Scalable Visible Light Synthesis of C2 Substituted Benzothiazole Hydroxyalkylated Derivatives for Commercial Production
The pharmaceutical and agrochemical industries are constantly seeking robust, sustainable, and cost-effective methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN111961011B, which discloses a novel preparation method for C2 substituted 2H-benzothiazole hydroxyalkylated derivatives. This technology leverages visible light induction coupled with inexpensive organic dye photosensitizers to drive the hydroxyalkylation reaction under remarkably mild conditions. Unlike traditional methods that rely on harsh reagents or expensive transition metals, this approach utilizes simple LED white light irradiation at room temperature, representing a paradigm shift towards greener chemical manufacturing. For R&D directors and procurement managers alike, this patent offers a compelling route to high-purity intermediates with a drastically simplified operational profile, addressing critical pain points regarding safety, cost, and environmental compliance in modern fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the C2 position on the benzothiazole ring has been fraught with significant technical and economic challenges. Conventional synthetic routes often necessitate the use of strong bases for deprotonation followed by reaction with aldehydes or ketones, or alternatively, the employment of highly reactive organometallic reagents such as n-BuLi, t-BuLi, or Grignard reagents. These methodologies impose severe constraints on the manufacturing process, including the absolute requirement for cryogenic conditions to manage exothermic risks and prevent reagent decomposition. Furthermore, alternative oxidative approaches utilizing tert-butyl hydroperoxide demand elevated temperatures around 120°C, introducing substantial safety hazards related to the handling of explosive organic peroxides. The reliance on transition metal catalysts, such as expensive Iridium complexes, further exacerbates the cost structure and introduces the persistent issue of heavy metal residue removal, which is a critical bottleneck for pharmaceutical grade intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN111961011B introduces a transformative visible light-promoted strategy that operates under ambient thermal conditions. By employing cheap and easily obtained organic dyes like Eosin B or Rose Bengal as photocatalysts, the process eliminates the need for precious metals entirely. The reaction proceeds efficiently at room temperature in common solvents like acetonitrile, driven solely by the energy from a standard LED white light lamp. This not only drastically reduces energy consumption associated with heating or cooling but also enhances the safety profile by removing hazardous oxidants and pyrophoric reagents from the workflow. The simplicity of the catalytic system allows for a streamlined workup procedure, typically involving solvent removal and standard column chromatography, thereby facilitating a more agile and cost-effective production cycle for high-value benzothiazole derivatives.
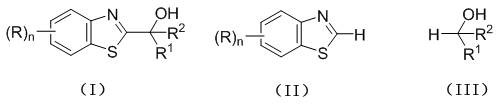
Mechanistic Insights into Visible Light Photocatalytic Hydroxyalkylation
The core of this innovation lies in the efficient generation of carbon-centered radicals via a photoredox cycle initiated by organic dye excitation. Upon irradiation with visible light, the organic dye photosensitizer absorbs photons to reach an excited state, which then facilitates a single electron transfer (SET) process with the fatty alcohol substrate. This interaction generates an alpha-hydroxy alkyl radical species from the alcohol, which is sufficiently nucleophilic to attack the electron-deficient C2 position of the benzothiazole ring. The subsequent rearomatization or proton transfer steps lead to the formation of the desired C2 hydroxyalkylated product while regenerating the ground state catalyst to continue the cycle. This mechanistic pathway bypasses the high energy barriers associated with thermal activation, allowing the reaction to proceed smoothly at room temperature without the need for aggressive chemical activators that often lead to complex impurity profiles.
From an impurity control perspective, the mildness of this photochemical protocol offers distinct advantages over thermal or strongly basic conditions. Traditional methods involving strong bases or organolithiums often suffer from poor chemoselectivity, leading to over-alkylation, ring opening, or polymerization side reactions that are difficult to separate. In contrast, the radical-mediated mechanism under visible light irradiation exhibits high selectivity for the C2 position, minimizing the formation of structural isomers or degradation byproducts. The use of stoichiometric excesses of fatty alcohol, as suggested in the patent, further drives the reaction to completion while acting as a benign solvent medium, ensuring that the final crude product contains fewer difficult-to-remove impurities. This results in a cleaner reaction profile that simplifies downstream purification, ultimately yielding a high-purity intermediate suitable for sensitive pharmaceutical applications without extensive recrystallization steps.
