Advanced Dithiophene Pyrrole Bridge Indoline Dyes for High-Efficiency Solar Applications
Advanced Dithiophene Pyrrole Bridge Indoline Dyes for High-Efficiency Solar Applications
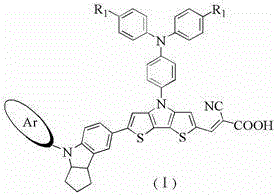
The global transition towards sustainable energy solutions has intensified the demand for high-performance photovoltaic materials, specifically within the realm of dye-sensitized solar cells (DSSC). Patent CN103554958A introduces a groundbreaking class of dithiophene pyrrole bridge-indoline organic dyes that address critical limitations found in traditional ruthenium-based sensitizers. These novel compounds feature a sophisticated molecular architecture where a dithiophene pyrrole bridge connects an electron-donating indoline unit to an electron-withdrawing cyanoacrylic acid acceptor. By incorporating bulky substituents such as fluorenyl or trimeric indenyl groups into the indoline moiety, the invention achieves a remarkable balance between light harvesting capability and charge recombination suppression. This technological leap represents a significant opportunity for manufacturers seeking reliable organic dye photosensitizers supplier partnerships to enhance the efficiency and cost-effectiveness of next-generation solar modules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the benchmark for high-efficiency DSSCs has been set by ruthenium polypyridyl complexes, which, despite their respectable performance, suffer from inherent economic and physical drawbacks that hinder widespread commercial adoption. The primary constraint is the reliance on noble metal ruthenium, which drives up raw material costs and introduces supply chain volatility associated with precious metal mining and refining. Furthermore, these metal-organic complexes often exhibit relatively low molar absorptivity coefficients compared to purely organic alternatives, necessitating thicker semiconductor films to capture sufficient light, which can impede electron transport. The separation and purification of ruthenium complexes are also notoriously difficult due to the presence of multiple isomers and the stability of the metal-ligand bonds, leading to complex downstream processing that increases the overall cost reduction in electronic chemical manufacturing efforts.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these issues by utilizing a metal-free organic framework built upon a dithiophene pyrrole bridge system. This design significantly enhances the molar absorptivity of the dye molecules, allowing for more efficient light harvesting even with thinner semiconductor layers. The strategic incorporation of large steric hindrance groups, specifically the fluorenyl or trimeric indenyl structures attached to the indoline nitrogen, serves a dual purpose: it acts as an insulating barrier to prevent the approach of electrolyte ions to the semiconductor surface, thereby inhibiting charge recombination. This structural modification directly translates to an increase in open-circuit voltage, a critical parameter for boosting the overall photoelectric conversion efficiency of the solar cell beyond the 8% threshold demonstrated in the embodiments.
Mechanistic Insights into Suzuki Coupling and Knoevenagel Condensation
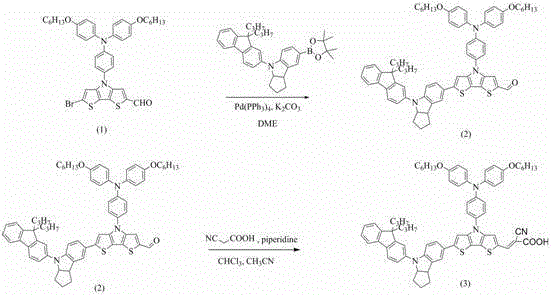
The synthesis of these advanced photosensitizers relies on a robust two-step sequence beginning with a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. In this critical step, an alkoxytriphenylamine-substituted dithiophene pyrrole bromoaldehyde reacts with a specialized indoline boronate ester in the presence of tetrakis(triphenylphosphine)palladium(0) and potassium carbonate. The reaction proceeds in glycol dimethyl ether (DME) under reflux conditions for approximately 8 hours, facilitating the formation of a carbon-carbon bond between the electron-rich indoline donor and the conjugated bridge. This step is pivotal for establishing the extended pi-conjugation system required for effective intramolecular charge transfer, and the use of mild basic conditions ensures the integrity of the sensitive aldehyde functionality needed for the subsequent step.
Following the coupling reaction, the intermediate aldehyde undergoes a Knoevenagel condensation with cyanoacetic acid to install the electron-withdrawing anchor group. This transformation is catalyzed by piperidine in a mixed solvent system of acetonitrile and chloroform, typically requiring a two-stage addition of reagents over a total reflux period of 16 hours to drive the equilibrium towards the product. The resulting cyanoacrylic acid moiety is essential for anchoring the dye onto the titanium dioxide surface of the solar cell, ensuring efficient electron injection from the excited state of the dye into the conduction band of the semiconductor. The high yields reported, reaching up to 95% in the second step for the trimeric indenyl derivative, underscore the reliability and reproducibility of this synthetic pathway for producing high-purity OLED material precursors and related optoelectronic compounds.
