Advanced Aqueous Salification Technology for Commercial Hydrocortisone Sodium Succinate Production
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for critical corticosteroid derivatives, and the preparation of Hydrocortisone Sodium Succinate stands as a prime example of process optimization. Based on the technological breakthroughs detailed in patent CN101613392A, we observe a significant departure from traditional, cumbersome salification methods towards a streamlined, aqueous-based protocol. This innovation addresses long-standing challenges in the manufacturing of anti-inflammatory agents, specifically targeting yield improvement and operational simplicity. The core of this advancement lies in the direct conversion of hydrocortisone monomester succinate into its sodium salt using a controlled aqueous environment, bypassing the need for complex buffer systems that previously complicated the supply chain. By leveraging this method, manufacturers can achieve a reaction profile that is not only faster but also generates significantly fewer side products, ensuring a higher quality final API intermediate.
The structural integrity of the final product is paramount for its efficacy in treating acute anaphylactic diseases and adrenal crises. As illustrated below, the molecule retains the critical steroid backbone while incorporating the water-soluble succinate moiety essential for rapid intravenous administration. Understanding this molecular architecture is crucial for R&D teams aiming to replicate or optimize the synthesis for large-scale production.
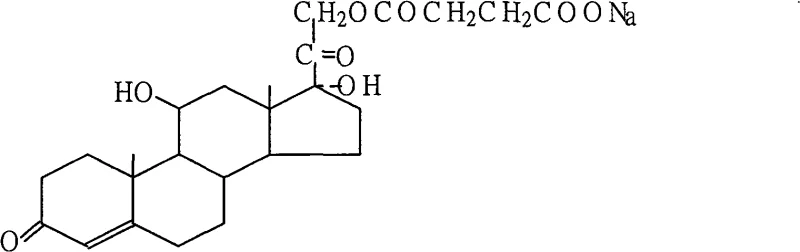
Historically, the industrial production of salifiable corticosteroids has been plagued by inefficiencies that drive up costs and extend lead times. Conventional methods often relied on reacting hydrocortisone with succinic acid directly in water, a process notorious for low yields and extremely difficult drying characteristics due to high moisture retention. Alternatively, earlier patented approaches utilized buffered solutions to facilitate the reaction, which inadvertently increased the complexity of the downstream processing and introduced additional impurities that required rigorous removal. These legacy techniques resulted in prolonged batch cycles and inconsistent product quality, creating bottlenecks for procurement managers who require reliable volumes of high-purity intermediates. The inability to efficiently remove residual solvents and water from the crystal lattice often meant extended drying times, consuming excessive energy and delaying shipment schedules.
In stark contrast, the novel approach outlined in the reference patent introduces a paradigm shift by utilizing hydrocortisone monomester succinate as the direct starting material for salification. This method operates within a precise pH window of 7.5 to 11.0, achieved by the addition of soda ash or sodium bicarbonate, followed by the strategic addition of acetone as an anti-solvent. This precipitation technique forces the product out of the aqueous solution rapidly and cleanly, avoiding the formation of hard-to-dry hydrates common in older methods. The result is a process that is not only chemically superior but also logistically advantageous, offering a stable reaction profile that minimizes the risk of batch failure. For supply chain heads, this translates to a more predictable manufacturing timeline and a product that is inherently easier to handle and package.
Mechanistic Insights into Aqueous Salification and Precipitation
From a mechanistic perspective, the success of this synthesis relies on the precise control of acid-base equilibrium and solubility parameters. The reaction begins with the dissolution of the acidic monoester in purified water, where the carboxylic acid group of the succinate side chain is deprotonated by the added base (sodium bicarbonate or hydroxide). This formation of the sodium carboxylate salt dramatically increases the polarity of the molecule, rendering it highly soluble in the aqueous phase initially. However, the subsequent addition of acetone alters the dielectric constant of the solvent system, drastically reducing the solubility of the ionic salt. This induces supersaturation and drives the nucleation and growth of the Hydrocortisone Sodium Succinate crystals. The critical nature of maintaining the temperature between 10-20°C during this phase cannot be overstated, as it controls the kinetics of crystallization, preventing the occlusion of impurities and ensuring the formation of a free-flowing powder rather than an amorphous gum.
Furthermore, the impurity profile is tightly managed through the integration of a decolorization step using activated carbon prior to the final concentration. This step is vital for removing trace organic impurities and colored byproducts that may arise from the precursor synthesis or minor oxidative degradation. By filtering the solution before the final vacuum concentration, the process ensures that the resulting solid meets the stringent visual and chemical purity requirements of pharmacopoeias like CP2005 and USP28. The low-temperature vacuum drying (T≤30°C) further protects the thermally sensitive steroid nucleus from decomposition, preserving the biological activity of the final API. This meticulous attention to thermal and chemical stability is what differentiates a lab-scale curiosity from a commercially viable manufacturing process capable of delivering high-purity pharmaceutical intermediates.
