Advanced Synthesis of 17-Beta-Hydroxy Steroid Impurities for Ulipristal Acetate Quality Control
The pharmaceutical industry faces rigorous challenges in maintaining the purity profiles of complex steroid hormones, particularly for selective progesterone receptor modulators like Ulipristal Acetate. Patent CN112010918A introduces a groundbreaking methodology for converting the 17-carbonyl group of steroid hormone substances directly into the 17-beta-hydroxy configuration, a critical stereoisomer often classified as a process-related impurity. This technical advancement addresses a significant gap in the availability of certified reference standards required for regulatory compliance and quality control. By shifting the reaction paradigm from traditional thermodynamic conditions to a kinetically controlled cyanation process, the invention enables the efficient production of ubbi ketal impurities with exceptional purity levels exceeding 96 percent. This development is pivotal for manufacturers seeking to validate their analytical methods and ensure the safety of final drug products without relying on hazardous heavy metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex steroid intermediates resembling the ubbi ketal structure has relied on multi-step sequences that pose severe environmental and operational risks. As illustrated in the prior art, conventional routes typically commence with an ethynylation reaction followed by deprotection and a subsequent hydrolysis step catalyzed by mercuric sulfate. The reliance on mercury salts introduces substantial toxicity concerns, complicating waste disposal and increasing the environmental footprint of the manufacturing process. Furthermore, these traditional pathways often suffer from low reaction yields and generate numerous side reactions, necessitating labor-intensive purification techniques such as column chromatography to isolate the target compound. The technical difficulty associated with removing trace heavy metals to meet pharmaceutical grade specifications adds another layer of complexity and cost, making these legacy processes unsustainable for modern large-scale production.
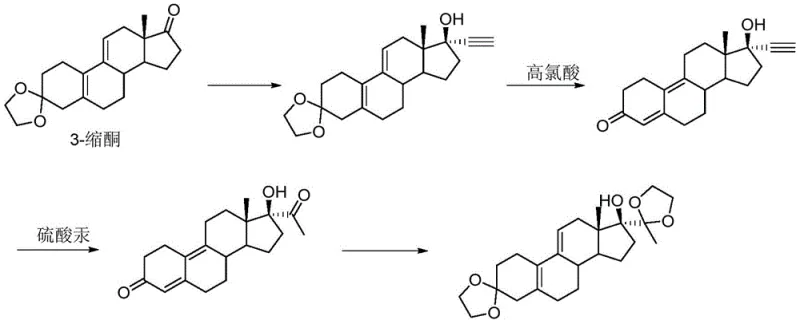
The Novel Approach
In stark contrast, the methodology disclosed in CN112010918A presents a streamlined, four-step synthetic strategy that eliminates the need for toxic transition metals entirely. The core innovation involves a direct cyanation of the 17-keto steroid using acetone cyanohydrin in the presence of an organic base and a specific ester or ether solvent system. This approach not only simplifies the reaction workflow but also fundamentally alters the stereochemical outcome to favor the beta-isomer through kinetic control. Following the initial cyanation, the route proceeds through selective ketal protection, a Grignard addition to install the C21 methyl group, and a final ketalization step. This sequence is robust, scalable, and avoids the use of column chromatography, relying instead on efficient crystallization techniques to achieve high purity. The elimination of mercury and the reduction in unit operations represent a significant leap forward in green chemistry principles applied to steroid manufacturing.
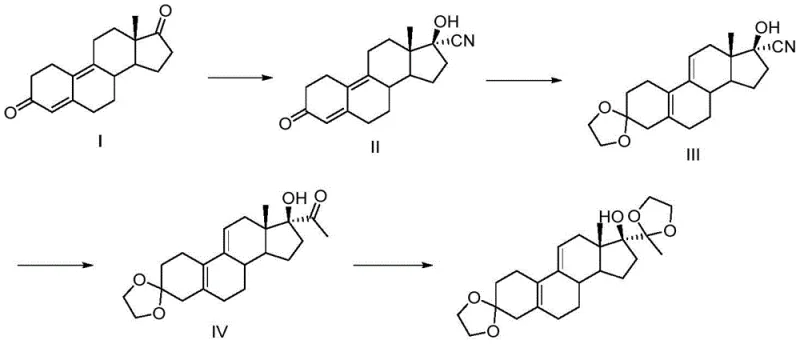
Mechanistic Insights into Kinetic Cyanation and Stereocontrol
The success of this synthesis hinges on a sophisticated understanding of reaction kinetics versus thermodynamics in steroid functionalization. In traditional cyanation reactions performed in alcoholic solvents with inorganic bases, the system tends to equilibrate towards the thermodynamically more stable 17-alpha-hydroxy product. However, the patented process strategically employs organic solvents such as ethyl acetate or methyl tert-butyl ether, which possess lower polarity compared to alcohols. Coupled with the use of organic bases like triethylamine or pyridine, the reaction environment creates a milder alkalinity that suppresses the epimerization of the initial adduct. Consequently, the nucleophilic attack of the cyanide ion on the 17-carbonyl group generates the kinetic product, the 17-beta-hydroxy nitrile, which is then rapidly isolated before it can convert to the alpha-isomer. This precise manipulation of solvent and base properties allows for the selective accumulation of the desired impurity standard with yields reaching up to 95 percent in the first step alone.
