Advanced Manufacturing of Dithiothreitol: A Safer, High-Yield Quaternary Ammonium Salt Route for Global Supply Chains
Introduction to Next-Generation Dithiothreitol Manufacturing
The global demand for high-purity reducing agents in biochemical and pharmaceutical applications continues to surge, driving the need for more efficient and safer synthesis routes. Patent CN115093352A introduces a groundbreaking preparation method for dithiothreitol (DTT), also known as Cleland's reagent, which addresses critical limitations in traditional manufacturing. This novel approach utilizes a quaternary ammonium salt strategy to stabilize reactive intermediates, thereby preventing unwanted side reactions that have historically plagued DTT production. By shifting the synthetic paradigm from direct oxidation of hazardous halides to a protected quaternary pathway, this technology offers a robust solution for producing high-purity pharmaceutical intermediates. The method encompasses a sophisticated five-step sequence involving quaternization, controlled oxidation, acetylation, thioacetate substitution, and final hydrolysis. For R&D directors and supply chain leaders, understanding this mechanistic shift is vital for securing a reliable dithiothreitol supplier capable of meeting stringent quality and volume requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of dithiothreitol typically involves the direct oxidation of 1,4-dibromo-2-butene using potassium permanganate. While conceptually straightforward, this legacy process suffers from severe chemical inefficiencies and safety hazards. The oxidation step inherently generates potassium hydroxide, a strong base, as a byproduct. In the presence of this alkaline environment, the halogen atoms on the butene backbone are highly susceptible to hydrolysis, leading to the degradation of the starting material before it can be converted into the desired diol. Consequently, the yield of this critical oxidation step is notoriously low, often hovering between only 30% and 40%. Furthermore, the raw material, 1,4-dibromo-2-butene, possesses strong irritant properties that pose significant occupational health risks, requiring operators to exercise extreme caution to prevent damage to skin and mucous membranes. These factors combine to create a manufacturing bottleneck that increases waste, drives up costs, and complicates regulatory compliance.
The Novel Approach
The innovative route disclosed in the patent fundamentally alters the reaction landscape by introducing a quaternization step prior to oxidation. By reacting 1,4-dibromo-2-butene with pyridine, the process generates a quaternary ammonium salt intermediate that serves as a stable precursor. 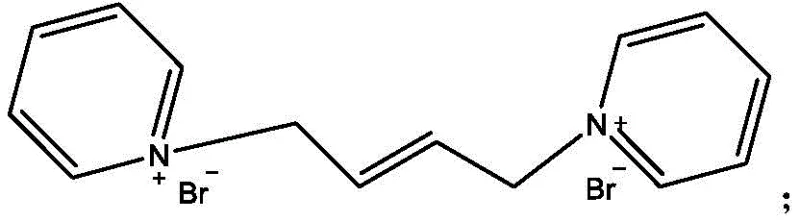 This structural modification is pivotal; the resulting quaternary salt is almost entirely free of the irritation associated with the parent halide, drastically improving workplace safety. More importantly, this intermediate exhibits remarkable stability against hydrolysis, even under the alkaline conditions required for subsequent oxidation. This stability ensures that the carbon-halogen bonds remain intact during the conversion to the diol, effectively bypassing the yield-killing side reactions of the conventional method. The result is a streamlined process that not only enhances operator safety but also delivers a substantial improvement in overall process efficiency and material throughput.
This structural modification is pivotal; the resulting quaternary salt is almost entirely free of the irritation associated with the parent halide, drastically improving workplace safety. More importantly, this intermediate exhibits remarkable stability against hydrolysis, even under the alkaline conditions required for subsequent oxidation. This stability ensures that the carbon-halogen bonds remain intact during the conversion to the diol, effectively bypassing the yield-killing side reactions of the conventional method. The result is a streamlined process that not only enhances operator safety but also delivers a substantial improvement in overall process efficiency and material throughput.
Mechanistic Insights into Quaternary Salt Stabilization and Oxidation
The core chemical innovation lies in the electronic and steric stabilization provided by the pyridinium moiety. In the conventional route, the electron-withdrawing nature of the adjacent bromine atoms makes the carbon backbone vulnerable to nucleophilic attack by hydroxide ions generated during oxidation. However, in the novel pathway, the formation of the quaternary ammonium salt alters the electronic distribution of the molecule. 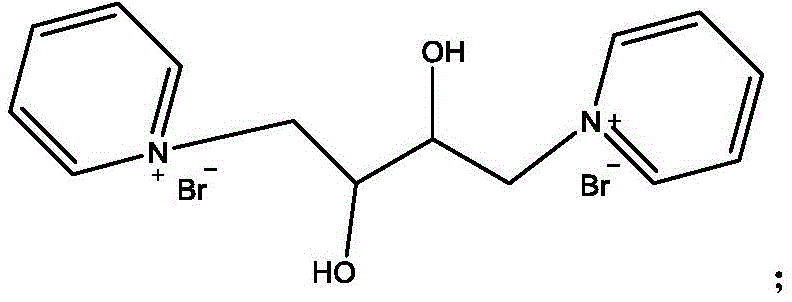 When the quaternary salt is subjected to oxidation using potassium permanganate at controlled low temperatures (0-5°C), the molecule resists hydrolytic degradation. The positive charge on the nitrogen atom and the bulk of the pyridine ring likely create a protective environment that hinders the approach of hydroxide ions to the reactive carbon centers. This allows the permanganate to selectively oxidize the alkene double bond to a vicinal diol without cleaving the carbon-bromine bonds. Following oxidation, the diol functionality is protected via acetylation, and the bromine atoms are subsequently displaced by thioacetate groups in a nucleophilic substitution reaction. This precise control over reactivity ensures that the sulfur groups are introduced only after the oxygen functionality is securely in place, minimizing the formation of complex impurity profiles.
