Advanced Photochemical Synthesis of Cefuroxime Sodium E-Type Impurity Intermediates for Global Pharma Supply Chains
The pharmaceutical industry faces rigorous demands for impurity profiling, particularly for widely used antibiotics like cefuroxime sodium. Patent CN109553629B introduces a groundbreaking preparation method for the E-type impurity compound of cefuroxime sodium intermediates, addressing a critical gap in quality control and regulatory compliance. This technology leverages a unique photochemical approach to overcome the thermodynamic barriers that typically favor the Z-isomer during synthesis. For R&D directors and quality assurance teams, accessing high-purity reference standards is essential for validating analytical methods and ensuring batch consistency. The disclosed method achieves purity levels greater than 97% through a streamlined two-step process involving phosphorus pentachloride activation under controlled illumination. This innovation not only simplifies the acquisition of EP Impurity F but also provides a robust pathway for synthesizing the corresponding finished drug impurity, EP Impurity E. By mastering this specific isomeric form, manufacturers can better support the declaration and registration of cefuroxime sodium products in global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of cefuroxime sodium relies on the Z-configured side chain, and the formation of E-type impurities is often an uncontrolled byproduct rather than a targeted outcome. Conventional liquid phase separation technologies struggle to isolate these E-isomers efficiently because the thermodynamic equilibrium heavily favors the Z-configuration. When standard acyl chlorinating agents like thionyl chloride are used without specific energetic inputs, the resulting mixture contains unpredictable ratios of isomers. This necessitates extensive and costly purification steps, such as preparative HPLC, to obtain the E-type impurity in quantities sufficient for analytical validation. Furthermore, the instability of the beta-lactam ring under harsh acidic conditions used in traditional activation can lead to degradation, further reducing overall yield and complicating the impurity spectrum. These inefficiencies create bottlenecks in the supply chain for reference standards, delaying critical quality control processes.
The Novel Approach
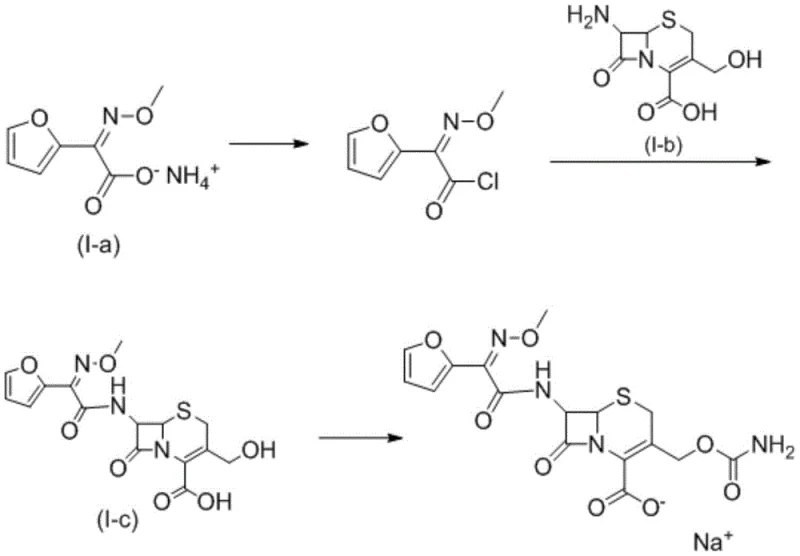
The patented method revolutionizes this landscape by introducing illumination as a critical process parameter to drive stereochemical selectivity. Instead of relying solely on thermal energy or chemical catalysts, the process utilizes specific light intensities ranging from 5000 Lx to 10000 Lx during the formation of the acyl chloride intermediate. This photochemical input provides the necessary energy to overcome the rotational barrier of the oxime double bond, facilitating the conversion from the Z-form to the desired E-form. The subsequent coupling reaction with the cephalosporin nucleus is also conducted under illumination, ensuring that the E-configuration is maintained throughout the amide bond formation. This targeted approach eliminates the need for complex separation of isomers post-reaction, as the reaction itself is biased towards the target impurity structure. The result is a direct, high-yield pathway to the E-type intermediate that is both economically viable and technically superior to random byproduct harvesting.
Mechanistic Insights into Photochemical Isomerization and Coupling
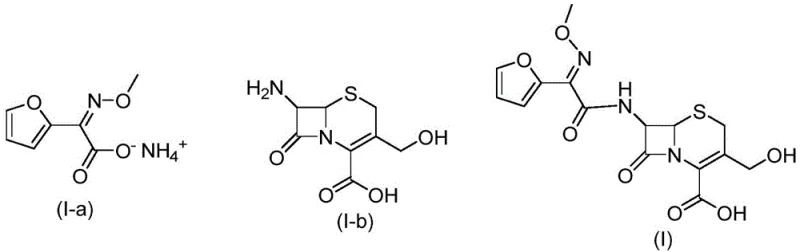
The core mechanism of this invention lies in the photo-induced isomerization of the methoxyimino group during the activation phase. When the ammonium acetate salt of the furan side chain reacts with phosphorus pentachloride, the formation of the reactive acyl chloride species occurs concurrently with light absorption. The photons absorbed by the reaction mixture excite the electrons in the C=N double bond, allowing for rotation that would otherwise be restricted at the operating temperatures of 30-50°C. This dynamic equilibrium is shifted towards the E-isomer, which is then trapped by the nucleophilic attack of the 7-amino cephalosporanic acid derivative. The use of phosphorus pentachloride is particularly effective here as it generates the acyl chloride rapidly, minimizing the time the sensitive intermediate is exposed to potential hydrolysis before coupling. The precise control of illuminance, preferably between 7000 Lx and 8000 Lx, ensures that the energy input is sufficient for isomerization without causing photodegradation of the sensitive beta-lactam core or the furan ring.
