Revolutionizing SGLT2 Inhibitor Production: A Deep Dive into High-Purity Canagliflozin Intermediate Manufacturing
The pharmaceutical landscape for Type 2 diabetes management has been significantly transformed by the advent of SGLT2 inhibitors, with Canagliflozin standing out as a cornerstone therapy. The commercial viability of this blockbuster drug relies heavily on the efficient and pure synthesis of its key precursors. Patent CN111560004A introduces a groundbreaking preparation method for a high-purity Canagliflozin intermediate, addressing long-standing challenges in impurity control and process stability. This technical insight report analyzes the proprietary methodology disclosed in the patent, highlighting its potential to redefine manufacturing standards for high-purity pharmaceutical intermediates. By shifting the purification burden to an earlier stage in the synthesis, this approach mitigates the risks associated with late-stage purification failures. The structural complexity of the target molecule demands precision, as illustrated by the final API structure below.
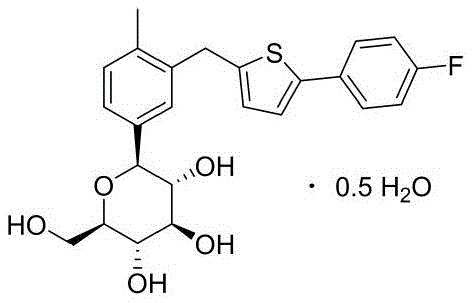
For R&D Directors, the implication is clear: access to intermediates with defined impurity profiles accelerates clinical development and regulatory approval. For Procurement Managers, the robustness of the described crystallization steps suggests a more predictable cost structure, reducing the volatility associated with yield losses. Finally, Supply Chain Heads will appreciate the scalability inherent in the solvent systems chosen, which favor industrial availability over laboratory exclusivity. This report dissects the chemical logic and commercial advantages of this novel pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Canagliflozin intermediates has been plagued by the inherent instability and poor crystallization behavior of the target hemiacetal structure, often referred to as Intermediate II. Conventional routes, such as those disclosed in prior art patents, typically attempt to construct this complex scaffold in a single pot or through direct coupling that yields a crude product rife with stereoisomers and byproducts. When using highly reactive reagents like n-butyllithium at low temperatures, the margin for error is razor-thin; any deviation in thermal control leads to significant formation of regioisomers that are structurally similar and notoriously difficult to separate. Furthermore, the direct crystallization of Intermediate II from reaction mixtures is often inefficient, requiring excessive solvent volumes and resulting in substantial mother liquor losses. This not only inflates the cost of goods sold but also creates environmental burdens due to high solvent waste. The reliance on direct purification of the final intermediate means that any impurity generated in the coupling step is carried forward, potentially compromising the safety profile of the final API.
The Novel Approach
The methodology presented in CN111560004A ingeniously circumvents these bottlenecks by introducing a stable, crystalline precursor known as Intermediate I. Instead of forcing the immediate formation of the cyclic hemiacetal, the process first generates an open-chain ketone derivative which exhibits exceptional crystallization properties. This strategic pivot allows manufacturers to perform a rigorous purification at the Intermediate I stage, where impurities can be effectively rejected into the mother liquor through standard crystallization techniques. The visual representation of this key intermediate highlights the structural features that enable this superior solid-state behavior.
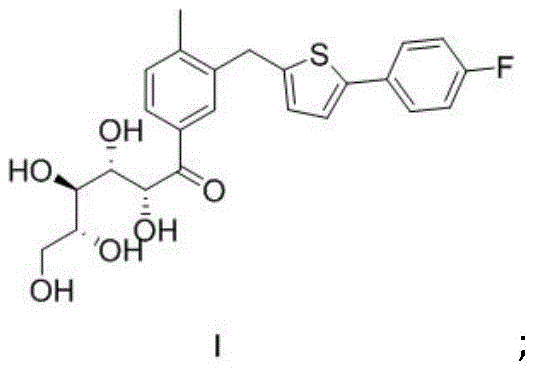
Once high-purity Intermediate I is secured, it is subsequently converted to the desired Intermediate II under controlled acidic conditions. This two-step sequence decouples the carbon-carbon bond formation from the cyclization event, granting process chemists independent control over each transformation. The result is a dramatic improvement in the overall purity profile without the need for complex chromatographic separations. For a reliable pharmaceutical intermediates supplier, this translates to a process that is forgiving of minor operational variances and consistently delivers material meeting stringent specifications. The ability to isolate a stable solid intermediate also enhances storage and logistics, reducing the risk of degradation during transport between manufacturing sites.
