Advanced Manufacturing of Cyclopropylethylamide: A Safer, High-Yield Route for Ciprofloxacin Intermediates
Introduction to Patent CN111269131B
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antibiotic intermediates, particularly for quinolones like ciprofloxacin hydrochloride. Patent CN111269131B introduces a transformative process for synthesizing cyclopropylethylamide, a pivotal precursor in this therapeutic class. This innovation addresses long-standing challenges in acylation chemistry by replacing hazardous reagents with safer, more efficient alternatives. Specifically, the technology utilizes tri-n-propylamine as a dual-function acid acceptor and solvent carrier, fundamentally altering the reaction thermodynamics and kinetics. By optimizing the reaction conditions to maintain temperatures between 40-60°C for condensation and 60-80°C for substitution, the process ensures high selectivity while minimizing degradation. This technical breakthrough not only enhances the purity profile of the final API intermediate but also aligns with modern green chemistry principles by facilitating easier solvent recovery and reducing toxic exposure risks for operators.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of cyclopropylethylamide relied heavily on tri-n-butylamine as the acid scavenger, a choice that introduced severe operational and economic bottlenecks. Tri-n-butylamine is classified as a highly toxic substance with a boiling point as high as 216.5°C, creating significant difficulties in separation and recovery processes. The high energy input required to distill off such a high-boiling solvent drastically increased manufacturing costs and carbon footprint. Moreover, the toxicity of tri-n-butylamine necessitated stringent safety protocols and specialized containment equipment to protect workers from ammonia-like vapors and potential health hazards. The difficulty in completely removing this high-boiling amine often led to residual impurities in the final product, complicating downstream purification steps and potentially affecting the quality of the final ciprofloxacin drug substance. These cumulative factors made the traditional route increasingly unsustainable in a cost-sensitive and regulation-heavy market environment.
The Novel Approach
The novel approach detailed in the patent elegantly circumvents these issues by substituting tri-n-butylamine with tri-n-propylamine, a reagent with a much more favorable physical profile. With a boiling point of only 156°C, tri-n-propylamine allows for significantly more energy-efficient recovery through distillation, directly translating to lower utility costs per kilogram of product. Its low toxicity classification removes the burden of handling hypertoxic chemicals, thereby improving the overall safety culture and reducing compliance costs associated with hazardous material management. Crucially, tri-n-propylamine acts not just as an acid acceptor but also as a solvent carrier, simplifying the reaction matrix and enhancing the conversion rates of both condensation and substitution steps to an impressive 97-99%. This dual functionality streamlines the workflow, reduces the total volume of auxiliary solvents needed, and boosts the overall yield to approximately 98%, offering a clear competitive advantage in commercial manufacturing scenarios.
Mechanistic Insights into Tri-n-propylamine Mediated Acylation
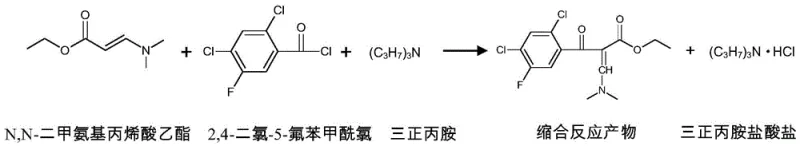
The core of this synthesis lies in the precise orchestration of nucleophilic acyl substitution, where the amine base plays a critical role in driving the equilibrium forward. In the initial condensation step, N,N-dimethylamino ethyl acrylate reacts with 2,4-dichloro-5-fluorobenzoyl chloride. The tri-n-propylamine rapidly scavenges the generated hydrogen chloride (HCl), preventing the protonation of the nucleophilic amine and avoiding the hydrolysis of the sensitive acid chloride moiety. This immediate neutralization is vital because the presence of free HCl could catalyze unwanted side reactions, such as the hydrolysis of the acid chloride into the corresponding carboxylic acid, which would act as a difficult-to-remove impurity. The reaction is maintained at a moderate 40-60°C to balance reaction kinetics with thermal stability, ensuring that the enamine structure remains intact while the acylation proceeds to completion.