Impurity control is further enhanced by the specific pH management during the coupling stage. The reaction is maintained at a pH of 7.5 to 8.5 using inorganic bases like sodium hydroxide or sodium carbonate. This slightly alkaline environment is crucial for neutralizing the hydrochloric acid generated during the amide bond formation while preventing the base-catalyzed opening of the beta-lactam ring. By carefully controlling the addition rate of the acyl chloride solution into the aqueous suspension of the nucleus, the local concentration of the reactive species is kept low, minimizing side reactions such as polymerization or esterification at the hydroxymethyl group. The final isolation step involves acidifying the aqueous layer to pH 1.5-2.5, which protonates the carboxylic acid group and reduces the solubility of the target compound, causing it to precipitate as a high-purity solid. This crystallization-driven purification effectively excludes remaining Z-isomers and other polar byproducts.
How to Synthesize Cefuroxime Sodium E-Type Impurity Efficiently
The synthesis protocol outlined in the patent offers a reproducible method for generating this critical reference material. The process begins with the suspension of the Z-configured ammonium salt in a suitable organic solvent such as dichloromethane or ethyl acetate. Phosphorus pentachloride is added, and the mixture is heated to reflux or maintained at 35-45°C under strong illumination for approximately 3 to 4 hours. Following the formation of the acyl chloride, the reaction mixture is quenched with ice water, and the organic layer is separated. This organic phase containing the E-enriched acyl chloride is then added dropwise to an aqueous solution of the cephalosporin nucleus, which has been solubilized with base. The coupling proceeds under continued illumination at temperatures between 20-45°C for 2 to 3 hours. Finally, the product is isolated by acidification and filtration.
- React (Z)-2-(furan-2-yl)-2-(methoxyimino) ammonium acetate with phosphorus pentachloride under illumination (5000-10000 Lx) at 30-50°C to form the acyl chloride.
- Couple the resulting acyl chloride solution with 7-amino-3-(hydroxymethyl)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid under continued illumination.
- Maintain pH 7.5-8.5 during coupling, then acidify the aqueous layer to pH 1.5-2.5 to precipitate the pure E-type impurity compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical synthesis route presents significant strategic benefits regarding cost structure and supply reliability. The primary advantage lies in the drastic simplification of the downstream processing requirements. Because the reaction selectively produces the E-isomer with high specificity, the need for expensive and time-consuming chromatographic purification is virtually eliminated. This reduction in processing complexity translates directly into lower operational expenditures and reduced consumption of high-grade solvents and silica-based stationary phases. Furthermore, the use of commodity chemicals like phosphorus pentachloride and sodium hydroxide ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic catalysts or specialized reagents.
- Cost Reduction in Manufacturing: The elimination of complex separation technologies significantly lowers the cost of goods sold for impurity standards. By achieving high crude purity through reaction control rather than post-reaction cleanup, manufacturers can reduce labor hours and equipment usage time. The ability to use standard glass-lined or stainless steel reactors equipped with simple lighting fixtures, rather than specialized photo-flow reactors, further minimizes capital expenditure requirements for scaling up production.
- Enhanced Supply Chain Reliability: The robustness of this method ensures consistent batch-to-batch quality, which is critical for maintaining the supply of reference standards needed for regulatory testing. Since the process parameters (light intensity, temperature, pH) are easily monitored and controlled, the risk of batch failure due to isomer contamination is minimized. This reliability allows for longer production runs and larger inventory buffers, ensuring that pharmaceutical clients never face shortages of critical impurity markers during their own quality control cycles.
- Scalability and Environmental Compliance: The process is inherently scalable as it does not rely on photon flux limitations typical of small-scale lab photochemistry; industrial lighting arrays can easily replicate the required lux levels in large vessels. Additionally, the aqueous workup and precipitation method generates less hazardous waste compared to solvent-intensive chromatography. The ability to recycle the organic solvent layer after quenching further aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process and simplifying waste disposal compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing quality control workflows. The answers are derived directly from the experimental data and process descriptions provided in the patent documentation.
Q: Why is the E-type impurity of cefuroxime sodium difficult to synthesize traditionally?
A: Traditional methods favor the thermodynamically stable Z-isomer. Conventional acyl chlorination often fails to induce sufficient Z-to-E isomerization, leading to low yields of the E-impurity and requiring complex, costly chromatographic separation to meet pharmacopoeial standards.
Q: How does the photochemical method improve purity compared to standard liquid phase separation?
A: By utilizing specific illumination intensities (e.g., 7000-8000 Lx) during the activation and coupling steps, the process drives the stereochemical conversion towards the E-configuration. This results in crude products with purity exceeding 97%, significantly reducing the need for downstream purification.
Q: Is this synthesis method scalable for commercial impurity reference standard production?
A: Yes, the method utilizes commercially available reagents like phosphorus pentachloride and standard organic solvents. The operation involves simple temperature control and pH adjustment, making it highly adaptable for large-scale manufacturing of reference standards required for quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Sodium E-Type Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex photochemical processes can be translated into robust manufacturing operations. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of cefuroxime sodium intermediate meets the exacting standards required for EP and USP compliance. Our commitment to technical excellence ensures that our clients receive materials that facilitate accurate analytical method validation and seamless regulatory filings.
We invite you to collaborate with us to optimize your supply chain for antibiotic intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can enhance your product quality and operational efficiency.
