Scalable Vitronectin Manufacturing: Advanced Acetal Protection Strategy for High-Purity Cosmetic Actives
Introduction to Next-Generation Vitronectin Synthesis
The landscape of cosmetic active ingredient manufacturing is undergoing a significant transformation driven by the need for higher purity and scalable processes. Patent CN113735810A introduces a groundbreaking method for synthesizing Vitronectin (also known as Boseine), a potent xylose derivative renowned for its anti-wrinkle and skin-restructuring properties. This technology addresses critical bottlenecks in traditional synthesis routes by employing a strategic acetal/ketal protection and reduction sequence. By leveraging the specific steric characteristics of the intermediate molecules, this novel approach achieves high stereoselectivity while completely bypassing the need for resource-intensive column chromatography. For industry leaders seeking a reliable cosmetic active ingredients supplier, this patent represents a pivotal shift towards more efficient and environmentally conscious manufacturing paradigms that align with modern green chemistry principles.
The core innovation lies in the manipulation of solubility profiles through selective protection. Traditional methods often struggle with the separation of boron-containing byproducts generated during reduction. In contrast, this new methodology converts key hydroxyl groups into lipophilic acetals or ketals early in the sequence. This modification not only stabilizes the molecule against unwanted side reactions but also fundamentally alters its physical properties, enabling the removal of water-soluble impurities through straightforward liquid-liquid extraction. This strategic intervention simplifies the downstream processing workflow, reduces solvent consumption, and enhances the overall yield of the bioactive S-configuration isomer, making it an ideal candidate for cost reduction in cosmetic active ingredients manufacturing.
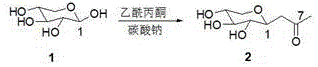
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, such as those disclosed in WO02/051828A1 and subsequent improvements by the Euraila team, have long dominated the production of C-glycoside derivatives like Vitronectin. These conventional routes typically involve the direct condensation of D-xylose with acetylacetone followed immediately by reduction using sodium borohydride. While effective in generating the carbon skeleton, these methods suffer from significant purification challenges. The use of sodium borohydride inevitably generates boric acid as a byproduct, which has a strong tendency to form stable complexes with the multiple hydroxyl groups present on the sugar moiety. These borate esters are notoriously difficult to separate from the desired product, often necessitating the use of column chromatography. For commercial operations, reliance on column chromatography is a major liability; it limits batch sizes, consumes vast quantities of silica gel and solvents, and creates substantial hazardous waste, thereby inflating production costs and complicating regulatory compliance for high-purity cosmetic actives.
The Novel Approach
The methodology described in CN113735810A circumvents these historical limitations through a clever reordering of synthetic steps. Instead of immediate reduction, the process first selectively protects the 3,4-dihydroxy groups of the initial condensation product. This protection step transforms the highly polar sugar derivative into a more lipophilic species. This change in polarity is the key enabler for purification; it allows manufacturers to wash away water-soluble impurities, including residual salts and polar byproducts, using simple aqueous workups. Furthermore, the protected intermediate exhibits superior chemical stability, preventing degradation during the subsequent reduction phase. By decoupling the protection and reduction steps, the process avoids the formation of tenacious boron complexes that plague earlier methods. This results in a cleaner reaction profile that can be purified via crystallization or standard extraction, facilitating the commercial scale-up of complex cosmetic intermediates without the burden of chromatographic purification.
Mechanistic Insights into Stereoselective Reduction and Protection
From a mechanistic perspective, the success of this synthesis relies heavily on the precise control of stereochemistry and solubility. The initial condensation of D-xylose with acetylacetone yields a beta-xyloside intermediate. The critical innovation occurs in the protection step, where reagents such as 2,3-butanedione or various orthoesters react selectively with the 3 and 4-position hydroxyl groups. This selectivity is driven by the relatively low steric hindrance at the 4-position compared to other sites on the pyranose ring. The resulting acetal or ketal ring locks the conformation of the sugar backbone, providing a rigid scaffold for the subsequent reduction. This rigidity is crucial because it minimizes conformational flexibility that could lead to non-selective reduction outcomes. The lipophilic nature of the protected intermediate also ensures compatibility with organic solvents like dichloromethane or toluene, further enhancing the efficiency of impurity removal.