Mechanistic Insights into Thiophene-Glucose Condensation and Cyclization
The core of this innovation lies in the precise manipulation of reaction thermodynamics and kinetics during the condensation phase. The process initiates with the deprotonation of a thiophene derivative using a strong base, such as an organolithium or Grignard reagent, within an inert solvent system comprising tetrahydrofuran and toluene. This mixture is carefully maintained at cryogenic temperatures, typically ranging from -80°C to 0°C, to ensure the formation of the nucleophilic species without triggering decomposition pathways. The subsequent addition of 2,3,4,6-tetra-O-(trimethylsilyl)-D-gluconolactone must be executed with meticulous dosing control to manage the exotherm and prevent local hot spots that could racemize the sensitive sugar moiety. The choice of solvent ratio is not arbitrary; the specific volume ratio of THF to toluene influences the solubility of the transition states and the subsequent precipitation of the product. Following the condensation, the reaction is quenched with a strong acid and water, which serves the dual purpose of neutralizing the base and hydrolyzing the silyl protecting groups to reveal the free hydroxyls of Intermediate I.
Following the isolation of Intermediate I, the conversion to Intermediate II involves an acid-catalyzed cyclization in methanol. This step leverages the equilibrium between the open-chain ketone and the cyclic hemiacetal forms. By employing a strong acid catalyst in a protic solvent, the reaction drives the equilibrium towards the thermodynamically stable cyclic form. The mechanistic elegance here is that because the starting material (Intermediate I) is already of high purity, the resulting equilibrium mixture is similarly clean, minimizing the formation of anomeric impurities. The comprehensive reaction scheme below illustrates the flow from the halogenated thiophene starting material through the ketone intermediate to the final cyclic product.
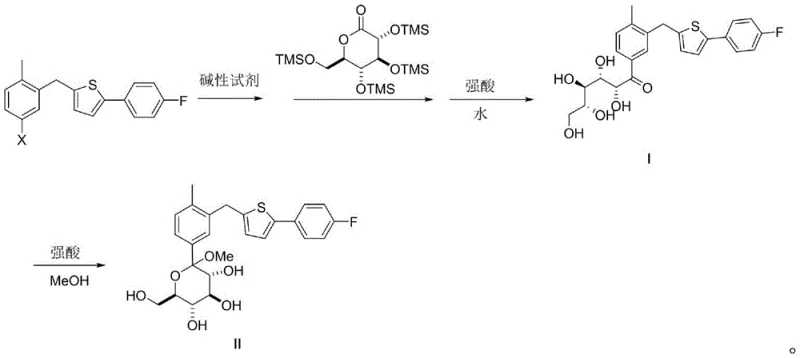
Understanding these mechanistic nuances is vital for cost reduction in pharmaceutical intermediates manufacturing. By optimizing the stoichiometry of the base and the acid quench, manufacturers can minimize reagent costs while maximizing yield. Furthermore, the separation of phases after the quench is facilitated by the specific solvent choices, allowing for a clean split between the organic product layer and the aqueous waste layer. This reduces the need for extensive washing cycles, saving both time and resources. The robustness of this chemistry ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved with high confidence, as the critical quality attributes are built into the process design rather than relying solely on end-product testing.
