Scalable Synthesis of Ergothioneine Key Intermediate via Novel Acetone Route
Scalable Synthesis of Ergothioneine Key Intermediate via Novel Acetone Route
The global demand for ergothioneine (EGT), a potent naturally occurring antioxidant, has surged due to its applications in functional foods, cosmetics, and pharmaceuticals. As a rare chiral amino acid, EGT plays a critical role in cellular protection against oxidative stress and UV radiation. However, traditional supply sources relying on natural extraction from fungi are limited by low yields and high costs, while biosynthetic routes face fermentation control challenges. Consequently, the chemical synthesis of ergothioneine and its precursors has become a focal point for industrial innovation. Patent CN113248442B, published in early 2022, introduces a groundbreaking preparation method for a key ergothioneine intermediate, 2-(benzylthio)-5-(bromomethyl)-1H-imidazole. This technology leverages inexpensive acetone as a starting material, offering a robust alternative to expensive histidine-based routes. The structural versatility of ergothioneine, existing in thiol-thione tautomeric forms as shown below, necessitates precise synthetic control to ensure high purity and stability in the final active ingredient.
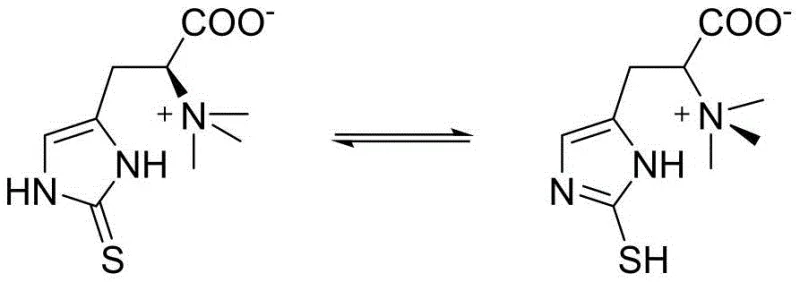
This patent represents a significant leap forward for reliable ergothioneine intermediate suppliers aiming to secure the supply chain for this high-value nutraceutical. By shifting the synthetic foundation from complex amino acids to simple ketones, the process fundamentally alters the economic landscape of EGT manufacturing. The methodology described not only simplifies the reaction sequence but also enhances environmental compliance by avoiding hazardous reagents commonly found in legacy protocols. For R&D directors and procurement managers alike, understanding the mechanistic nuances and commercial implications of this acetone-based pathway is essential for optimizing future production strategies and achieving cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of ergothioneine has heavily relied on L-histidine as the primary starting scaffold. While this approach mimics the natural biosynthetic precursor, it introduces severe economic and technical bottlenecks. L-histidine is a relatively expensive amino acid, and its utilization requires intricate protection and deprotection strategies to manage the reactivity of the alpha-amino and carboxyl groups. Furthermore, literature precedents, such as those cited in the patent background, often employ phosphorus tribromide (PBr3) to introduce the necessary bromomethyl functionality. PBr3 is notoriously hazardous, generating corrosive phosphorous acid waste that complicates downstream processing and increases environmental disposal costs. The reliance on chiral starting materials also means that any racemization during the harsh bromination steps leads to significant yield losses and difficult purification challenges. As illustrated in the prior art pathway below, the multi-step nature involving protection groups and hazardous halogenating agents creates a fragile supply chain vulnerable to raw material price fluctuations.

The Novel Approach
In stark contrast, the methodology disclosed in CN113248442B constructs the imidazole core de novo from acetone, trimethyl orthoformate, and bromine. This strategy bypasses the need for expensive chiral pools at the initial stages, utilizing commodity chemicals that are available in bulk quantities globally. The process flow is remarkably streamlined: acetone is first converted into 1,3-dibromo-2,2-dimethoxypropane, which then undergoes cyclization with potassium thiocyanate and ammonium acetate to form the imidazole ring directly. This eliminates the need for the dangerous PBr3 reagent entirely, replacing it with a safer bromine/methanol system and standard alkyl halides for the final protection step. The resulting route, depicted in the following scheme, demonstrates superior atom economy and operational simplicity. By decoupling the synthesis from the constraints of amino acid chemistry, this novel approach offers a scalable platform for commercial scale-up of complex pharmaceutical intermediates, ensuring a more stable and cost-effective supply of the critical 2-(benzylthio)-5-(bromomethyl)-1H-imidazole building block.