When the quaternary salt is subjected to oxidation using potassium permanganate at controlled low temperatures (0-5°C), the molecule resists hydrolytic degradation. The positive charge on the nitrogen atom and the bulk of the pyridine ring likely create a protective environment that hinders the approach of hydroxide ions to the reactive carbon centers. This allows the permanganate to selectively oxidize the alkene double bond to a vicinal diol without cleaving the carbon-bromine bonds. Following oxidation, the diol functionality is protected via acetylation, and the bromine atoms are subsequently displaced by thioacetate groups in a nucleophilic substitution reaction. This precise control over reactivity ensures that the sulfur groups are introduced only after the oxygen functionality is securely in place, minimizing the formation of complex impurity profiles.
Furthermore, the final hydrolysis step is optimized to remove the acetyl protecting groups while preserving the delicate dithiol structure. The use of sulfuric acid in methanol under reflux conditions facilitates the cleavage of both the thioester and acetate ester bonds simultaneously. This convergent deprotection strategy simplifies the downstream processing, as it eliminates the need for multiple isolation steps between deprotection events. The resulting crude product can be purified through crystallization, yielding dithiothreitol with high stereochemical integrity. For R&D teams, this mechanistic clarity translates to a predictable impurity profile, making validation and quality control significantly more manageable compared to the erratic byproduct formation seen in direct oxidation methods.
How to Synthesize Dithiothreitol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing temperature control and stoichiometry to maximize yield. The process begins with the exothermic quaternization in ethanol, followed by a delicate low-temperature oxidation in water. Subsequent steps involve organic solvent extractions and crystallizations to ensure purity at each stage. While the general workflow is robust, precise adherence to the reaction parameters—such as maintaining the oxidation temperature below 5°C and controlling the pH during workup—is essential for replicating the high yields reported in the embodiments.
- React 1,4-dibromo-2-butene with pyridine in ethanol below 35°C to form a stable quaternary ammonium salt intermediate.
- Oxidize the quaternary salt using potassium permanganate at 0-5°C to generate the diol oxide without hydrolyzing the halogen groups.
- Protect the hydroxyl groups by reacting with acetic anhydride, followed by substitution with potassium thioacetate and final acid hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this quaternary salt-based synthesis offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic reduction of operational risk and the enhancement of supply continuity. By eliminating the handling of highly irritating raw materials in the later stages of the process, the facility reduces its reliance on specialized personal protective equipment and minimizes the potential for workplace incidents that could halt production. Moreover, the significant improvement in chemical yield directly correlates to cost reduction in pharmaceutical intermediates manufacturing. Higher yields mean less raw material is required per kilogram of finished product, lowering the variable cost of goods sold and reducing the volume of chemical waste that requires disposal.
- Cost Reduction in Manufacturing: The elimination of yield-killing hydrolysis side reactions means that the overall process efficiency is markedly higher than traditional methods. In the conventional route, losing 60-70% of material during the oxidation step represents a massive financial drain. By stabilizing the intermediate, this new method recovers that value, leading to substantial cost savings on raw materials. Additionally, the simplified purification process reduces the consumption of solvents and energy required for extensive chromatography or recrystallization, further driving down the manufacturing cost base without compromising quality.
- Enhanced Supply Chain Reliability: The stability of the quaternary ammonium salt intermediate allows for greater flexibility in production scheduling. Unlike unstable intermediates that must be processed immediately, this salt can potentially be stored or transported with lower risk, decoupling the upstream quaternization step from the downstream oxidation and substitution steps. This modularity enhances the resilience of the supply chain, allowing manufacturers to build inventory buffers at the intermediate stage to mitigate disruptions in the final synthesis steps, ensuring a reliable dithiothreitol supplier status even during market fluctuations.
- Scalability and Environmental Compliance: The reaction conditions described, such as oxidation at 0-5°C and substitution at 30-35°C, are readily achievable in standard stainless steel reactors without the need for exotic cryogenic or high-pressure equipment. This ease of scale-up facilitates the commercial scale-up of complex reducing agents from pilot batches to multi-ton annual production. Furthermore, the reduction in waste generation due to higher selectivity aligns with increasingly stringent environmental regulations, reducing the burden on wastewater treatment facilities and lowering the carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this technology for large-scale production. Understanding these details is crucial for stakeholders evaluating the transition to this improved manufacturing platform.
Q: Why is the quaternary ammonium salt route superior to direct oxidation for DTT production?
A: Direct oxidation of 1,4-dibromo-2-butene generates strong bases that cause hydrolysis of the halogen groups, leading to low yields (30-40%). The quaternary salt route prevents this hydrolysis, significantly improving yield and safety.
Q: What are the safety advantages of this new synthesis method?
A: The intermediate quaternary ammonium salt is almost free of irritation compared to the raw material 1,4-dibromo-2-butene, reducing risks to skin and mucous membranes during operation and storage.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes mild reaction conditions (0-35°C) and common solvents like ethanol and water, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dithiothreitol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality. As a premier CDMO partner, we possess the technical expertise to adapt and optimize this quaternary salt route for industrial-scale manufacturing. Our engineering teams are equipped to handle the specific thermal and mixing requirements of the oxidation and substitution steps, ensuring that the high yields demonstrated in the lab are replicated in our production facilities. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our rigorous QC labs enforce stringent purity specifications, ensuring that every batch of dithiothreitol meets the exacting standards required for biochemical and pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can secure your position as a leader in the life sciences industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
