Advanced Manufacturing of Articaine Hydrochloride via Optimized Amidation and Amination Routes
Introduction to the Optimized Synthesis of Articaine Hydrochloride
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational safety, a challenge effectively addressed by the preparation method disclosed in patent CN102060840B. This technology outlines a streamlined three-step synthetic route for Articaine Hydrochloride, a potent amide-type local anesthetic widely used in dental and surgical procedures. Unlike legacy processes that rely on hazardous reagents and complex purification sequences, this novel approach utilizes a sequential amidation, amination, and salification strategy that significantly enhances process reliability. By shifting away from toxic aromatic solvents and employing mild reaction conditions, the method not only improves the overall yield coefficient but also aligns with modern green chemistry principles essential for sustainable pharmaceutical intermediates production. For R&D directors and procurement specialists, understanding this pathway offers a clear window into how next-generation synthesis can drive both quality assurance and supply chain resilience.
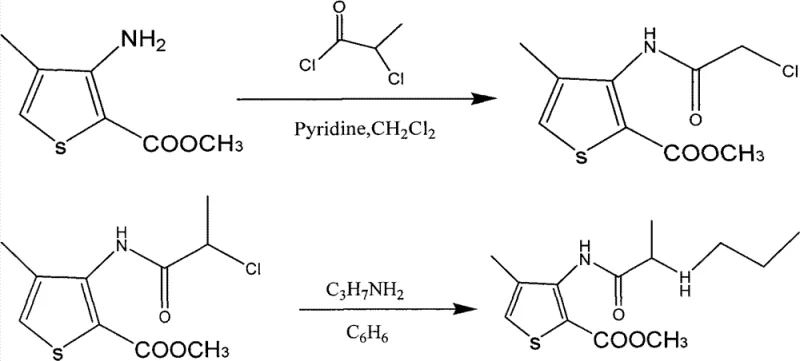
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Articaine Hydrochloride has been plagued by significant operational hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional literature reports often describe multi-step sequences requiring the use of benzene, a known carcinogen, and pyridine, which poses severe toxicity and odor issues in a plant environment. These conventional routes typically suffer from rigorous reaction conditions, including extreme temperatures or pressures, which demand expensive specialized equipment and rigorous safety protocols. Furthermore, the yield efficiency in these older methods is notably suboptimal, with amidation steps frequently achieving less than 70% conversion and subsequent steps dropping below 65%. This cumulative loss of material not only inflates the cost of goods sold but also generates substantial chemical waste, creating a heavy burden on waste treatment facilities and complicating regulatory compliance for manufacturers aiming to maintain a reliable API intermediate supplier status.
The Novel Approach
In stark contrast, the methodology presented in CN102060840B introduces a refined protocol that drastically simplifies the operational landscape while boosting output metrics. The new route replaces hazardous solvents with dichloromethane for the initial acylation and dimethylformamide (DMF) for the amination step, creating a safer working environment and reducing the complexity of solvent recovery systems. The process achieves a remarkable improvement in yield, with the initial amidation step reaching up to 85% and the subsequent amination step achieving approximately 90% efficiency under optimized conditions. By utilizing triethylamine as a superior catalyst over inorganic alternatives, the reaction kinetics are accelerated without necessitating harsh thermal inputs. This strategic shift allows for a more predictable production schedule and facilitates cost reduction in pharmaceutical manufacturing by minimizing raw material waste and energy consumption associated with high-temperature processing.
Mechanistic Insights into Triethylamine-Catalyzed Amidation
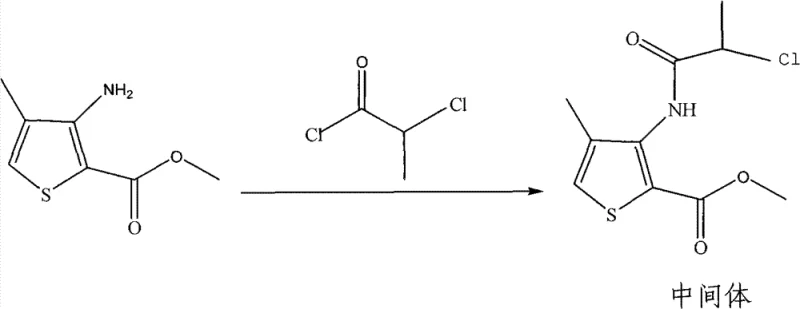
The cornerstone of this improved synthesis lies in the precise control of the initial amidation reaction, where 4-methyl-3-aminothiophene-2-methyl formate reacts with 2-chloropropionyl chloride. The selection of triethylamine as the acid scavenger is critical, as it effectively neutralizes the hydrochloric acid byproduct generated during the acylation, driving the equilibrium forward towards the desired amide intermediate. Operating at a controlled temperature range of 0-30°C is vital to suppress potential side reactions, such as the hydrolysis of the acid chloride or over-acylation, which could lead to difficult-to-remove impurities. The mechanistic advantage here is the formation of a soluble triethylamine hydrochloride salt that can be easily separated during the aqueous workup, unlike inorganic salts that might complicate filtration. This careful orchestration of reagent stoichiometry, specifically maintaining a molar ratio of roughly 1:1.2 between the amine and the acid chloride, ensures that the reaction proceeds to completion while minimizing the presence of unreacted starting materials in the crude mixture.