How to Synthesize C2 Substituted Benzothiazole Hydroxyalkylated Derivatives Efficiently
Implementing this visible light synthesis requires careful attention to the ratio of reactants and the intensity of the light source to maximize yield and reproducibility. The patent outlines a straightforward protocol where substituted 2H-benzothiazole is mixed with a significant excess of fatty alcohol, typically in a molar ratio ranging from 1:5.0 to 1:50.0, to ensure complete conversion of the heterocycle. A catalytic amount of organic dye, such as 1.0 to 10.0 mol% of Eosin B, is introduced into the reaction vessel containing a solvent like acetonitrile. The mixture is then subjected to continuous stirring under nitrogen protection while being irradiated by a 10-50W LED white light lamp. Reaction progress is monitored via TLC, with typical completion times ranging from 8 to 38 hours depending on the specific substrate electronics. Detailed standardized synthesis steps follow below.
- Mix substituted 2H-benzothiazole with fatty alcohol and an organic dye photosensitizer (e.g., Eosin B) in a solvent like acetonitrile under nitrogen protection.
- Irradiate the reaction mixture with an LED white light lamp (10-50W) at room temperature while stirring for 8 to 38 hours until TLC indicates completion.
- Concentrate the reaction solution to remove the solvent and purify the crude product via column chromatography using a petroleum ether and ethyl acetate mixed solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional metallation or high-temperature oxidation to this visible light protocol presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The elimination of expensive transition metal catalysts like Iridium removes a major cost driver and supply chain vulnerability, as organic dyes are commodity chemicals with stable pricing and abundant availability. Furthermore, the removal of hazardous reagents such as organolithiums and organic peroxides significantly lowers the regulatory burden and insurance costs associated with storage and transport, while also reducing the complexity of waste disposal. The ability to run reactions at room temperature translates to substantial energy savings by eliminating the need for specialized cryogenic cooling equipment or high-temperature heating mantles, making the process inherently more scalable and safer for large-scale manufacturing facilities.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive organic dyes results in a drastic reduction in raw material costs per kilogram of product. Additionally, the simplified reaction setup reduces the capital expenditure required for specialized reactor hardware capable of handling extreme temperatures or pressures. The mild conditions also extend the lifespan of equipment by reducing corrosion and thermal stress, leading to lower maintenance overheads and a more favorable total cost of ownership for the production line.
- Enhanced Supply Chain Reliability: By relying on widely available organic dyes and common fatty alcohols rather than specialized organometallic reagents, the supply chain becomes far more robust against market fluctuations and geopolitical disruptions. The stability of the reagents allows for longer shelf life and easier logistics, reducing the risk of production delays caused by expired or degraded materials. This reliability ensures consistent batch-to-batch quality and uninterrupted supply continuity for downstream pharmaceutical customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The absence of heavy metals and hazardous oxidants simplifies the environmental compliance landscape, making it easier to obtain necessary permits for expansion. The process generates less hazardous waste, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The straightforward workup procedure involving simple solvent evaporation and chromatography is easily adaptable to continuous flow chemistry or larger batch reactors, facilitating a seamless transition from laboratory discovery to multi-ton commercial production without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalysis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this method for industrial applications. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing workflows.
Q: What are the advantages of using organic dye photocatalysts over transition metals for benzothiazole synthesis?
A: Organic dyes like Eosin B and Rose Bengal are significantly cheaper and more readily available than transition metal complexes like Iridium. Furthermore, they eliminate the risk of toxic metal residue in the final pharmaceutical intermediate, simplifying purification and regulatory compliance.
Q: Can this visible light method be scaled for industrial production?
A: Yes, the process operates at room temperature and atmospheric pressure using standard LED irradiation, which avoids the safety hazards associated with high-temperature peroxide oxidants or cryogenic organolithium reagents, making it highly suitable for commercial scale-up.
Q: What is the substrate scope for this hydroxyalkylation reaction?
A: The method demonstrates broad substrate tolerance, accommodating various substituents on the benzothiazole ring (such as methoxy, cyano, chloro, and nitro groups) and different fatty alcohols, yielding products with good efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiazole Hydroxyalkylated Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in modernizing the synthesis of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111961011B are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzothiazole derivative we deliver meets the highest standards of quality and consistency required by global regulatory bodies.
We invite forward-thinking partners to collaborate with us to leverage this cost-effective and sustainable technology for their supply chains. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this metal-free route can optimize your COGS. Please contact our technical procurement team today to request specific COA data for our benzothiazole portfolio and discuss route feasibility assessments for your next project, ensuring a secure and efficient supply of high-quality intermediates for your critical drug development programs.
