Scalable Production of High-Purity Anamorelin Intermediates Using Novel Palladium Catalysis
Introduction to Next-Generation Anamorelin Intermediate Synthesis
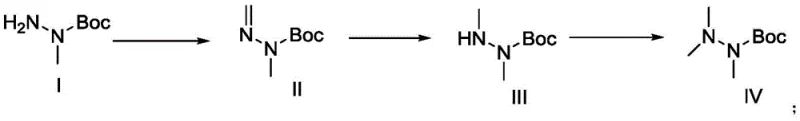
The pharmaceutical industry continuously seeks robust methodologies for constructing complex peptide mimetics, particularly for ghrelin receptor agonists like Anamorelin, which addresses cachexia in non-small cell lung cancer patients. A pivotal breakthrough in this domain is documented in Chinese Patent CN108129357B, which discloses a highly efficient preparation method for the critical hydrazine moiety found within the Anamorelin structure. This patent introduces a transformative one-pot synthetic strategy that converts 1-Boc-1-methylhydrazine into N,N',N'-trimethylhydrazine carboxylic acid tert-butyl ester through alternating reactions with formaldehyde compounds and palladium catalysts. Unlike legacy processes that struggle with hazardous reagents and unstable intermediates, this innovation leverages controlled catalytic hydrogenation to achieve superior yield and purity profiles. The significance of this technology extends beyond mere academic interest; it represents a viable pathway for reliable pharmaceutical intermediates supplier networks aiming to secure the supply chain for oncology therapeutics. By addressing the inherent instability of trimethylhydrazine derivatives, this method ensures that the resulting hydrochloride salt is obtained as a high-purity crystalline product, fundamentally altering the manufacturability of this essential building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N',N'-trimethylhydrazine and its salts has been plagued by severe safety hazards and operational inefficiencies that hinder industrial scalability. Traditional routes, such as those described in earlier literature, often rely on 1,1-dimethylhydrazine as a starting material, a substance known for its extreme toxicity, flammability, and explosive potential, making it notoriously difficult to store and transport safely. Furthermore, alternative pathways utilizing sodium hydride (NaH) for methylation require stringent anhydrous and oxygen-free conditions at low temperatures, introducing significant process risks and requiring specialized equipment to prevent fire hazards. Even when successfully synthesized, the resulting trimethylhydrazine products from these conventional methods typically exist as oils or solutions that cannot be stored for extended periods, necessitating immediate use or complex distillation procedures. The final salt formation steps in these older protocols often yield hygroscopic oily substances rather than stable solids, complicating purification and rendering the materials unsuitable for large-scale industrial production due to consistency and stability issues.
The Novel Approach
In stark contrast to these hazardous precedents, the novel approach detailed in the patent utilizes 1-Boc-1-methylhydrazine as a safe and stable precursor, effectively bypassing the need for handling volatile and toxic dimethylhydrazine species. This methodology employs a sophisticated one-pot technique where formaldehyde compounds and a palladium catalyst work in tandem with a hydrogen source to sequentially methylate the hydrazine nitrogen atoms without isolating reactive intermediates. The process operates under moderate conditions, utilizing common solvents like ethanol and standard hydrogenation pressures, which drastically simplifies the operational complexity compared to cryogenic or strictly anhydrous requirements of the past. Crucially, this route culminates in the formation of N,N,N'-trimethylhydrazine hydrochloride as a distinct crystalline solid with low hygroscopicity, a physical form that is exceptionally convenient for preservation and long-term storage. This shift from unstable oils to stable crystals not only enhances product quality but also streamlines the downstream processing required for cost reduction in API manufacturing, making it an ideal candidate for commercial adoption.
Mechanistic Insights into Palladium-Catalyzed Reductive Methylation
The core chemical innovation driving this process is a sequential reductive amination mechanism facilitated by palladium catalysis, which allows for the precise installation of methyl groups onto the hydrazine scaffold. The reaction initiates with the condensation of the starting hydrazine with a formaldehyde source, such as paraformaldehyde, to generate an imine intermediate in situ. This imine is subsequently subjected to catalytic hydrogenation using a palladium catalyst, such as Pd/C or Pd(OH)2, under a hydrogen atmosphere typically maintained around 9.8 atm. The catalyst activates the hydrogen gas, enabling the reduction of the carbon-nitrogen double bond to a single bond, thereby installing the first methyl group efficiently. This cycle of condensation and reduction is repeated within the same reaction vessel to install the second methyl group, demonstrating the robustness of the catalytic system in tolerating the evolving chemical environment. The use of ethanol as a preferred solvent further supports this mechanism by providing excellent solubility for both the organic substrates and the gaseous hydrogen, ensuring homogeneous reaction kinetics throughout the transformation.