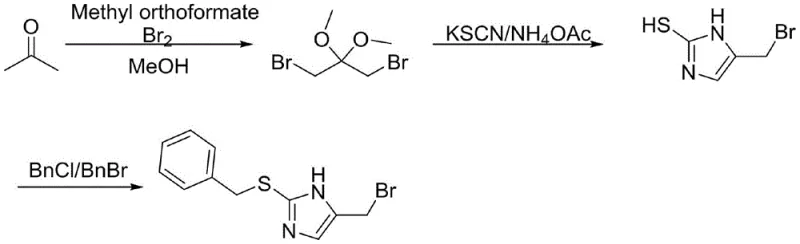
Mechanistic Insights into Acetone-Based Imidazole Cyclization
The core innovation of this patent lies in the efficient construction of the 2-thioimidazole ring from a linear dibromo-acetal precursor. The reaction begins with the bromination of acetone in the presence of trimethyl orthoformate and methanol. This step generates 1,3-dibromo-2,2-dimethoxypropane, a versatile electrophile that serves as a three-carbon synthon. The subsequent cyclization involves the nucleophilic attack of the thiocyanate anion (from KSCN) and the nitrogen source (ammonium acetate) on the electrophilic carbons of the dibromo species. The mechanism likely proceeds through an initial S-alkylation followed by intramolecular condensation to close the five-membered imidazole ring. The use of ammonium acetate is critical here, as it provides the necessary nitrogen atoms under mild conditions (35-45°C), preventing the degradation of the sensitive bromomethyl side chain. This mild thermal profile is a key differentiator from high-temperature cyclizations that often lead to polymerization or tar formation, thereby ensuring a cleaner reaction profile and higher crude purity.
Following the ring formation, the final step involves the S-alkylation of the 5-bromomethyl-1H-imidazole-2-thiol with benzyl chloride or benzyl bromide. This protection step is vital for stabilizing the thiol group against oxidation during subsequent synthetic transformations toward the final ergothioneine molecule. The patent specifies the use of inorganic bases like potassium carbonate or sodium carbonate in polar aprotic solvents such as acetonitrile or acetone. This choice of base and solvent system minimizes side reactions, such as N-alkylation, which is a common impurity in imidazole chemistry. The high selectivity for S-alkylation ensures that the nitrogen atoms on the imidazole ring remain free for future functionalization or salt formation. By controlling the stoichiometry and temperature (50-70°C), the process achieves high conversion rates while maintaining the integrity of the bromomethyl group, which is susceptible to hydrolysis or elimination under harsher basic conditions. This precise control over chemoselectivity is paramount for producing high-purity ergothioneine intermediates that meet stringent regulatory standards for human consumption.
How to Synthesize 2-(benzylthio)-5-(bromomethyl)-1H-imidazole Efficiently
The synthesis protocol outlined in the patent provides a clear, three-step roadmap for producing the target intermediate with high efficiency. The process is designed to be telescoped where possible, particularly between the formation of the dibromo-acetal and the cyclization step, minimizing isolation losses and solvent exchanges. Operators should pay close attention to temperature control during the initial bromination, maintaining the reaction between -5°C and +5°C to prevent poly-bromination or decomposition of the orthoformate. The subsequent cyclization requires careful monitoring of pH and water content to drive the equilibrium toward ring closure. Finally, the alkylation step benefits from the use of anhydrous conditions to maximize the yield of the benzyl-protected product. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- React acetone with trimethyl orthoformate and bromine in methanol at low temperature (-5°C to +5°C) to synthesize 1,3-dibromo-2,2-dimethoxypropane.