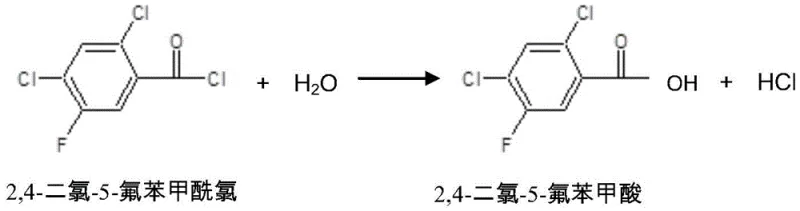
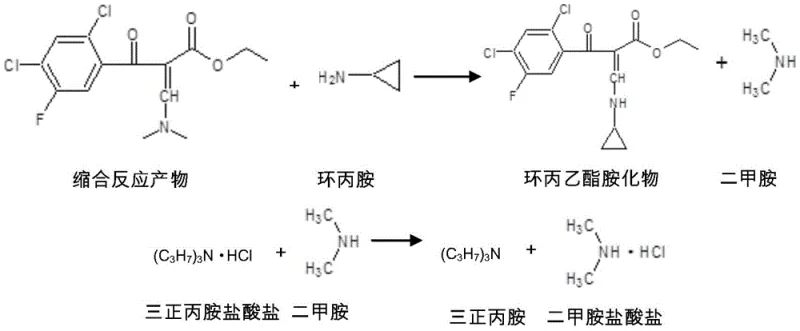
Following the condensation, the process moves to the substitution reaction where cyclopropylamine displaces the dimethylamino group. This step is equally sensitive to reaction conditions, requiring a temperature range of 60-80°C to overcome the activation energy barrier without degrading the cyclopropyl ring or the ester linkage. The mechanistic pathway involves a nucleophilic attack by the cyclopropylamine on the beta-carbon of the acrylate system, facilitated by the electron-withdrawing nature of the adjacent carbonyl groups. The presence of tri-n-propylamine continues to be beneficial here, maintaining a basic environment that keeps the cyclopropylamine in its free base form, maximizing its nucleophilicity. Furthermore, the process design includes a water washing step that selectively extracts the dimethylamine hydrochloride salt into the aqueous phase, effectively purifying the organic layer before crystallization. This strategic phase separation is key to achieving the high purity specifications required for pharmaceutical intermediates.
How to Synthesize Cyclopropylethylamide Efficiently
The synthesis protocol outlined in the patent provides a robust framework for manufacturing high-purity cyclopropylethylamide suitable for GMP environments. The process begins with the careful metering of raw materials into a reaction vessel, followed by controlled heating and stirring to ensure homogeneity. Critical parameters such as the mass ratio of reactants (specifically maintaining the tri-n-propylamine excess) and the precise temperature ramps are essential for reproducibility. The subsequent workup involving water washing, ethanol-assisted crystallization, and closed-system centrifugation ensures that the product is isolated with minimal solvent retention. For a comprehensive understanding of the specific operational parameters, including exact flow rates and equipment specifications, please refer to the standardized synthesis steps provided below.
- Conduct condensation reaction between N,N-dimethylamino ethyl acrylate, 2,4-dichloro-5-fluorobenzoyl chloride, and tri-n-propylamine at 40-60°C.
- Perform substitution reaction by adding cyclopropylamine to the mixture at 60-80°C to form the crude amide.