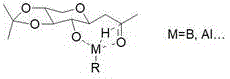
The reduction step is equally sophisticated, utilizing the inherent directing effects of the remaining free hydroxyl group at the 2-position. When a reducing agent such as lithium aluminum hydride or diisobutylaluminum hydride is introduced, the 2-OH group coordinates with the metal center of the reductant. This coordination creates a cyclic transition state that directs the hydride attack specifically to one face of the ketone carbonyl at the 7-position. This intramolecular delivery mechanism ensures that the reduction proceeds with high stereoselectivity, predominantly yielding the desired S-configuration alcohol. Achieving high diastereomeric excess is vital for biological activity, as the S-isomer of Vitronectin is significantly more potent in stimulating glycosaminoglycan synthesis than its R-counterpart. By engineering the molecule to exploit this directing effect, the process guarantees a high-quality impurity profile, reducing the burden on quality control laboratories and ensuring consistent batch-to-batch performance for reducing lead time for high-purity cosmetic actives.
How to Synthesize Vitronectin Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent stoichiometry to maximize yield and stereoselectivity. The process begins with the alkaline condensation of xylose and acetylacetone, followed by the critical protection step using acidic catalysts. The subsequent reduction must be performed under controlled temperatures to maintain the integrity of the protecting groups while ensuring complete conversion of the ketone. Finally, mild acidic hydrolysis removes the protecting groups to reveal the final bioactive molecule. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures derived from the patent examples, are outlined below to guide process development teams.
- Condense D-xylose with acetylacetone under alkaline conditions to form the beta-xyloside intermediate.
- Protect the 3,4-dihydroxy groups using a ketalizing agent to enhance lipophilicity and facilitate impurity removal.
- Perform stereoselective reduction of the ketone group guided by the 2-OH group to achieve S-configuration.
- Execute acidic deprotection and crystallization to isolate the final Vitronectin product without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acetal protection strategy offers profound economic and operational benefits. The most significant advantage is the elimination of column chromatography, which is traditionally the most expensive and time-consuming unit operation in fine chemical synthesis. By replacing chromatography with extraction and crystallization, manufacturers can drastically reduce solvent usage, lower waste disposal costs, and increase throughput capacity. This shift from batch-limited purification to continuous-friendly separation techniques directly translates to improved margins and a more resilient supply chain. Additionally, the use of readily available starting materials like D-xylose and acetylacetone ensures raw material security, mitigating risks associated with specialty reagent shortages.
- Cost Reduction in Manufacturing: The removal of column chromatography eliminates the need for expensive silica gel and the large volumes of elution solvents required for purification. This simplification of the downstream process significantly lowers the variable cost per kilogram of the final product. Furthermore, the ability to remove impurities via aqueous extraction reduces the complexity of the workup, saving labor hours and energy consumption associated with solvent recovery. The high stereoselectivity also means less material is lost to inactive isomers, improving the overall mass balance and atom economy of the process.
- Enhanced Supply Chain Reliability: The robustness of the acetal protection method makes the supply chain less vulnerable to disruptions. Since the process does not rely on specialized chromatographic columns or hard-to-source purification media, it can be easily replicated across different manufacturing sites. The stability of the protected intermediates also allows for potential storage or transport between steps if multi-site manufacturing is required. This flexibility ensures consistent availability of high-purity cosmetic actives even during periods of high market demand or logistical constraints.
- Scalability and Environmental Compliance: Scaling a chromatographic process is notoriously difficult due to pressure drop and bed compression issues, whereas extraction and crystallization scale linearly with vessel size. This new method is inherently designed for large-scale production, allowing for seamless transition from pilot plant to commercial tonnage. Moreover, the reduction in hazardous solvent waste and the avoidance of boron-containing sludge align with increasingly stringent environmental regulations. This eco-friendly profile enhances the sustainability credentials of the final product, a key factor for major cosmetic brands committed to green sourcing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process feasibility and product quality. Understanding these details is essential for technical teams evaluating the integration of this route into existing production lines.
Q: How does the new method improve purification compared to prior art?
A: Unlike traditional methods requiring column chromatography due to boric acid impurities, this method uses acetal protection to make intermediates fat-soluble, allowing water-soluble impurities to be removed via simple extraction.
Q: What ensures the high stereoselectivity of the reduction step?
A: The process utilizes the directing effect of the 2-position hydroxyl group during reduction, ensuring the 7-position ketone is reduced specifically to the S-configuration hydroxyl.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, by eliminating column chromatography and relying on extraction and crystallization, the process is significantly more scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitronectin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the acetal protection synthesis route for producing high-quality Vitronectin. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical S-configuration ratio, guaranteeing that every batch meets the highest standards for cosmetic and pharmaceutical applications. We are committed to delivering excellence in every gram of material we produce.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this valuable active ingredient.