How to Synthesize Hydrocortisone Sodium Succinate Efficiently
To implement this synthesis effectively, operators must adhere to strict parameter controls regarding temperature and solvent ratios. The process begins with the dissolution of the monoester in water, followed by pH adjustment and the slow, controlled dripping of acetone to induce precipitation. Detailed standard operating procedures regarding stirring speeds, specific addition rates, and filtration techniques are critical for reproducibility. For a comprehensive, step-by-step guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized protocol below.
- Dissolve hydrocortisone monomester succinate in purified water at a controlled temperature between 0-60°C to ensure complete solubilization.
- Adjust the pH value to the range of 7.5-11.0 by adding soda ash, sodium bicarbonate, or sodium hydroxide to initiate the salification reaction.
- Slowly drip acetone into the solution while maintaining the temperature at 10-20°C, followed by filtration and vacuum drying at temperatures not exceeding 30°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced synthesis route offers tangible benefits that extend beyond mere chemical yield. The elimination of complex buffering agents and the reduction in organic solvent usage directly correlate to a simplified waste management profile and lower raw material costs. By shifting the primary solvent system to water with acetone recovery, the process aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing facility. This simplification also means fewer unit operations are required, which decreases the potential for human error and equipment downtime, thereby enhancing the overall reliability of the supply chain for critical corticosteroid ingredients.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the significant simplification of the downstream processing. Traditional methods often require extensive drying cycles to remove tenacious water molecules, consuming vast amounts of energy and time. In this novel method, the product precipitates in a form that is much easier to dewater and dry, leading to substantial energy savings. Additionally, the high yield reported in the patent embodiments implies less raw material waste per kilogram of finished product, directly improving the cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is contingent upon consistent batch-to-batch quality and predictable lead times. This synthesis route offers a robust operational window, with tolerance for slight variations in temperature and pH without catastrophic failure. The use of commodity chemicals like water, acetone, and sodium bicarbonate ensures that raw material availability is never a bottleneck, even during global supply disruptions. This stability allows suppliers to maintain consistent inventory levels and meet just-in-time delivery schedules required by major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the linear nature of this salification and precipitation method makes it highly scalable from pilot plants to multi-ton reactors. The process generates minimal hazardous waste, primarily consisting of aqueous filtrates that are easier to treat than mixed organic waste streams. This ease of waste treatment facilitates compliance with increasingly strict environmental regulations, ensuring long-term operational continuity for manufacturing sites. The ability to scale without re-engineering the core chemistry provides a secure foundation for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Hydrocortisone Sodium Succinate. These answers are derived directly from the patented methodology and practical experience in steroid chemistry, providing clarity for stakeholders evaluating this technology for their supply chains.
Q: What are the primary advantages of this aqueous salification method over traditional buffered processes?
A: This novel method eliminates the need for complex buffered solutions, significantly simplifying the operational workflow. By utilizing direct salification in water followed by acetone precipitation, the process reduces environmental pollution, shortens reaction time, and achieves consistently higher yields compared to conventional techniques that often suffer from difficult drying and lower efficiency.
Q: How does the process control impurities during the synthesis of Hydrocortisone Sodium Succinate?
A: Impurity control is achieved through strict temperature regulation and the use of activated carbon. The reaction is maintained at a low temperature range of 10-20°C during the critical precipitation phase to prevent hydrolysis or degradation of the steroid backbone. Furthermore, the inclusion of a decolorization step with activated carbon ensures the removal of organic impurities, resulting in a white or off-white powder that meets stringent pharmacopoeia standards.
Q: Is this synthesis route scalable for industrial manufacturing of corticosteroid intermediates?
A: Yes, the process is highly amenable to commercial scale-up. The reliance on common, inexpensive solvents like water and acetone, combined with simple unit operations such as dissolution, pH adjustment, and filtration, allows for easy transition from laboratory to multi-ton production. The low drying temperature requirement also facilitates energy-efficient processing in large-scale rotary evaporators or spray dryers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Sodium Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this aqueous salification process are fully realized in a GMP-compliant environment. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against global pharmacopoeia standards.
We invite you to collaborate with us to optimize your sourcing strategy for this critical anti-inflammatory intermediate. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient synthesis route can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of quality, reliability, and scientific innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
