Advanced Two-Step Synthesis of 2-Pentylanthraquinone for Industrial Hydrogen Peroxide Production
Advanced Two-Step Synthesis of 2-Pentylanthraquinone for Industrial Hydrogen Peroxide Production
The global demand for high-efficiency hydrogen peroxide production has driven significant innovation in the synthesis of its key carrier molecule, 2-pentylanthraquinone. Traditional manufacturing routes have long been plagued by environmental concerns and complex multi-step procedures, creating a critical bottleneck for scalable green chemistry. A breakthrough detailed in patent CN107602368B introduces a streamlined two-step methodology that fundamentally redefines the production landscape for this essential fine chemical intermediate. By leveraging advanced heterogeneous catalysis, this novel approach replaces hazardous liquid acids with regenerable solid catalysts, achieving theoretical atom utilization rates of 100% while drastically simplifying the downstream purification workflow. For R&D directors and procurement strategists alike, this technology represents a pivotal shift towards sustainable, cost-effective manufacturing that aligns perfectly with modern environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-pentylanthraquinone has relied heavily on the classical Friedel-Crafts acylation route, a process notorious for its operational complexity and severe environmental footprint. This traditional pathway typically necessitates a three-step sequence: initial synthesis of pentylbenzene, followed by acylation to form 2-(4-pentylbenzoyl)benzoic acid, and finally, a dehydration cyclization step requiring large quantities of oleum or concentrated sulfuric acid. The reliance on such corrosive media not only accelerates equipment degradation, leading to frequent maintenance downtime and capital expenditure, but also generates substantial volumes of acidic wastewater that require expensive neutralization and treatment protocols. Furthermore, the solubility limitations of older carriers like 2-ethylanthraquinone often force manufacturers into inefficient operating regimes with excessive hydrogenation and degradation, whereas the transition to 2-pentyl variants has been hindered by these prohibitive production costs and import dependencies.
The Novel Approach
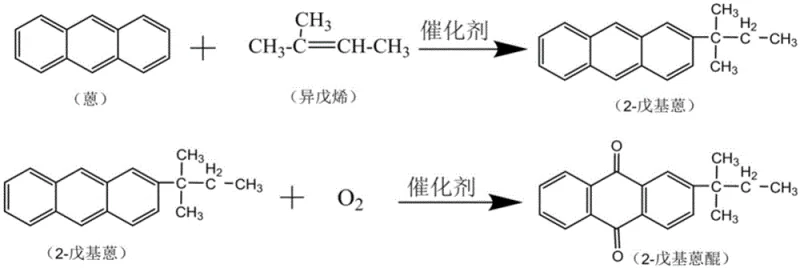
In stark contrast, the innovative two-step process outlined in the patent data offers a paradigm shift by utilizing direct alkylation and catalytic oxidation to bypass the need for corrosive acylation agents entirely. The first stage involves the direct alkylation of anthracene with isopentene over a solid Mg-MWW molecular sieve catalyst, a reaction that proceeds under controlled pressure and temperature to yield 2-pentylanthracene with high selectivity. This is immediately followed by a second oxidation stage where the intermediate is converted to the final quinone using molecular oxygen or air over a supported transition metal oxide catalyst. This methodology not only eliminates the generation of acidic waste streams but also enables the continuous regeneration and reuse of the solid catalysts, thereby transforming a linear, waste-generating process into a circular, resource-efficient system that significantly lowers the total cost of ownership for chemical manufacturers.
Mechanistic Insights into Mg-MWW Zeolite Alkylation and Supported Oxide Oxidation
The core of this technological advancement lies in the sophisticated design of the catalytic systems employed in both reaction stages, which ensure high conversion rates while minimizing byproduct formation. In the initial alkylation phase, the Mg-MWW molecular sieve acts as a solid acid catalyst where the isopentene substrate is protonated at the acid centers to form a reactive tert-amyl carbocation intermediate. This electrophilic species then attacks the anthracene ring in a highly regioselective manner, preferentially substituting at the 2-position to form 2-pentylanthracene, which serves as the rate-controlling step of the entire sequence. The use of a shape-selective zeolite framework prevents the formation of bulky poly-alkylated impurities, ensuring a cleaner reaction profile that simplifies subsequent separation processes and enhances the overall purity of the intermediate stream before it enters the oxidation reactor.
Following the alkylation, the oxidation mechanism leverages the redox properties of transition metals such as manganese, iron, or magnesium supported on a gamma-alumina carrier to facilitate the insertion of oxygen into the anthracene backbone. Under mild thermal conditions ranging from 50°C to 200°C, the supported metal oxides activate molecular oxygen to generate reactive oxygen species that selectively oxidize the central ring of the 2-pentylanthracene molecule. This heterogeneous catalytic cycle avoids the safety hazards associated with high-concentration hydrogen peroxide or harsh chemical oxidants like chromic acid, which are prone to causing runaway reactions. The robust nature of the gamma-alumina support ensures thermal stability during the exothermic oxidation, allowing for precise temperature control that suppresses over-oxidation and ring-opening degradation, ultimately delivering a high-purity 2-pentylanthraquinone product suitable for sensitive hydrogen peroxide working solutions.
