Advanced Manufacturing of Naphthoquine Ester: A Technical Breakdown for Global Supply Chains
The pharmaceutical and veterinary industries are constantly seeking robust synthetic pathways that balance high purity with economic viability, particularly for broad-spectrum anticoccidial agents. Patent CN113979940A introduces a transformative methodology for the synthesis of naphthoquine ester, a critical quinoline derivative known chemically as 6-Butyl-1,4-dihydro-4-oxo-7-(phenyl methoxy)-3-quinolinecarboxylic acid methyl ester. This document details a strategic shift from traditional, high-cost precursors to more accessible starting materials, specifically replacing compound 3 with the economically superior compound 5. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediates supplier options, this patent represents a significant opportunity to optimize manufacturing costs while maintaining stringent quality standards. The disclosed route not only simplifies the operational complexity associated with high-temperature cyclization but also enhances the overall conversion efficiency, addressing long-standing pain points in the commercial production of this vital veterinary active ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of naphthoquine ester has been plagued by significant process inefficiencies that hinder large-scale commercialization. The prior art typically relies on the condensation of compound 2 with compound 3 to generate intermediate 4, followed by a rigorous cyclization step. A critical bottleneck in this legacy approach is the extreme sensitivity of the cyclization reaction to time and temperature; the reaction window is notoriously narrow, often requiring completion within less than 10 minutes at elevated temperatures. If the reaction time extends beyond this brief period, the nequinate product undergoes rapid thermal degradation and deterioration, severely impacting the purity profile and complicating downstream purification efforts. Furthermore, the reliance on compound 3 as a starting material imposes a substantial financial burden due to its higher market price compared to alternative reagents. These factors collectively result in low overall yields, difficult product isolation, and a manufacturing process that is inherently risky for industrial batch reactors, creating supply chain vulnerabilities for buyers of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to the limitations of the legacy route, the novel method disclosed in the patent leverages compound 5 (diethyl ethoxymethylenemalonate) to drive the condensation reaction, offering a distinct cost advantage and operational flexibility. By shifting the synthetic strategy to form intermediate 6 prior to cyclization, the process decouples the condensation and ring-closing steps, allowing for better control over reaction kinetics. The new protocol permits a cyclization window of 5 to 30 minutes at temperatures between 240°C and 260°C, a significant improvement over the precarious <10-minute window of the past. This extended reaction time reduces the risk of operator error and equipment failure, facilitating a more stable production environment. Additionally, the ability to recycle mother liquor for unreacted compound 6 ensures that raw material utilization is maximized, directly contributing to cost reduction in pharmaceutical intermediates manufacturing. The result is a streamlined pathway that achieves product yields up to 78 percent, demonstrating superior efficiency and scalability.
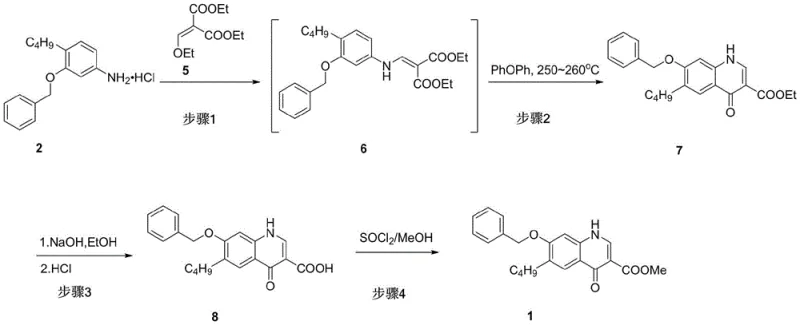
Mechanistic Insights into Condensation and Thermal Cyclization
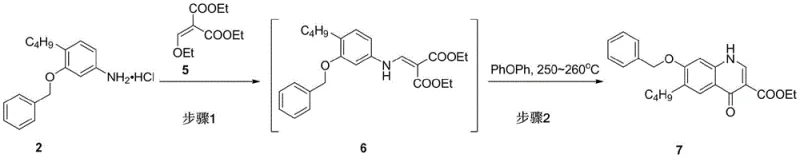
The core of this innovative synthesis lies in the precise orchestration of the condensation and subsequent thermal cyclization mechanisms. In the initial step, the free amine derived from compound 2 reacts with the ethoxy-methylene group of compound 5. This condensation is facilitated by heating in a solvent-free environment or within organic solvents like ethyl acetate at temperatures ranging from 60°C to 120°C. The mechanism involves the nucleophilic attack of the aniline nitrogen on the electron-deficient double bond of the malonate derivative, followed by the elimination of ethanol to form the enamine intermediate 6. This step is crucial as it sets the stage for the intramolecular ring closure. Unlike the prior art which struggled with unstable intermediates, this route generates a robust crude product 6 that can be carried forward directly without extensive purification, thereby reducing solvent consumption and processing time.