How to Synthesize Dithiophene Pyrrole Bridge Indoline Dyes Efficiently
The preparation method described offers a streamlined pathway for generating these high-value intermediates, leveraging standard laboratory equipment that translates seamlessly to pilot and production scales. The process begins with the rigorous exclusion of oxygen via nitrogen protection to maintain the activity of the palladium catalyst during the coupling phase. Following the reaction, the crude product is isolated through aqueous workup and extraction with ethyl acetate, followed by purification via column chromatography using a petroleum ether and ethyl acetate gradient. The final condensation step similarly utilizes standard extraction and chromatographic techniques, ensuring that the final dye meets the stringent purity specifications required for photovoltaic applications. For a detailed breakdown of the specific stoichiometric ratios and temperature profiles, please refer to the standardized synthesis guide below.
- Perform Suzuki coupling between alkoxytriphenylamine-substituted dithiophene pyrrole bromoaldehyde and indoline boronate ester using Pd(PPh3)4 catalyst in DME solvent under reflux.
- Purify the intermediate fluorenyl or trimeric indenyl indoline-bis-thiophene pyrrole formaldehyde via column chromatography using petroleum ether and ethyl acetate.
- Conduct Knoevenagel condensation with cyanoacetic acid and piperidine in acetonitrile/chloroform mixture under reflux to yield the final organic dye.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from ruthenium-based complexes to these organic dyes presents a compelling value proposition driven by raw material availability and process simplicity. The elimination of precious metals removes a significant cost driver and mitigates the risks associated with geopolitical supply disruptions common in the mining sector. Furthermore, the synthetic route utilizes commodity chemicals such as potassium carbonate, piperidine, and common organic solvents, which are readily accessible from multiple global suppliers, enhancing supply chain resilience. The high reaction yields observed in the patent examples suggest that waste generation is minimized, aligning with modern environmental compliance standards and reducing the burden on waste treatment facilities.
- Cost Reduction in Manufacturing: The absence of expensive noble metal catalysts and precursors fundamentally alters the cost structure of the photosensitizer production. By replacing ruthenium with abundant organic building blocks, manufacturers can achieve substantial cost savings without compromising on performance metrics. Additionally, the simplified purification protocols, which rely on standard chromatography rather than complex metal scavenging techniques, reduce both the time and consumables required for final product isolation, further driving down the operational expenditure per kilogram of produced dye.
- Enhanced Supply Chain Reliability: The reliance on widely available organic intermediates such as indoline derivatives and thiophene aldehydes ensures a stable supply base that is not subject to the volatility of the precious metals market. This stability allows for more accurate long-term planning and inventory management, reducing the lead time for high-purity organic dye photosensitizers delivery. The robustness of the Suzuki coupling reaction also means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain fluctuations.
- Scalability and Environmental Compliance: The reaction conditions employed, such as refluxing in DME or acetonitrile, are well-suited for scale-up in standard stainless steel reactors, facilitating the commercial scale-up of complex organic intermediates. The use of non-halogenated solvents in the initial coupling step and the ability to recover solvents through distillation contribute to a greener manufacturing footprint. This alignment with environmental regulations reduces the regulatory burden on production facilities and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dye technology in solar cell manufacturing. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance expectations and process feasibility. Understanding these details is crucial for R&D teams evaluating the integration of these materials into existing production lines.
Q: What are the key advantages of dithiophene pyrrole bridge indoline dyes over ruthenium complexes?
A: These organic dyes offer significantly higher molar absorptivity and eliminate the reliance on expensive noble metals like ruthenium, leading to substantial cost reductions and easier purification processes.
Q: How does the steric hindrance in these dyes improve solar cell performance?
A: The introduction of bulky groups such as fluorenyl or trimeric indenyl moieties effectively suppresses electron recombination at the semiconductor interface, thereby increasing the open-circuit voltage and overall photoelectric conversion efficiency.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis utilizes standard palladium-catalyzed cross-coupling and condensation reactions with readily available precursors, making it highly amenable to commercial scale-up without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dithiophene Pyrrole Bridge Indoline Organic Dyes Supplier
As the demand for efficient and cost-effective renewable energy technologies grows, the need for high-quality photosensitizers has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of dye meets the exacting standards required for high-performance DSSC applications. We understand the nuances of handling sensitive organic intermediates and have optimized our logistics to ensure timely delivery to our global partners.
We invite you to collaborate with us to explore how these advanced dyes can enhance your product portfolio. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your journey towards more sustainable and profitable solar energy solutions.
