Advanced Metal-Free Synthesis of Substituted Acrylates for Commercial Pharmaceutical Manufacturing
Advanced Metal-Free Synthesis of Substituted Acrylates for Commercial Pharmaceutical Manufacturing
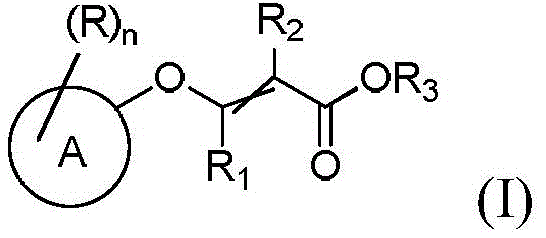
The pharmaceutical industry constantly seeks robust, scalable, and economically viable synthetic routes for critical intermediates. Patent CN114085146A, published in early 2022, introduces a groundbreaking methodology for the preparation of substituted acrylate compounds of general formula (I). These compounds serve as indispensable building blocks in the synthesis of complex active pharmaceutical ingredients (APIs). The disclosed technology represents a significant leap forward by addressing longstanding challenges associated with traditional synthesis methods, specifically the reliance on transition metal catalysts and harsh reaction conditions. By leveraging a novel nucleophilic substitution strategy using activated sulfonate or phosphonate intermediates, this invention enables the production of high-purity substituted acrylate compounds under mild, metal-free conditions. This advancement is particularly relevant for manufacturers aiming to streamline their supply chains and reduce the environmental footprint of their chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
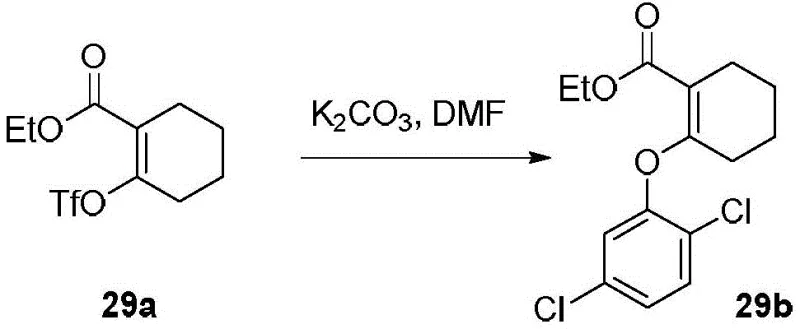
Prior to this innovation, the synthesis of substituted acrylates often relied on methodologies that presented significant operational and economic hurdles for large-scale manufacturing. A notable example found in the background art, specifically WO2013096771a1, describes a reaction involving the coupling of a triflate-substituted cyclohexene derivative with a dichlorophenol. While functional, this prior art method suffers from several critical deficiencies that impact commercial viability. Firstly, the reaction requires extremely high temperatures, typically around 120°C, which increases energy consumption and poses safety risks related to thermal runaway or solvent degradation. Secondly, the reported yields are suboptimal, often hovering around 59%, which leads to substantial material loss and increased cost of goods sold (COGS). Furthermore, the use of triflate (OTf) leaving groups, while reactive, can be expensive and generate corrosive byproducts that complicate waste management. The reliance on such harsh conditions and costly reagents makes the conventional approach less attractive for the cost-sensitive pharmaceutical market.