The subsequent cyclization step is a high-energy thermal transformation that constructs the quinoline core. When crude compound 6 is subjected to temperatures between 250°C and 255°C in a high-boiling solvent such as diphenyl ether, an electrocyclic ring closure occurs. This process involves the migration of the ethyl ester group and the loss of ethanol to aromatize the pyridone ring, yielding compound 7. The choice of solvent is mechanistically significant; diphenyl ether provides the necessary thermal stability and solvating power to sustain the reaction at 240-260°C without decomposing the sensitive quinoline scaffold. Following cyclization, the ethyl esters are hydrolyzed under basic conditions (NaOH or KOH) to form the dicarboxylic acid intermediate, which is then selectively decarboxylated and re-esterified using a methanol/thionyl chloride system. This final esterification step ensures the formation of the target methyl ester 1 with high regioselectivity and purity, effectively controlling the impurity profile by removing residual ethyl ester byproducts.
How to Synthesize Naphthoquine Ester Efficiently
The synthesis of naphthoquine ester via this novel route is designed for industrial robustness, beginning with the liberation of the free amine from its hydrochloride salt form. The process flows through a condensation phase to build the carbon skeleton, followed by a high-temperature cyclization to close the quinoline ring, and concludes with hydrolysis and esterification to finalize the functional groups. Each step has been optimized to minimize waste and maximize throughput, making it an ideal candidate for commercial scale-up of complex veterinary drug intermediates. Operators must pay close attention to the thermal parameters during the cyclization phase to ensure complete conversion while avoiding thermal stress on the product. For a comprehensive understanding of the specific reagent quantities, stirring rates, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Dissociate compound 2 with base, extract, and react with compound 5 at 60-120°C to form crude intermediate 6.
- Heat crude 6 in diphenyl ether at 240-260°C for 5-30 minutes to effect cyclization into compound 7.
- Hydrolyze compound 7 with alkali followed by acidification to obtain carboxylic acid 8.
- Perform methyl esterification on compound 8 using thionyl chloride and methanol to yield final product 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemistry. The primary driver for value creation is the substitution of expensive raw materials with cost-effective alternatives, which fundamentally alters the cost structure of the final API intermediate. By eliminating the dependency on compound 3 and utilizing the more abundant compound 5, manufacturers can achieve substantial cost savings that can be passed down the supply chain. Furthermore, the process improvements regarding reaction time windows reduce the likelihood of batch failures, ensuring a more consistent and reliable supply of material. This reliability is critical for maintaining continuous production schedules in the veterinary pharmaceutical sector, where demand for anticoccidial agents remains steady and predictable throughout the year.
- Cost Reduction in Manufacturing: The economic impact of switching to compound 5 cannot be overstated, as it removes a major cost center associated with the traditional synthesis of naphthoquine ester. The elimination of expensive precursors combined with the ability to operate without solvent in the initial condensation step significantly lowers the bill of materials. Additionally, the high conversion rates observed in the cyclization step mean that less raw material is wasted in the form of unreacted starting material or degradation byproducts. This efficiency translates directly into a lower cost per kilogram of the final product, providing a competitive edge in pricing negotiations for bulk purchases of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the new process parameters directly correlates to improved supply chain security. The extended reaction time window of 5 to 30 minutes, as opposed to the critical <10 minutes of the old method, reduces the operational risk associated with equipment lag or human error. This flexibility allows for the use of standard industrial reactors rather than specialized fast-response systems, broadening the base of potential manufacturing partners. Consequently, buyers can expect shorter lead times and greater consistency in delivery schedules, as the manufacturing process is less prone to the bottlenecks and batch rejections that characterized the legacy production methods.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the new route offers distinct advantages in waste management and energy usage. The ability to recycle mother liquor containing unreacted intermediate 6 minimizes the generation of hazardous chemical waste, aligning with modern green chemistry principles. Moreover, the use of common solvents like diphenyl ether and ethyl acetate simplifies solvent recovery and recycling processes. The process is inherently scalable, having been demonstrated to work effectively in multi-gram to kilogram scales in the patent examples, suggesting a smooth path to ton-scale production. This scalability ensures that suppliers can meet surging market demands without compromising on the stringent purity specifications required for veterinary drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of naphthoquine ester using the methodology described in patent CN113979940A. These insights are derived directly from the experimental data and process descriptions provided in the intellectual property documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains or formulation pipelines.
Q: What is the primary cost advantage of the new synthesis method described in CN113979940A?
A: The new method utilizes compound 5 (diethyl ethoxymethylenemalonate) instead of the more expensive compound 3 used in prior art, resulting in significant raw material cost reductions without compromising yield.
Q: How does the new process address the thermal degradation issues of previous methods?
A: Unlike the prior art which required extremely short reaction times (<10 minutes) leading to degradation, the new cyclization step allows for a controlled window of 5-30 minutes at 240-260°C, improving conversion rates and purity.
Q: What solvents are recommended for the high-temperature cyclization step?
A: High-boiling solvents such as diphenyl ether, biphenyl, glycerol, or Dowtherm A are suitable, with diphenyl ether being the preferred choice for optimal heat transfer and solubility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthoquine Ester Supplier
As the global demand for effective veterinary anticoccidial drugs continues to rise, securing a stable source of high-quality intermediates is paramount for pharmaceutical manufacturers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep technical expertise to deliver complex molecules with unmatched consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the largest multinational corporations. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of naphthoquine ester meets the highest international standards, providing our partners with the confidence they need to focus on their core drug development activities.
We invite you to collaborate with us to explore how this novel synthesis route can optimize your procurement strategy and reduce your overall manufacturing costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how NINGBO INNO PHARMCHEM can become your trusted partner in the sustainable production of advanced veterinary pharmaceuticals.
