Advanced Synthesis of Alpha-Oximido Aliphatic Ketones for Commercial Scale-Up
Advanced Synthesis of Alpha-Oximido Aliphatic Ketones for Commercial Scale-Up
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the urgent need for processes that balance high purity with operational efficiency. A pivotal advancement in this domain is documented in Chinese Patent CN101693671B, which discloses a highly optimized preparation method for alpha-oximido aliphatic ketones. This technology represents a significant departure from traditional multi-cycle protocols, introducing a streamlined single-pass nitrosation-oximation reaction that drastically reduces experimental periods while maintaining exceptional product quality. For R&D directors and procurement specialists seeking reliable pharma intermediates suppliers, understanding the mechanistic nuances of this patent is crucial, as it offers a pathway to cost reduction in pharmaceutical intermediates manufacturing through simplified unit operations and enhanced solvent recovery strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the state-of-the-art synthesis of alpha-oximinoketones was heavily reliant on methodologies exemplified by US Patent 3,697,597, which, while effective in achieving high purity, suffered from severe procedural inefficiencies. The conventional approach necessitated a tedious repetitive cycle where the nitrosation and oximation reactions had to be performed up to nine distinct times to drive the conversion of aliphatic ketones to completion. This multi-step recursion not only consumed excessive amounts of reagents and energy but also required complex experimental setups involving repeated alkaline extractions and acidifications, making the process inherently unsuitable for large-scale industrial application. Furthermore, the reliance on vacuum distillation for purification in older methods introduced additional thermal stress on the product and extended the overall production timeline, creating bottlenecks that hindered the ability to meet tight supply chain deadlines for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in CN101693671B achieves total quantity control through a sophisticated gradient addition of acid catalysts combined with real-time monitoring of reaction conditions. By reducing the requirement for repeated nitrosation-oximation cycles from nine iterations down to a single, highly efficient pass, the new method shortens the preparation cycle significantly, reportedly by more than 85%. This breakthrough is achieved by dissolving the aliphatic ketone in solvents like isopropyl ether or n-butyl ether and introducing methyl nitrite gas under strictly controlled pH conditions that shift progressively from 1.5 to 3.0. The result is a process that not only simplifies the operational workflow but also facilitates the direct crystallization of the product from the acidified layer, yielding white scaly crystals of superior purity without the need for energy-intensive distillation steps.
Mechanistic Insights into Acid-Catalyzed Nitrosation-Oximation
The core of this technological leap lies in the precise management of the nitrosation-oximation mechanism, which is fundamentally driven by the in situ generation and immediate utilization of alkyl nitrites. The process initiates with the preparation of methyl nitrite gas, typically generated by reacting sodium nitrite with methanol in the presence of dilute sulfuric acid, ensuring a continuous and uniform supply of the nitrosating agent.

Following generation, the methyl nitrite gas is introduced into the reaction vessel containing the aliphatic ketone substrate. The reaction kinetics are tightly regulated by the gradual addition of mineral or organic acids, which maintains the reaction medium within a specific acidic window (pH 1-3). This gradient pH control is critical; it prevents the degradation of the nitrite species while ensuring sufficient protonation of the carbonyl oxygen to facilitate nucleophilic attack by the nitrosonium ion. The use of ether solvents plays a dual role, acting both as a reaction medium and as an extractant that allows for the seamless separation of the product in later stages.
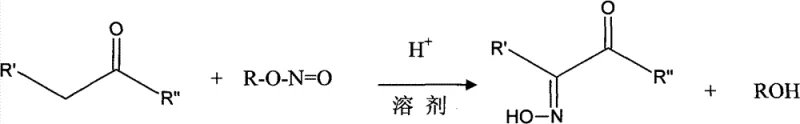
Furthermore, the impurity profile is meticulously managed through the strategic use of alkaline extraction followed by acid neutralization. By extracting the reaction mixture with a sodium hydroxide and sodium bisulfite solution, unreacted ketones and acidic byproducts are effectively separated from the desired oxime, which remains in the aqueous phase upon acidification. This selective partitioning ensures that the final crystallization step yields a product with purity levels often exceeding 99%, as the oily precipitate formed upon acidification is allowed to crystallize slowly at low temperatures, promoting the formation of well-defined lattice structures that exclude impurities.