The Novel Approach
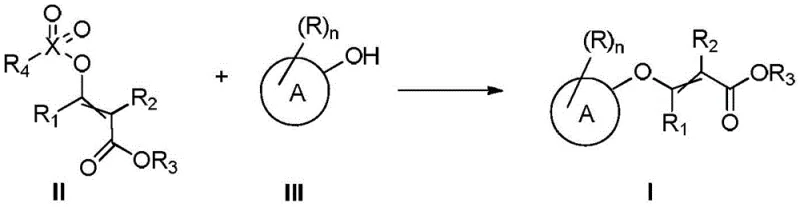
In stark contrast to the limitations of the prior art, the method disclosed in CN114085146A offers a refined and efficient pathway to the target molecules. The core of this innovation lies in the utilization of a compound of formula (II), which features a sulfonate or phosphonate leaving group, reacting with a hydroxyl-containing compound of formula (III) in the presence of a base. This strategic shift allows the reaction to proceed at significantly milder temperatures, typically ranging from 50°C to 80°C, thereby enhancing process safety and energy efficiency. Moreover, the invention explicitly avoids the use of metal catalysts, which is a major advantage for pharmaceutical applications where residual metal levels are strictly regulated. By eliminating the need for extensive purification steps to remove trace metals, manufacturers can achieve substantial cost reduction in API manufacturing. The versatility of this approach is further highlighted by its compatibility with a wide range of solvents, including green solvents like isopropyl acetate, facilitating easier downstream processing and solvent recovery.
Mechanistic Insights into Base-Promoted Nucleophilic Substitution
The chemical elegance of this process is rooted in a base-promoted nucleophilic substitution mechanism that ensures high selectivity and yield. The reaction initiates with the activation of the phenolic or heteroaryl hydroxyl group in compound (III) by the base, generating a highly nucleophilic phenoxide or alkoxide species. This nucleophile then attacks the electron-deficient carbon center of the activated intermediate (Formula II), displacing the sulfonate or phosphonate leaving group. The choice of the leaving group in Formula II is critical; unlike the unstable or overly reactive triflates used in older methods, the sulfonate and phosphonate groups provide an optimal balance of stability and reactivity. This balance prevents premature decomposition of the starting material while ensuring rapid conversion to the desired product under mild thermal conditions. The absence of transition metals means the reaction proceeds through a purely ionic mechanism, avoiding radical pathways that often lead to complex impurity profiles and side reactions.
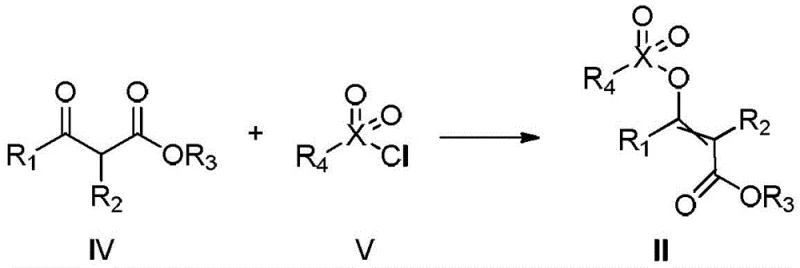
Furthermore, the preparation of the key intermediate (Formula II) itself is optimized for industrial feasibility. As illustrated in the patent data, the intermediate can be synthesized by reacting a beta-keto ester (Formula IV) with a sulfonyl or phosphoryl chloride (Formula V). This precursor step is highly efficient and utilizes readily available raw materials. The ability to generate the activated intermediate in situ or isolate it with high purity provides flexibility in process design. For instance, the use of p-toluenesulfonyl chloride allows for the formation of a stable tosylate intermediate that can be handled safely before the subsequent coupling step. This two-step sequence ensures that the final coupling reaction is clean and predictable, minimizing the formation of regioisomers or elimination byproducts that often plague acrylate syntheses. The result is a robust process capable of delivering high-purity substituted acrylate compounds suitable for sensitive downstream transformations.
How to Synthesize Substituted Acrylate Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The process generally involves a two-stage sequence: first, the activation of the beta-keto ester to form the sulfonate or phosphonate intermediate, and second, the nucleophilic displacement with the chosen phenol or alcohol. The patent provides detailed guidance on stoichiometry, suggesting a molar ratio of the intermediate to the nucleophile of approximately 1:1 to 1:1.2, which helps drive the reaction to completion without excessive waste. The choice of base is also pivotal, with inorganic bases like potassium carbonate or organic amines like DABCO proving effective depending on the specific substrate solubility and reactivity profile. Solvent selection plays a crucial role in heat transfer and product isolation, with esters like isopropyl acetate offering a favorable balance of polarity and boiling point. For a comprehensive understanding of the specific operational steps and safety considerations, please refer to the standardized synthesis guide below.