Controlling impurities in hydrazine chemistry is notoriously challenging due to the propensity for over-alkylation or decomposition, yet this mechanistic pathway offers inherent selectivity advantages. The Boc protecting group on the starting material plays a critical role in directing the methylation to the desired nitrogen atoms while preventing unwanted side reactions at the protected site. Furthermore, the final step involves reacting the fully methylated Boc-protected intermediate with hydrochloric acid in an organic solvent like ethyl acetate at controlled low temperatures, typically around 0°C. This acidolysis step simultaneously removes the Boc group and forms the hydrochloride salt, inducing crystallization directly from the reaction mixture. The ability to precipitate the product as a high-purity crystal without the need for column chromatography or extensive distillation indicates that the mechanistic pathway effectively suppresses the formation of closely related structural impurities, ensuring the final material meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize N,N',N'-Trimethylhydrazine Hydrochloride Efficiently
The practical execution of this synthesis involves a streamlined sequence that merges multiple chemical transformations into a unified operational flow, significantly reducing the footprint of the manufacturing process. Operators begin by reacting 1-Boc-1-methylhydrazine with paraformaldehyde in ethanol under reflux to establish the initial imine equilibrium, followed by the introduction of the palladium catalyst and hydrogen gas to drive the reduction. Once the first methylation is complete, additional formaldehyde is introduced to the same pot to repeat the cycle, achieving the fully methylated state without intermediate workups. The detailed standardized synthetic steps, including exact molar ratios, temperature profiles, and pressure settings required to replicate this high-yield process, are outlined below for technical reference.
- React 1-Boc-1-methylhydrazine with paraformaldehyde in ethanol at 80°C to form the imine intermediate.
- Perform catalytic hydrogenation using Pd/C at 60°C and 9.8 atm pressure to reduce the imine to the mono-methylated species.
- Add additional paraformaldehyde and repeat the hydrogenation cycle to achieve the fully methylated Boc-protected hydrazine, followed by HCl salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of highly hazardous raw materials like 1,1-dimethylhydrazine and sodium hydride fundamentally de-risks the supply chain, reducing the regulatory burden and insurance costs associated with handling dangerous goods. Moreover, the transition from producing unstable oily intermediates to stable crystalline salts dramatically enhances inventory management capabilities, allowing for longer shelf life and more flexible logistics planning without the fear of product degradation during transit. This stability is crucial for maintaining continuous supply lines to downstream API manufacturers, ensuring that production schedules are not disrupted by the need for immediate processing of perishable intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive and labor-intensive purification steps such as column chromatography which are mandatory in older synthetic routes. By achieving high purity through direct crystallization, the method significantly reduces solvent consumption and waste disposal costs, leading to substantial cost savings in the overall production budget. Additionally, the one-pot nature of the reaction minimizes unit operations, reducing energy consumption and labor hours required for multiple isolation and drying steps, thereby optimizing the cost structure for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The use of commercially available and safe starting materials ensures that the supply chain is resilient against disruptions caused by the scarcity of specialized or regulated reagents. Since the process does not rely on exotic catalysts or hard-to-source precursors, sourcing becomes straightforward, and lead times for raw material acquisition are significantly shortened. This reliability is further bolstered by the robustness of the reaction conditions, which tolerate standard industrial equipment rather than requiring bespoke reactors designed for extreme safety containment, facilitating easier technology transfer between manufacturing sites.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process aligns perfectly with green chemistry principles by reducing the E-factor through minimized solvent use and waste generation. The absence of heavy metal contaminants often associated with other catalytic systems simplifies the purification of the final API, easing the burden on quality control laboratories. The scalability is proven by the use of standard hydrogenation equipment and common solvents, making the commercial scale-up of complex hydrazine derivatives feasible from pilot plant to multi-ton production without encountering the thermal runaway risks associated with traditional hydrazine alkylations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative data. These insights are intended to clarify the operational benefits and technical feasibility for potential partners evaluating this route for their supply chain integration. Understanding these nuances is essential for making informed decisions about adopting this advanced manufacturing protocol.
Q: Why is the new crystalline salt form superior to previous oily products?
A: The novel process yields N,N,N'-trimethylhydrazine hydrochloride as a stable crystalline solid with low hygroscopicity, eliminating the storage instability and purification difficulties associated with the oily products generated by conventional methods.
Q: What safety advantages does this route offer over traditional hydrazine synthesis?
A: This method avoids the use of highly toxic and explosive 1,1-dimethylhydrazine and dangerous reagents like sodium hydride (NaH), utilizing safer starting materials and standard catalytic hydrogenation conditions instead.
Q: How does the one-pot strategy impact production efficiency?
A: By combining condensation and reduction steps in a single vessel without isolating intermediates, the process significantly reduces solvent consumption, operational time, and waste generation compared to multi-step isolation protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anamorelin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key intermediates for the global oncology market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our facilities are equipped with rigorous QC labs and advanced analytical capabilities to guarantee stringent purity specifications for every batch of Anamorelin intermediate we produce, adhering to the highest international standards for pharmaceutical ingredients.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this patented technology for their upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bill of materials. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, quality, and economic efficiency.
