Advanced Recovery of 3-Mercapto-5-Methyl-1,2,4-Triazole for Sustainable API Manufacturing
The pharmaceutical industry is constantly seeking innovative solutions to optimize the production of critical antibiotic intermediates, particularly for blockbuster drugs like Ceftriaxone sodium. A groundbreaking approach detailed in patent CN111057017B introduces a highly efficient method for recovering 3-mercapto-5-methyl-1,2,4-triazole directly from triazine ring cyclization mother liquor. This technology addresses a significant pain point in the supply chain by transforming what was previously considered hazardous waste into a high-value pharmaceutical intermediate. By leveraging a specialized catalytic system involving stearates, the process achieves exceptional purity levels while drastically minimizing environmental impact. For global procurement teams and R&D directors, this represents a pivotal shift towards circular economy principles within fine chemical manufacturing, ensuring a more sustainable and cost-effective source of essential heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
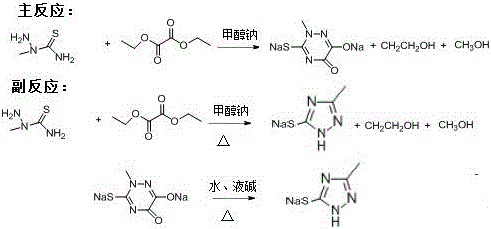
Traditionally, the production of triazine rings, which are vital precursors for Ceftriaxone sodium, generates substantial quantities of cyclization mother liquor that are often treated merely as waste streams. In conventional processing facilities, this mother liquor, rich in solvents like methanol and ethanol alongside organic byproducts, typically undergoes basic distillation or incineration, leading to significant economic loss and environmental burden. The primary drawback of these legacy methods is the failure to isolate and purify the valuable 3-mercapto-5-methyl-1,2,4-triazole contained within the mixture, resulting in a linear consumption of raw materials without resource recovery. Furthermore, the disposal of such chemically complex waste requires rigorous environmental compliance measures, driving up operational expenditures and complicating the supply chain logistics for large-scale API manufacturers. Without a targeted recovery strategy, the potential value embedded in these byproducts remains locked, contributing to higher overall production costs for the final antibiotic.
The Novel Approach
The innovative methodology outlined in the patent revolutionizes this workflow by introducing a targeted recovery sequence that utilizes stearate catalysts to selectively isolate the desired triazole derivative. Instead of discarding the mother liquor, the process begins with the addition of calcium or sodium stearate, followed by reduced pressure distillation to remove volatile solvents, effectively concentrating the target compounds. A crucial step involves adjusting the pH of the solution to a neutral or slightly alkaline range (pH 7-9) using liquid alkali, which prepares the mixture for a controlled reflux reaction. This precise manipulation of reaction conditions allows for the separation of the triazole species from the complex matrix, enabling its extraction and subsequent purification to pharmaceutical-grade standards. This approach not only recovers a valuable intermediate but also streamlines the waste treatment process, creating a dual benefit of revenue generation and cost avoidance.
Mechanistic Insights into Stearate-Catalyzed Recovery and Purification
The core of this technological advancement lies in the specific interaction between the stearate catalyst and the components of the cyclization mother liquor under thermal stress. The stearate acts as a phase-transfer and stabilizing agent that facilitates the rearrangement and isolation of the 3-mercapto-5-methyl-1,2,4-triazole during the reflux stage at temperatures between 90°C and 110°C. By maintaining the pH within the narrow window of 7 to 9, the process ensures that the triazole remains in a stable ionic form suitable for subsequent extraction, preventing degradation or polymerization that often plagues harsh acidic or basic treatments. The mechanism effectively separates the target molecule from the triazine ring sodium salt and other organic impurities, leveraging differences in solubility and reactivity that are enhanced by the presence of the long-chain fatty acid salts. This selective catalysis is key to achieving the reported high purity without the need for complex chromatographic separations.
Furthermore, the purification mechanism relies on a robust solid-liquid separation followed by solvent extraction using petroleum ether, which selectively dissolves the organic triazole product while leaving inorganic salts behind. The final drying step at 100°C ensures the removal of residual moisture and solvents, yielding a free-flowing powder with consistent quality. This mechanistic pathway is designed to be robust against variations in the initial composition of the mother liquor, making it highly suitable for industrial application where feedstock consistency can vary. The ability to consistently achieve purity levels greater than 99.4% demonstrates the efficacy of the stearate-mediated pathway in controlling impurity profiles, a critical factor for downstream API synthesis where trace contaminants can affect drug safety and efficacy.