- Prepare the activated intermediate (Formula II) by reacting a beta-keto ester with a sulfonyl or phosphoryl chloride in the presence of a base.
- React the intermediate (Formula II) with a phenol or heteroaryl alcohol (Formula III) using an inorganic or organic base in a suitable solvent like isopropyl acetate.
- Maintain reaction temperature between 50-80°C, monitor completion, and purify the final substituted acrylate product via standard workup procedures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating transition metal catalysts, the process removes the need for expensive scavenger resins or complex filtration steps required to meet strict residual metal specifications (e.g., ICH Q3D guidelines). This reduction in downstream processing directly correlates to lower operational expenditures and shorter batch cycle times. Additionally, the use of mild reaction temperatures reduces the energy load on manufacturing facilities, contributing to lower utility costs and a smaller carbon footprint. The robustness of the reaction conditions also implies a wider operating window, reducing the risk of batch failures due to minor temperature fluctuations, thereby enhancing overall supply reliability.
- Cost Reduction in Manufacturing: The economic impact of this metal-free process is profound. Traditional methods often require precious metal catalysts such as palladium or copper, which not only carry high upfront costs but also necessitate rigorous recovery systems to prevent loss. By replacing these with inexpensive inorganic bases and organic amines, the raw material cost profile is significantly improved. Furthermore, the avoidance of triflate leaving groups, which are notoriously expensive and moisture-sensitive, in favor of stable sulfonates or phosphonates, reduces the cost of goods. The high yields reported in the examples minimize the amount of starting material required per kilogram of final product, optimizing material efficiency. These factors combined create a compelling economic case for switching to this newer methodology, allowing for more competitive pricing in the final API market.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. The reagents utilized in this process, such as beta-keto esters, sulfonyl chlorides, and common phenols, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages that can occur with specialized catalysts or exotic reagents. Moreover, the stability of the intermediates allows for potential stocking strategies, where the activated intermediate can be prepared in bulk and stored for later use, decoupling the two reaction steps and providing greater flexibility in production scheduling. This flexibility is crucial for responding to sudden spikes in demand or unexpected disruptions in the supply of specific starting materials, ensuring continuous production flow.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the ton-scale often reveals hidden hazards, but this methodology is inherently designed for safety. The exothermic nature of the reaction is manageable under the recommended conditions (50-80°C), reducing the need for complex cooling systems or specialized pressure vessels. From an environmental perspective, the ability to use greener solvents like isopropyl acetate instead of chlorinated solvents or high-boiling amides like DMF simplifies waste treatment and solvent recovery. The absence of heavy metals in the waste stream significantly lowers the cost and complexity of effluent treatment, ensuring compliance with increasingly stringent environmental regulations. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and embodiments described in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers cover aspects ranging from substrate scope to purification strategies, ensuring a holistic view of the technology's potential.
Q: What are the key advantages of this new synthesis method over prior art?
A: The novel method eliminates the need for expensive and difficult-to-remove metal catalysts, operates at significantly milder temperatures (50-80°C vs 120°C), and utilizes safer, non-triflate leaving groups to achieve higher yields and purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scale-up, utilizing commercially available raw materials, common solvents like isopropyl acetate, and avoiding hazardous reagents, which simplifies safety management and waste treatment.
Q: What types of substituents are compatible with this reaction?
A: The method demonstrates broad substrate scope, accommodating various aryl and heteroaryl rings, different alkyl and haloalkyl groups at the alpha and beta positions, and diverse ester functionalities, making it versatile for generating compound libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Acrylate Compounds Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with an experienced CDMO is essential for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from development to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of this metal-free synthesis, including precise temperature control and advanced solvent recovery systems. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of substituted acrylate compounds meets the highest international standards, providing you with the confidence needed for regulatory filings.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By collaborating with us, you gain access to cutting-edge chemical technologies and a reliable partner dedicated to supporting your long-term growth in the pharmaceutical market.