- Cyclize the dibromo intermediate using potassium thiocyanate and ammonium acetate in methanol at 35-45°C to form 5-bromomethyl-1H-imidazole-2-thiol.
- Perform S-alkylation by reacting the thiol intermediate with benzyl chloride or benzyl bromide in the presence of an inorganic base (e.g., potassium carbonate) to yield the final protected intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from histidine-based to acetone-based synthesis represents a strategic opportunity to optimize cost structures and mitigate supply risks. The reliance on L-histidine ties the production of ergothioneine intermediates to the volatile amino acid market, where prices can fluctuate based on fermentation capacity and agricultural feedstock costs. In contrast, acetone is a massive-volume commodity chemical with a stable and diversified supply base. This fundamental change in raw material sourcing drastically reduces the exposure to upstream price shocks. Furthermore, the elimination of phosphorus tribromide removes a significant logistical burden; PBr3 requires special handling, storage, and hazardous waste disposal protocols, all of which add hidden costs to the manufacturing process. By adopting this safer, simpler chemistry, manufacturers can achieve substantial cost savings not just in raw materials, but in operational overhead and environmental compliance.
- Cost Reduction in Manufacturing: The replacement of expensive chiral starting materials with commodity acetone fundamentally lowers the bill of materials. Additionally, the simplified workflow reduces the number of unit operations, leading to lower energy consumption and labor costs. The avoidance of complex protection/deprotection sequences associated with amino acid chemistry further streamlines the process, resulting in a more economically viable production model that supports competitive pricing for the final ergothioneine product.
- Enhanced Supply Chain Reliability: Sourcing acetone, bromine, and benzyl chloride is significantly more reliable than securing high-purity L-histidine, especially during global supply disruptions. The robustness of the chemical synthesis route ensures consistent output regardless of biological fermentation yields, which can be unpredictable. This stability allows for better production planning and inventory management, reducing lead time for high-purity pharmaceutical intermediates and ensuring uninterrupted supply to downstream formulation partners.
- Scalability and Environmental Compliance: The process operates under mild conditions and uses common solvents like methanol and acetonitrile, which are easily recovered and recycled on a large scale. The absence of heavy metal catalysts or highly corrosive reagents like PBr3 simplifies waste treatment and reduces the environmental footprint of the facility. This aligns with modern green chemistry principles and facilitates easier regulatory approval for commercial plants, making the commercial scale-up of complex intermediates smoother and faster.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in CN113248442B, providing clarity on how this technology compares to existing methods. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new pathway for their own production lines or for sourcing partners who utilize this specific chemistry.
Q: What are the primary cost advantages of the acetone-based route compared to histidine-based methods?
A: The acetone-based route utilizes commodity chemicals which are significantly cheaper and more readily available than L-histidine. Furthermore, it eliminates the need for complex chiral protection strategies in the early stages, streamlining the overall production cost.
Q: How does this new method improve safety compared to prior art involving phosphorus tribromide?
A: Conventional methods often rely on phosphorus tribromide (PBr3) for bromination, which is highly corrosive and hazardous. The patented method avoids PBr3 entirely by using a bromine/methanol system and standard alkyl halides, significantly reducing operational hazards and waste treatment complexity.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights the simplicity and stability of the operation. The use of common solvents like methanol and acetonitrile, along with mild reaction temperatures (mostly below 80°C), makes the process highly amenable to scale-up from pilot plant to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(benzylthio)-5-(bromomethyl)-1H-imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the acetone-based synthesis route for ergothioneine production. As a leading CDMO partner, we possess the technical expertise to translate laboratory patents into robust, industrial-scale processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the exacting standards required for pharmaceutical and nutraceutical applications. Our infrastructure is designed to handle the specific solvent systems and reaction conditions outlined in this patent, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Whether you require custom synthesis services or bulk procurement of the protected imidazole intermediate, our technical sales team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with NINGBO INNO PHARMCHEM can drive value and security into your ergothioneine supply chain.