Furthermore, the subsequent steps are designed to maintain this stereochemical integrity while building molecular complexity. The protection of the C3-ketone as an ethylene ketal prevents unwanted side reactions during the Grignard addition. The use of methyl magnesium chloride at controlled low temperatures ensures selective addition to the nitrile or ketone functionality without disturbing the sensitive double bonds in the steroid backbone. The final ketalization at the C20 position locks the structure into the ubbi ketal impurity form. Each step is optimized to minimize byproduct formation, ensuring that the final impurity reference standard is free from genotoxic impurities and other related substances that could interfere with analytical validation. This level of control is essential for producing materials that meet the stringent requirements of pharmacopoeial standards.
How to Synthesize 3,3,20,20-bis(ethylenedioxy)-17β-hydroxy-19-norpregna-5(10),9(11)-diene Efficiently
The synthesis of this complex steroid impurity requires precise adherence to reaction conditions to ensure the correct stereochemistry and high purity. The process begins with the kinetic cyanation of the starting ketone, followed by sequential protection and carbon-chain extension steps. Detailed operational parameters regarding temperature control, reagent stoichiometry, and workup procedures are critical for replicating the high yields reported in the patent literature. For process chemists looking to implement this route, understanding the nuances of the crystallization steps is just as important as the reaction chemistry itself, as these purification stages are key to achieving the required specification limits.
- React 17-carbonyl steroid with acetone cyanohydrin in ethyl acetate using triethylamine to selectively form the 17-beta-hydroxy nitrile.
- Protect the C3-ketone as an ethylene ketal using ethylene glycol and p-toluenesulfonic acid in dichloromethane.
- Perform a Grignard reaction with methyl magnesium chloride to introduce the C21 methyl group, followed by final ketal protection at C20.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits over traditional methods. The primary advantage lies in the drastic simplification of the manufacturing process, which directly translates to reduced operational expenditures and enhanced supply reliability. By removing the requirement for mercuric sulfate, manufacturers eliminate the need for expensive heavy metal scavenging resins and complex wastewater treatment protocols associated with toxic effluent. This not only lowers the direct cost of goods sold but also mitigates regulatory risks associated with environmental compliance. Additionally, the high conversion rates and the ability to purify the product via simple crystallization rather than chromatography significantly reduce solvent consumption and processing time, leading to substantial cost savings in raw material utilization and facility throughput.
- Cost Reduction in Manufacturing: The elimination of column chromatography is a major driver for cost efficiency in this process. Chromatographic separations are notoriously expensive at scale due to high solvent usage, silica gel costs, and low throughput. By designing a route where the product precipitates or crystallizes directly from the reaction mixture or after a simple solvent swap, the process becomes inherently more economical. Furthermore, the use of readily available organic bases and common ester solvents replaces specialized or hazardous reagents, stabilizing the raw material supply chain against price volatility. The high yield of the initial cyanation step ensures that expensive steroid starting materials are utilized with maximum efficiency, minimizing waste and maximizing the output per batch.
- Enhanced Supply Chain Reliability: Dependence on toxic heavy metals like mercury can create supply bottlenecks due to strict transportation and storage regulations. By transitioning to a metal-free organic synthesis, the supply chain becomes more resilient and less susceptible to regulatory disruptions. The robustness of the reaction conditions, which tolerate a range of temperatures and use common industrial solvents, facilitates easier technology transfer between manufacturing sites. This flexibility ensures that production can be scaled up or shifted to different facilities without significant re-validation efforts, guaranteeing a continuous supply of critical impurity standards needed for ongoing drug quality testing.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in any fine chemical plant, such as stirred tank reactors and filtration units. The absence of toxic mercury waste simplifies the environmental permitting process and reduces the long-term liability associated with hazardous waste disposal. This aligns perfectly with the growing corporate mandate for sustainable manufacturing practices. The ability to produce high-purity materials with a smaller environmental footprint enhances the brand reputation of the supplier and meets the increasingly strict ESG (Environmental, Social, and Governance) criteria demanded by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this steroid impurity standard. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities.
Q: Why is the 17-beta-hydroxy isomer considered an impurity in Ulipristal Acetate?
A: In the synthesis of Ulipristal Acetate, the desired active pharmaceutical ingredient requires a 17-alpha-hydroxy configuration. The 17-beta-hydroxy isomer is a stereoisomeric byproduct that must be strictly controlled and quantified to ensure drug safety and efficacy, necessitating high-purity reference standards.
Q: How does the new cyanation method improve upon traditional ethynylation routes?
A: Traditional routes often rely on ethynylation followed by mercuric sulfate hydrolysis, which involves highly toxic heavy metals and complex purification via column chromatography. The new method utilizes a metal-free cyanation strategy with organic bases, offering a safer, more environmentally friendly profile with higher yields and simpler crystallization workups.
Q: What represents the key technical breakthrough in controlling stereochemistry?
A: The breakthrough lies in switching from polar alcohol solvents and inorganic bases to non-polar ester/ether solvents and organic bases. This shifts the reaction from thermodynamic control (favoring the alpha-isomer) to kinetic control, effectively trapping the desired 17-beta-hydroxy configuration before it can equilibrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3,20,20-bis(ethylenedioxy)-17β-hydroxy-19-norpregna-5(10),9(11)-diene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality impurity standards in the development and lifecycle management of pharmaceutical products like Ulipristal Acetate. Our team of expert process chemists has extensively evaluated the synthetic route described in CN112010918A and possesses the technical capability to execute this complex steroid functionalization with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 3,3,20,20-bis(ethylenedioxy)-17β-hydroxy-19-norpregna-5(10),9(11)-diene we deliver meets the highest industry standards for reference materials.
We invite you to collaborate with us to optimize your supply chain for steroid intermediates and impurity standards. By leveraging our expertise in green chemistry and process intensification, we can help you achieve significant efficiencies in your quality control workflows. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your regulatory and commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