- Execute water washing, layering, cooling crystallization, and centrifugal drying to isolate the high-purity finished product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented process offers substantial strategic benefits that extend beyond simple yield improvements. The shift to tri-n-propylamine eliminates the need for managing highly regulated toxic substances, simplifying logistics, storage, and disposal procedures. This change significantly reduces the administrative burden and insurance costs associated with hazardous chemical handling. Additionally, the lower boiling point of the solvent carrier translates directly into reduced energy consumption during the recovery phase, offering a tangible reduction in manufacturing overheads without compromising on throughput. The ability to recycle ethanol and water within a closed-loop system further enhances the sustainability profile of the operation, making it attractive for companies with strict environmental, social, and governance (ESG) targets.
- Cost Reduction in Manufacturing: The elimination of high-boiling, toxic solvents like tri-n-butylamine removes the need for energy-intensive distillation columns capable of handling extreme temperatures. By utilizing tri-n-propylamine, which boils at a much lower 156°C, the facility can utilize standard glass-lined or stainless steel reactors with conventional heating systems, lowering capital expenditure (CAPEX) and operational expenditure (OPEX). The dual role of the amine as both reagent and solvent reduces the total volume of materials required, leading to smaller batch sizes for the same output or higher throughput in existing vessels. Furthermore, the high conversion rate of 97-99% minimizes raw material waste, ensuring that expensive starting materials like 2,4-dichloro-5-fluorobenzoyl chloride are utilized with maximum efficiency.
- Enhanced Supply Chain Reliability: Tri-n-propylamine is a widely available commodity chemical with a stable global supply chain, unlike specialized or restricted toxic amines that may face regulatory shipping constraints. This availability ensures consistent raw material sourcing, reducing the risk of production stoppages due to supply shortages. The simplified purification process, which relies on straightforward crystallization and centrifugation rather than complex chromatographic separations, shortens the overall cycle time per batch. Faster cycle times mean increased production capacity and the ability to respond more agilely to fluctuating market demands for ciprofloxacin intermediates. The robustness of the process also implies fewer failed batches, guaranteeing a steady flow of high-quality material to downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as washing, layering, and crystallization that are easily transferable from pilot plant to commercial scale (100 kgs to 100 MT). The closed-system design for centrifugation and drying prevents the release of volatile organic compounds (VOCs) into the workplace, ensuring compliance with occupational health standards. The integrated recovery systems for ethanol and water significantly reduce liquid waste discharge, lowering wastewater treatment costs and environmental impact. By converting the dimethylamine hydrochloride byproduct into a usable resource for producing N,N-dimethylamino ethyl acrylate, the process moves towards a circular economy model, minimizing the generation of hazardous solid or liquid waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent literature. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement officers assessing supplier capabilities. The focus is on practical implementation details, yield consistency, and the specific advantages of the tri-n-propylamine system over legacy methods.
Q: Why is tri-n-propylamine preferred over tri-n-butylamine in this synthesis?
A: Tri-n-propylamine offers a significantly lower boiling point (156°C vs 216.5°C), drastically reducing energy consumption during solvent recovery. Furthermore, it possesses low toxicity compared to the highly toxic tri-n-butylamine, ensuring a safer operational environment for personnel.
Q: What is the expected yield and conversion rate for this process?
A: The patented process achieves a conversion rate of 97-99% for both condensation and substitution reactions. The total product yield reaches approximately 98%, representing a tangible improvement over traditional methods.
Q: How does this process handle waste and solvent recycling?
A: The method incorporates a closed-loop system for recovering ethanol, tri-n-propylamine, and water. The aqueous dimethylamine hydrochloride byproduct is also separated for potential reuse in producing N,N-dimethylamino ethyl acrylate, minimizing environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropylethylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for essential antibiotic intermediates like cyclopropylethylamide. Our technical team has extensively analyzed the patented tri-n-propylamine route and validated its potential for large-scale commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are fully realized in practice. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical synthesis. We are committed to delivering high-purity intermediates that support the global demand for affordable and effective antimicrobial therapies.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of our material with your downstream processes. Let us collaborate to secure a sustainable and cost-effective source of high-quality cyclopropylethylamide for your ciprofloxacin production needs.