How to Synthesize Canagliflozin Intermediate Efficiently
Implementing this synthesis requires adherence to strict operational parameters to replicate the high purity reported in the patent data. The process begins with the preparation of the reaction vessel under an inert nitrogen atmosphere, ensuring that moisture and oxygen are excluded to prevent reagent degradation. The thiophene substrate is dissolved in the designated solvent blend, and the temperature is lowered to the specified cryogenic range before the slow addition of the base. Once the intermediate anion is formed, the gluconolactone derivative is introduced, and the mixture is allowed to stir until conversion is complete as monitored by HPLC. The quenching step is critical; the acid must be added dropwise to control gas evolution and heat generation. After phase separation and concentration, the crystallization of Intermediate I is induced by the addition of anti-solvents, followed by filtration and drying.
- Condense thiophene compound with gluconolactone derivative under inert low-temperature conditions to form Intermediate I.
- Purify Intermediate I via crystallization using specific solvent systems like toluene and heptane.
- Convert Intermediate I to Final Intermediate II using strong acid in methanol followed by extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages for organizations seeking to optimize their supply chains for SGLT2 inhibitors. The primary value driver is the significant enhancement in process reliability, which directly correlates to supply continuity. Traditional methods that struggle with purity often lead to batch failures or the need for reprocessing, both of which introduce unpredictable delays into the production schedule. By contrast, the crystallization-driven purification of Intermediate I provides a robust checkpoint that ensures only material meeting strict criteria proceeds to the next stage. This predictability allows procurement teams to plan inventory levels more accurately and reduce the need for safety stock buffers. Additionally, the use of common industrial solvents like toluene, methanol, and heptane simplifies the sourcing of raw materials, mitigating the risk of supply disruptions associated with specialty chemicals.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps results in substantial cost savings. Traditional routes often require expensive silica gel columns or preparative HPLC to remove stubborn impurities, which are costly to operate and scale. This new method relies on crystallization, a unit operation that is inherently cheaper and more scalable in a multi-ton environment. Furthermore, the improved yield stability means that less starting material is wasted per kilogram of final product, driving down the effective cost of goods. The ability to recover and recycle solvents from the crystallization mother liquors further enhances the economic efficiency of the process, contributing to a leaner manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of the chemical process translates directly into shorter and more reliable lead times. Because the reaction conditions are less sensitive to minor fluctuations in temperature or addition rates compared to direct lithiation methods, the failure rate of production batches is drastically reduced. This consistency is crucial for maintaining the continuous flow of materials required for downstream API synthesis. Suppliers utilizing this technology can offer more dependable delivery schedules, reducing the administrative burden on supply chain managers who otherwise have to constantly expedite orders to cover for production shortfalls. The stability of Intermediate I also allows for strategic stockpiling, decoupling the production of the precursor from the immediate demand for the final intermediate.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard equipment and well-understood unit operations. The avoidance of hazardous or exotic reagents simplifies the safety protocols required for large-scale manufacturing, reducing the capital expenditure needed for specialized containment systems. From an environmental standpoint, the reduced solvent consumption and the potential for solvent recycling align with modern green chemistry principles. This helps manufacturing sites maintain compliance with increasingly stringent environmental regulations regarding volatile organic compound emissions and waste disposal. The overall process mass intensity is improved, making the production of high-purity pharmaceutical intermediates more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing supply network.
Q: Why is Intermediate I critical for purity control?
A: Intermediate I possesses a ketone structure with superior crystallization properties compared to the hemiacetal Intermediate II, allowing impurities to be removed effectively before the final cyclization step.
Q: What are the safety considerations for the low-temperature step?
A: The process requires strict temperature control between -80°C and 0°C using cryogenic cooling systems to manage the exothermic nature of the organolithium or Grignard addition safely.
Q: How does this method improve supply chain reliability?
A: By utilizing robust crystallization steps and common solvents like toluene and methanol, the method reduces dependency on exotic reagents and simplifies scale-up operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Canagliflozin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We understand that stringent purity specifications are non-negotiable in the pharmaceutical sector, which is why our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality ensures that the intermediates we supply seamlessly integrate into your downstream processes, minimizing the risk of deviations during API manufacturing.
We invite you to collaborate with us to optimize your supply chain for SGLT2 inhibitors. By leveraging our technical expertise, you can secure a stable source of high-quality materials that support your long-term business goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency and reduce your overall procurement costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
