of production delays due to failed quality control tests. This reliability allows for more accurate forecasting and inventory planning, ensuring that critical pharmaceutical intermediates are available when needed without interruption.
- Scalability and Environmental Compliance: The green nature of this process, characterized by low pollution and mild conditions, simplifies regulatory compliance and environmental permitting. The reduction in hazardous waste generation lowers the costs associated with waste treatment and disposal, making the facility more sustainable. The process is inherently scalable from laboratory to commercial tonnage without significant re-engineering, facilitating rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this decarboxylation technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The insights provided here clarify the operational parameters and quality expectations associated with the saline-mediated synthesis method.
Q: Why is saturated saline used instead of pure water for this decarboxylation?
A: Saturated saline optimizes the solubility profile and reaction kinetics, allowing effective decarboxylation at lower temperatures (105-115°C) compared to pure water methods which often require over 150°C and strict pH control.
Q: What is the expected purity of the final 5-methylpyrazine-2-carboxylic acid?
A: The patented process consistently achieves an HPLC purity of up to 98%, with isomer impurities tightly controlled between 0.5% and 3% due to the high selectivity of the saline medium.
Q: Does this process require expensive organic solvents for the reaction step?
A: No, the core reaction occurs entirely in an aqueous saline solution without added organic solvents, significantly reducing raw material costs and environmental pollution, though butanone is used subsequently for extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Methylpyrazine-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of high-value pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the saline decarboxylation process can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in process optimization allows us to identify and mitigate potential scale-up risks early, guaranteeing a smooth transition from pilot plant to full-scale manufacturing.
We invite you to collaborate with us to optimize your supply chain for 5-methylpyrazine-2-carboxylic acid and related heterocyclic compounds. By leveraging our technical capabilities, you can achieve a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project is built on a foundation of scientific certainty and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
