Advanced Manufacturing of Triazole Fungicide Intermediates via Lewis Acid Catalysis
Advanced Manufacturing of Triazole Fungicide Intermediates via Lewis Acid Catalysis
The global demand for high-efficacy fungicides such as propiconazole and difenoconazole necessitates robust, environmentally sustainable, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN113336715B, which discloses a novel preparation method for triazole compounds containing a dioxolane ring. This technology represents a paradigm shift from traditional halogenation-heavy processes to a streamlined Lewis acid-catalyzed acylation strategy. By directly introducing the triazolyl group via 1H-1,2,4-triazole-1-acetic acid derivatives, the invention effectively circumvents the formation of problematic 1,3,4-triazole isomers that plague conventional nucleophilic substitution methods. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: a pathway that significantly enhances reaction yield while drastically simplifying downstream purification, ultimately delivering a reliable agrochemical intermediate supplier solution that aligns with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
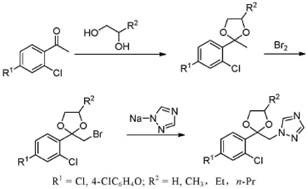
Historically, the synthesis of dioxolane-containing triazoles has relied heavily on multi-step sequences involving bromination, which introduces severe operational and environmental bottlenecks. Traditional routes often employ elemental bromine to activate the acetophenone precursor, a process characterized by poor atom utilization where nearly half the bromine mass is wasted as hydrogen bromide gas or acidic wastewater. Furthermore, the subsequent condensation of the brominated intermediate with 1,2,4-triazole is a nucleophilic substitution that frequently suffers from low yields and the generation of approximately 15% isomeric byproducts, specifically the 1,3,4-triazole variant, which complicates purification immensely. As illustrated in the reaction schemes of legacy processes, these methods often require harsh refining techniques, such as nitric acid salt formation or high-temperature distillation around 300°C, posing significant safety hazards and risking thermal decomposition of the sensitive triazole moiety.
The Novel Approach
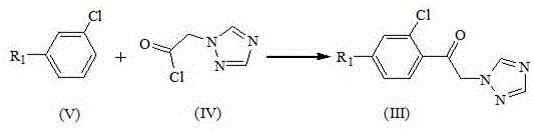
In stark contrast, the methodology outlined in CN113336715B adopts a direct Friedel-Crafts acylation strategy that fundamentally alters the economic and safety profile of the synthesis. Instead of brominating a ketone and then substituting, this novel approach reacts a substituted aromatic compound (Formula V) directly with 2-(1H-1,2,4-triazol-1-yl)-acetyl chloride (Formula IV) in the presence of a Lewis acid. This single-step construction of the carbon-carbon bond between the aromatic ring and the triazole-bearing side chain eliminates the need for hazardous bromine handling entirely. The process operates under mild conditions, typically between 0°C and 30°C, which preserves the integrity of the triazole ring and prevents the formation of the troublesome 1,3,4-isomer. By shifting the triazole introduction to the very beginning of the sequence, the subsequent ketalization step proceeds with exceptional cleanliness, allowing for product isolation via simple solvent crystallization rather than energy-intensive distillation.
Mechanistic Insights into Lewis Acid-Catalyzed Acylation and Ketalization
The core of this technological breakthrough lies in the precise control of electrophilic aromatic substitution mediated by strong Lewis acids such as anhydrous aluminum chloride, ferric chloride, or boron trifluoride. In the first stage, the Lewis acid activates the acid chloride moiety of the triazole-acetyl chloride, generating a highly reactive acylium ion complex. This electrophile attacks the electron-rich positions of the chlorobenzene or diphenyl ether substrate with high regioselectivity, driven by the steric and electronic properties of the existing substituents. The reaction is exothermic and requires strict temperature control, ideally maintained between 5°C and 10°C during the addition phase, to suppress poly-acylation and ensure the formation of the desired mono-substituted ketone (Formula III). The use of non-polar or moderately polar solvents like 1,2-dichloroethane or dichloromethane facilitates the solubility of the Lewis acid complex while allowing for easy aqueous workup to quench the catalyst and recover the product.
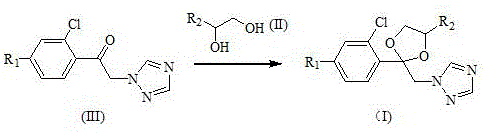
Following the isolation of the triazole ketone intermediate, the second mechanistic phase involves acid-catalyzed ketalization to form the dioxolane ring. This transformation utilizes p-toluenesulfonic acid as a proton source to activate the carbonyl oxygen of the ketone, rendering the carbonyl carbon susceptible to nucleophilic attack by the diol (Formula II). The reaction proceeds through a hemiketal intermediate, which subsequently undergoes dehydration to close the five-membered dioxolane ring. Crucially, the patent specifies the use of a Dean-Stark apparatus or similar water-separation technique during reflux in solvents like toluene or cyclohexane. This continuous removal of water drives the equilibrium towards the product side, ensuring near-quantitative conversion. The result is a structurally robust dioxolane-triazole hybrid (Formula I) that serves as the active scaffold for potent fungicides, achieved without the impurity profiles associated with older halogen-exchange methodologies.