How to Synthesize 2-Pentylanthraquinone Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and catalyst longevity, particularly regarding the pressure and temperature profiles in the alkylation reactor. The process begins by charging anthracene and isopentene into a sealed high-pressure vessel along with the Mg-MWW catalyst, where the system is purged with nitrogen and heated to a preferred range of 200°C to 300°C under a pressure of approximately 2.5 MPa. Once the alkylation is complete, the mixture undergoes vacuum distillation to separate the liquid product from the solid catalyst and unreacted starting materials, allowing the catalyst to be recycled directly back into the reactor. The resulting 2-pentylanthracene is then dissolved in a solvent such as mesitylene and subjected to oxidation in the presence of the supported metal oxide catalyst, with oxygen or air introduced at a controlled flow rate to maintain safe and efficient conversion kinetics.
- Perform liquid-phase alkylation of anthracene and isopentene using Mg-MWW molecular sieve catalyst at 200-300°C and 2.5 MPa.
- Separate the intermediate 2-pentylanthracene via vacuum distillation to recover unreacted anthracene and catalyst.
- Oxidize 2-pentylanthracene using oxygen or air over a supported metal oxide catalyst (e.g., Mn/Fe on gamma-Al2O3) at 50-200°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this two-step catalytic technology translates into tangible strategic advantages that extend far beyond simple unit cost metrics. By eliminating the dependency on imported 2-pentylanthraquinone and the volatile pricing of specialized acylation reagents, manufacturers can secure a more stable and predictable supply chain for their hydrogen peroxide production lines. The shift from liquid acid catalysts to solid, regenerable molecular sieves removes the logistical burden of handling and disposing of hazardous sulfuric acid waste, thereby reducing regulatory compliance costs and minimizing the risk of environmental penalties that can disrupt operations. Furthermore, the simplified two-step workflow reduces the overall processing time and energy consumption compared to the traditional three-step Friedel-Crafts route, enabling faster throughput and improved asset utilization rates across the production facility.
- Cost Reduction in Manufacturing: The elimination of concentrated sulfuric acid and oleum from the process workflow removes the significant costs associated with acid procurement, storage, and neutralization, while the ability to regenerate solid catalysts multiple times drastically lowers the recurring expense of catalytic materials. This structural change in the bill of materials allows for a leaner cost base that is less susceptible to fluctuations in raw material markets, providing a competitive edge in pricing negotiations with downstream hydrogen peroxide producers who are increasingly demanding greener supply chains.
- Enhanced Supply Chain Reliability: Utilizing readily available bulk commodities like anthracene and isopentene as primary feedstocks reduces reliance on complex, multi-sourced intermediate supply chains that are prone to disruption. The robustness of the solid catalyst system ensures consistent production output without the frequent shutdowns required for cleaning corroded equipment, thereby guaranteeing a steady flow of high-purity intermediates to meet the continuous demand of large-scale peroxide plants and mitigating the risk of stockouts that could halt customer production lines.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction system facilitates straightforward scale-up from pilot batches to commercial tonnage without the engineering challenges posed by handling massive volumes of corrosive liquids. This scalability is matched by superior environmental performance, as the process generates minimal hazardous waste and utilizes oxygen from air as the primary oxidant, aligning perfectly with global sustainability goals and helping corporate partners meet their carbon reduction and green chemistry targets without compromising on production volume.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this novel two-step synthesis pathway for 2-pentylanthraquinone. These answers are derived directly from the experimental data and mechanistic descriptions provided in the patent literature, offering clarity on catalyst selection, reaction safety, and product quality assurance for technical teams evaluating this technology for adoption.
Q: What are the advantages of the two-step method over traditional Friedel-Crafts acylation?
A: The two-step method eliminates the use of corrosive concentrated sulfuric acid and oleum, significantly reducing wastewater treatment costs and equipment corrosion risks while improving atom economy.
Q: Can the catalysts used in this process be recycled?
A: Yes, both the Mg-MWW molecular sieve for alkylation and the supported metal oxides for oxidation are solid heterogeneous catalysts that can be separated and regenerated for repeated use.
Q: Why is 2-pentylanthraquinone preferred over 2-ethylanthraquinone for hydrogen peroxide production?
A: 2-Pentylanthraquinone exhibits higher solubility in working fluids, allowing for higher hydrogen efficiency and reduced degradation products compared to 2-ethylanthraquinone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pentylanthraquinone Supplier
As the chemical industry pivots towards more sustainable and efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands at the forefront as a premier partner capable of translating complex patent technologies into commercial reality. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that the transition from laboratory discovery to industrial application is seamless and risk-mitigated. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-pentylanthraquinone meets the exacting standards required for high-efficiency hydrogen peroxide working solutions, providing our clients with the confidence needed to optimize their own downstream processes.
We invite forward-thinking organizations to collaborate with us to unlock the full potential of this green synthesis technology for their specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current production volumes, and ask for specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can enhance your supply chain resilience and profitability.