How to Synthesize Alpha-Oximido Aliphatic Ketone Efficiently
The implementation of this synthesis route requires careful attention to the stoichiometry of reagents and the timing of acid addition to maximize yield and minimize waste. The protocol outlined in the patent provides a robust framework for converting various aliphatic ketones, including those with alpha-reactive hydrogen atoms, into their corresponding oximido derivatives with high efficiency. Detailed standardized synthesis steps see the guide below.
- Generate methyl nitrite gas by reacting sodium nitrite with methanol and dilute sulfuric acid.
- Conduct the nitrosation-oximation reaction by passing methyl nitrite into aliphatic ketone dissolved in ether solvent with controlled acid addition to maintain pH 1-3.
- Extract with alkaline solution, acidify the aqueous layer to precipitate the product, and crystallize via cooling to obtain high-purity white flakes.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from multi-cycle legacy processes to this single-pass methodology offers profound economic and logistical benefits. The elimination of repetitive reaction cycles translates directly into a drastic reduction in utility consumption, labor hours, and equipment occupancy time, thereby lowering the overall cost of goods sold. Moreover, the ability to recycle the organic solvent phase from one batch directly into the next creates a closed-loop system that minimizes raw material expenditure and reduces the environmental footprint associated with solvent disposal.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the collapse of the production timeline; by avoiding the need to repeat the nitrosation and extraction sequence up to nine times, manufacturers can achieve substantial cost savings in terms of energy usage and reactor throughput. The process eliminates the need for expensive vacuum distillation purification steps, relying instead on simple cooling crystallization, which further reduces capital expenditure on specialized equipment and lowers operational complexity. Additionally, the high atom economy of the single-pass reaction ensures that raw materials are utilized more efficiently, reducing the cost per kilogram of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: The simplified workflow inherently reduces the risk of batch failure and variability, leading to more predictable lead times for high-purity pharmaceutical intermediates. Since the process does not rely on complex, multi-stage recycling loops that are prone to accumulation of impurities or mechanical failures, the continuity of supply is significantly strengthened. The use of common, commercially available solvents like isopropyl ether and standard mineral acids ensures that raw material sourcing remains stable and unaffected by niche supply chain disruptions, providing a secure foundation for long-term production planning.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent waste and the ability to recycle the organic phase align perfectly with modern green chemistry principles and regulatory standards. The process generates fewer aqueous waste streams compared to the exhaustive extraction methods of the past, simplifying wastewater treatment requirements and lowering compliance costs. Furthermore, the straightforward nature of the reaction conditions—operating at mild temperatures between 15-35°C and atmospheric pressure—makes the technology highly scalable from pilot plant to commercial production without the need for exotic high-pressure reactors or cryogenic cooling systems.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability, safety, and purity profiles of this synthesis method, derived directly from the experimental data and claims within the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: How does this method improve upon US3,697,597?
A: Unlike the prior art which required repeating the nitrosation and extraction cycle up to 9 times to achieve acceptable yield, this novel method completes the conversion in a single pass through precise pH gradient control and optimized gas ventilation, reducing processing time by over 85%.
Q: What solvents are compatible with this synthesis?
A: The process demonstrates high efficacy using ethers such as isopropyl ether, n-butyl ether, or tetrahydrofuran (THF), with the added advantage that the organic phase can be recycled directly for subsequent batches.
Q: What is the expected purity of the final crystal?
A: By employing direct cooling crystallization of the acidified oily layer followed by cold water washing and vacuum drying, the method consistently yields white flake-like crystals with purity exceeding 99%, eliminating the need for complex distillation purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Oximido Aliphatic Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of patents like CN101693671B are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-oximido aliphatic ketone meets the exacting standards required by global drug developers.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits for your supply chain. Please contact us today to索取 specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-quality chemical solutions.