How to Synthesize Triazole Dioxolane Compounds Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the Lewis acid catalysis. The process begins with the preparation of the acylium species in an inert atmosphere, followed by the controlled addition of the aromatic substrate. Post-reaction quenching must be performed cautiously to manage the heat of hydrolysis of the aluminum salts. Once the ketone intermediate is secured, the cyclization step demands efficient water removal to push the equilibrium. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and crystallization protocols validated in the patent examples, are provided in the guide below to ensure reproducible high-yield manufacturing.
- React a substituted chlorobenzene or diphenyl ether derivative with 2-(1H-1,2,4-triazol-1-yl)-acetyl chloride in the presence of a Lewis acid catalyst like anhydrous aluminum chloride at 0-30°C.
- Isolate the resulting triazole ketone intermediate through aqueous workup and solvent crystallization.
- Cyclize the ketone intermediate with a suitable diol (e.g., 1,2-pentanediol or 1,2-propanediol) using p-toluenesulfonic acid under reflux with water separation to form the final dioxolane product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages beyond mere chemical elegance. The elimination of elemental bromine not only removes a highly regulated and hazardous material from the supply chain but also eradicates the associated costs of corrosion-resistant equipment and specialized waste treatment for hydrobromic acid. By simplifying the purification process from high-temperature distillation or salt formation to straightforward solvent crystallization, the method significantly reduces energy consumption and processing time. This streamlining translates directly into lower operational expenditures and a more resilient production schedule, as the reliance on complex, failure-prone unit operations is minimized. Furthermore, the high selectivity of the reaction ensures a consistent supply of high-purity intermediates, reducing the risk of batch rejection and enhancing overall supply chain reliability for downstream formulation partners.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the workflow. By avoiding the use of expensive bromine and the subsequent neutralization of acidic byproducts, raw material costs are optimized. Additionally, the ability to isolate the final product through simple crystallization rather than energy-intensive vacuum distillation at 300°C leads to substantial savings in utility costs. The high yields reported, often exceeding 97%, mean that less raw material is wasted per kilogram of finished product, further driving down the cost of goods sold and improving margin potential for large-scale agrochemical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of widely available and stable starting materials. The reliance on commodity chemicals like chlorobenzenes, acetyl chlorides, and common diols reduces exposure to the volatility of specialized reagent markets. Moreover, the robustness of the reaction conditions—operating at near-ambient temperatures for the acylation step—reduces the likelihood of thermal runaways or equipment failures that can halt production lines. This stability ensures that delivery timelines for critical fungicide intermediates can be met consistently, supporting the just-in-time manufacturing models of major agricultural clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is inherently greener. The absence of bromine waste streams and nitric acid salt byproducts simplifies wastewater treatment and reduces the facility's environmental footprint. The process is designed for scalability, with exotherms that are manageable even in large reactors due to the low operating temperatures. This makes the technology ideal for commercial scale-up of complex agrochemical intermediates, allowing manufacturers to expand capacity without incurring prohibitive costs for new pollution control infrastructure or high-pressure high-temperature vessels.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production facilities or for sourcing partners who utilize this specific technology.
Q: How does this new route improve upon traditional bromination methods?
A: The new route eliminates the use of elemental bromine, thereby avoiding the generation of corrosive hydrogen bromide waste and improving atom economy. It also prevents the formation of difficult-to-separate 1,3,4-triazole isomers common in nucleophilic substitution methods.
Q: What are the critical quality parameters for the triazole ketone intermediate?
A: Critical parameters include maintaining a reaction temperature between 0-30°C to prevent side reactions and achieving a purity of over 98% through simple solvent crystallization, avoiding the need for high-temperature distillation which can degrade the product.
Q: Is this process scalable for industrial production of difenoconazole?
A: Yes, the process uses standard industrial reagents like anhydrous aluminum chloride and p-toluenesulfonic acid, operates at mild temperatures, and utilizes simple workup procedures like water washing and crystallization, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN113336715B is critical for maintaining competitiveness in the global agrochemical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Lewis acid-catalyzed process are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of triazole intermediate meets the exacting standards required for final API formulation, providing our partners with the confidence needed to navigate complex regulatory landscapes.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge technology for their fungicide portfolios. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on the foundation of the most efficient and sustainable chemistry available.