Furthermore, the impurity profile of the final product is tightly managed through the specific choice of solvents in the subsequent amination and salification steps. In the second stage, the use of DMF as a polar aprotic solvent facilitates the nucleophilic attack of n-propylamine on the chloro-intermediate, ensuring high conversion to the articaine base without requiring excessive heat that could degrade the thiophene ring. The final salification in acetone allows for the selective precipitation of the hydrochloride salt, leveraging the differential solubility of the product versus organic impurities. This crystallization step is crucial for achieving the reported purity of 99.3%, as it effectively excludes non-ionic byproducts and residual solvents. For quality control teams, this mechanism implies a robust process capable of consistently meeting stringent pharmacopeial standards, thereby reducing the risk of batch rejection and ensuring a steady supply of high-purity pharmaceutical intermediates for downstream formulation.
How to Synthesize Articaine Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the defined parameters to replicate the high yields and purity described in the patent data. The process begins with the preparation of the amide intermediate in dichloromethane, followed by a direct amination in DMF, and concludes with salt formation in acetone. Each transition between steps involves specific workup procedures, such as pH adjustment and solvent exchange, which are critical for removing byproducts like triethylamine hydrochloride before they interfere with subsequent reactions. Operators must monitor the exothermic nature of the amination step carefully to maintain the temperature between 45-55°C, preventing thermal degradation. The detailed standardized synthesis steps, including exact reagent quantities and stirring times, are outlined in the guide below to ensure reproducibility across different manufacturing scales.
- Perform amidation of 4-methyl-3-aminothiophene-2-methyl formate with 2-chloropropionyl chloride using triethylamine in dichloromethane at 0-30°C.
- Conduct amination of the resulting intermediate with n-propylamine in DMF solvent at 45-55°C to form the articaine base.
- Execute salification by reacting the articaine base with concentrated hydrochloric acid in acetone at 55-65°C to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of benzene and pyridine from the process inventory significantly reduces the regulatory overhead and insurance costs associated with handling Class 1 carcinogens and high-risk reagents. This shift not only simplifies the procurement of raw materials, as dichloromethane and DMF are commodity chemicals with stable global supply chains, but also mitigates the risk of production stoppages due to environmental compliance audits. Moreover, the mild reaction conditions eliminate the need for specialized high-pressure reactors or cryogenic cooling systems, allowing existing general-purpose chemical infrastructure to be utilized for production. This flexibility enhances asset utilization rates and lowers the barrier to entry for scaling production volumes to meet fluctuating market demands for local anesthetics.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substantial increase in overall yield and the reduction in waste disposal costs. By achieving yields of 85-90% in key steps compared to the sub-70% rates of older methods, the amount of expensive starting material required per kilogram of final product is drastically lowered. Additionally, the removal of toxic solvents like benzene eliminates the need for complex and energy-intensive incineration or specialized recycling processes, leading to significant operational expenditure savings. The use of triethylamine, while a reagent cost, is offset by its efficiency in driving the reaction to completion, reducing the need for reprocessing off-spec batches. These factors combine to create a leaner cost structure that improves margin potential in a competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on widely available and stable solvents such as dichloromethane, DMF, and acetone ensures that production is not vulnerable to the supply shocks often associated with specialty or highly regulated chemicals. The simplicity of the three-step sequence also shortens the total cycle time per batch, allowing for faster turnover and more responsive inventory management. This agility is crucial for maintaining continuity of supply for critical medications like Articaine, where shortages can have immediate clinical impacts. Furthermore, the robustness of the process against minor variations in reaction conditions means that technology transfer between different manufacturing sites can be executed with lower risk, diversifying the supply base and reducing single-point failures.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing route is markedly smaller, aligning with the increasing corporate sustainability goals of major pharmaceutical buyers. The absence of persistent organic pollutants like benzene simplifies wastewater treatment and reduces the volume of hazardous solid waste generated. Scalability is inherently supported by the ambient pressure and moderate temperature requirements, meaning that scaling from pilot plant to commercial tonnage does not require exponential increases in engineering complexity. This ease of scale-up ensures that suppliers can rapidly ramp up production capacity in response to market spikes without compromising on safety or quality, securing long-term partnerships with clients who prioritize responsible sourcing and reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and comparative analysis within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement officers assessing supplier capabilities. The answers provided reflect the specific advantages of the triethylamine-catalyzed pathway and its impact on final product quality and process safety.
Q: What are the critical safety advantages of this new synthesis route compared to traditional methods?
A: The patented method eliminates the use of highly toxic solvents like benzene and pyridine, replacing them with safer alternatives such as dichloromethane and DMF, thereby reducing occupational health risks and environmental compliance burdens.
Q: How does the choice of catalyst impact the yield of the amidation step?
A: Experimental data indicates that using triethylamine as the catalyst provides superior catalytic activity compared to inorganic bases like potassium carbonate, resulting in higher conversion rates and easier workup procedures.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild temperature conditions (0-65°C) and atmospheric pressure, avoiding the need for specialized high-pressure reactors, which makes it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Articaine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to optimized synthetic routes like the one described in CN102060840B requires a partner with deep technical expertise and proven manufacturing capability. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our facilities are equipped with the necessary corrosion-resistant reactors and solvent recovery systems to handle the specific chemistry of thiophene derivatives safely. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Articaine Hydrochloride meets the highest international standards, providing our clients with the confidence needed for regulatory filings and market launch.
We invite you to engage with our technical team to explore how this advanced synthesis method can be integrated into your supply chain to drive efficiency and quality. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic benefits specific to your volume requirements. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than assumptions. Let us collaborate to secure a sustainable and cost-effective supply of this critical anesthetic agent for the global market.