How to Synthesize 3-Mercapto-5-Methyl-1,2,4-Triazole Efficiently
Implementing this recovery process requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The procedure involves a sequential operation of distillation, pH adjustment, reflux, and extraction, each step critical to the success of the isolation. Operators must carefully monitor the vacuum degree during the initial solvent removal and maintain precise temperature controls during the reflux phase to prevent side reactions.
- Add a stearate catalyst (calcium or sodium stearate) to the triazine ring cyclization mother liquor and perform reduced pressure distillation to remove solvents.
- Adjust the pH of the residual solution to between 7 and 9 using 30% liquid alkali, followed by refluxing at 90-110°C for 2-5 hours.
- Perform solid-liquid separation, extract the liquid phase with petroleum ether, distill off the solvent, and dry the resulting product at 100°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this recovery technology offers profound strategic advantages beyond simple chemical synthesis. By converting a waste stream into a saleable or usable intermediate, manufacturers can significantly reduce the net cost of producing triazine rings, effectively subsidizing the primary production line with revenue from the byproduct. This internal sourcing of 3-mercapto-5-methyl-1,2,4-triazole reduces dependency on external suppliers for this specific building block, thereby enhancing supply chain resilience and mitigating the risk of market volatility. The elimination of expensive waste disposal fees associated with hazardous mother liquor further contributes to substantial cost savings, improving the overall margin profile of the antibiotic manufacturing process.
- Cost Reduction in Manufacturing: The implementation of this stearate-catalyzed recovery route eliminates the need for purchasing fresh 3-mercapto-5-methyl-1,2,4-triazole for certain applications, directly lowering raw material expenditure. By avoiding the capital and operational costs associated with traditional waste incineration or complex chemical treatment, facilities can realize significant operational efficiency gains. The process utilizes inexpensive and readily available catalysts like sodium stearate, ensuring that the cost of goods sold remains low while maximizing the value extracted from every batch of mother liquor.
- Enhanced Supply Chain Reliability: Integrating this recovery step creates a closed-loop system that buffers the supply chain against external disruptions in the availability of triazole intermediates. Since the feedstock is generated internally as a byproduct of the main triazine ring production, the continuity of supply is inherently linked to the primary manufacturing schedule, reducing lead times and logistical complexities. This self-sufficiency allows for better inventory management and ensures that production targets for Ceftriaxone sodium are met without interruption due to raw material shortages.
- Scalability and Environmental Compliance: The process is designed for easy scale-up, utilizing standard unit operations such as distillation, filtration, and reflux that are common in existing fine chemical plants. From an environmental perspective, the reduction in waste volume simplifies compliance with increasingly stringent global regulations on industrial effluent and hazardous waste. This eco-friendly profile not only reduces environmental processing costs but also enhances the corporate sustainability credentials of the manufacturer, appealing to environmentally conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recovery technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation to assist decision-makers in evaluating feasibility.
Q: What is the primary advantage of using stearate catalysts in this recovery process?
A: The use of calcium or sodium stearate facilitates the efficient separation and purification of 3-mercapto-5-methyl-1,2,4-triazole from complex mother liquor matrices, achieving purity levels exceeding 99.4% without requiring expensive transition metal catalysts.
Q: How does this method impact the environmental footprint of Ceftriaxone production?
A: By recovering valuable byproducts from the cyclization mother liquor, this method significantly reduces the volume of hazardous waste requiring disposal and lowers the overall environmental protection processing costs associated with antibiotic intermediate manufacturing.
Q: What are the typical purity and recovery rates achieved with this technique?
A: According to patent data, the process consistently yields 3-mercapto-5-methyl-1,2,4-triazole with a purity of ≥99.4% and a recovery rate ranging from 1.0% to 1.5% relative to the total weight of the mother liquor processed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Mercapto-5-Methyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate recovery in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex recovery processes like the one described in CN111057017B are executed with precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of recovered triazole meets the highest standards required for API synthesis. We are committed to helping our partners optimize their production economics through advanced chemical engineering and process innovation.
We invite you to discuss how our expertise can be leveraged to implement this cost-effective recovery strategy in your own supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible value of partnering with us for your high-purity pharmaceutical intermediate needs.
