Advanced Manufacturing of Triphenylbutene Derivatives for Selective Estrogen Receptor Modulators
Advanced Manufacturing of Triphenylbutene Derivatives for Selective Estrogen Receptor Modulators
The pharmaceutical landscape for Selective Estrogen Receptor Modulators (SERMs) continues to evolve, driven by the critical need for effective treatments for menopausal symptoms, osteoporosis, and cardiovascular health in postmenopausal women. Central to this therapeutic class are triphenylbutene derivatives, specifically ospemifene and fispemifene, which exhibit a unique tissue-specific profile combining estrogenic and anti-estrogenic properties. The efficient production of these high-value active pharmaceutical ingredients relies heavily on robust synthetic methodologies that ensure stereochemical purity and process scalability. Patent CN101636372A discloses a pivotal advancement in this domain, outlining a streamlined process for the preparation of therapeutically valuable triphenylbutene derivatives. This technology addresses historical bottlenecks in isomer separation and protecting group manipulation, offering a viable pathway for reliable pharmaceutical intermediate supplier networks aiming to secure the global supply of these critical medications.
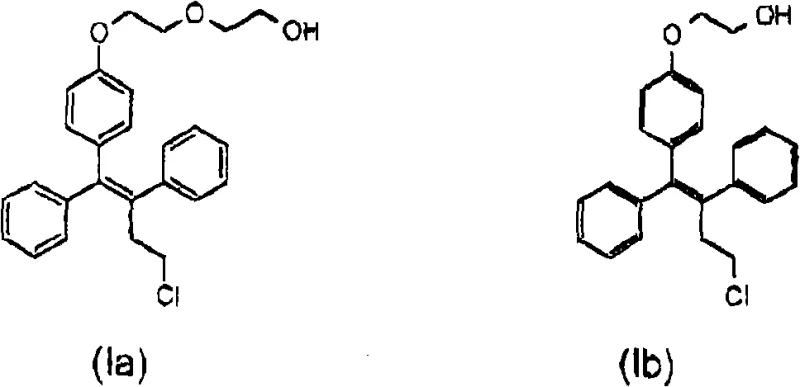
The core innovation lies in the strategic construction of the triphenylbutene backbone and the subsequent functionalization of the phenolic hydroxyl group. As illustrated in the structural representations, the target molecules possess a complex architecture featuring a chloro-substituted butenyl chain linked to a phenolic core, which is further extended by ethoxy chains. Achieving the correct Z-configuration at the double bond is paramount for biological activity, as the E-isomer often lacks the desired therapeutic efficacy. The disclosed methodology leverages the inherent stereoselectivity of transition metal-mediated coupling reactions to favor the Z-isomer directly, thereby reducing the reliance on extensive recrystallization campaigns that typically erode overall process yield and increase manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of compounds such as ospemifene and fispemifene has been plagued by multi-step sequences that introduce significant inefficiencies into the manufacturing workflow. Prior art, such as that described in WO02/090305, often necessitates the formation of dihydroxy-substituted butane intermediates which are subsequently converted to chloro-substituted triphenylbutenes. A major drawback in these legacy routes is the difficulty associated with isolating the desired Z-isomer from the reaction mixture, often requiring tedious fractional crystallization or chromatographic separation that is impractical on an industrial scale. Furthermore, conventional protection strategies frequently employ benzyl groups to mask the hydroxyl functionality during chain extension. The removal of benzyl protecting groups typically demands catalytic hydrogenation over palladium or harsh chemical conditions, which poses safety risks and can lead to the degradation of the sensitive olefinic bond, resulting in lower purity profiles and increased impurity burdens that complicate regulatory approval.
The Novel Approach
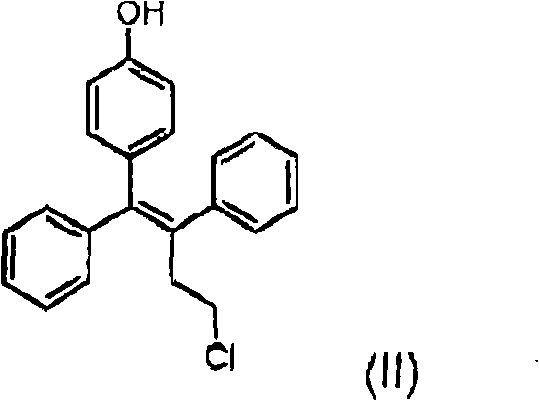
In stark contrast, the novel approach detailed in the patent data utilizes a convergent strategy centered on the direct formation of the alkene linkage via a McMurry-type reductive coupling. This method starts with readily available ketone precursors, specifically 4-hydroxyphenylbenzophenone and 3-chloropropiophenone, which are coupled in the presence of low-valent titanium species generated in situ. This reaction pathway is highly advantageous because it predominantly yields the Z-isomer of the phenolic intermediate, effectively solving the stereochemical control issue at the source. Additionally, the process introduces a superior protecting group strategy using tetrahydropyranyl (THP) moieties. Unlike the recalcitrant benzyl groups, THP protection allows for mild deprotection under acidic conditions, ensuring the structural integrity of the final molecule is maintained while facilitating a cleaner workup procedure. This shift in synthetic logic represents a substantial optimization in cost reduction in pharmaceutical intermediates manufacturing by minimizing unit operations and waste generation.
Mechanistic Insights into McMurry Reductive Coupling and Alkylation
The heart of this synthetic breakthrough is the McMurry reaction, a powerful tool for constructing carbon-carbon double bonds from carbonyl precursors. The mechanism involves the generation of low-valent titanium species, typically through the reduction of titanium tetrachloride with zinc dust in a solvent like tetrahydrofuran. These electron-rich titanium centers facilitate a single-electron transfer to the carbonyl oxygen of the ketone substrates, generating ketyl radical intermediates. These radicals rapidly dimerize to form a 1,2-diolate-titanium complex, which subsequently undergoes deoxygenation to yield the alkene. The stereoselectivity for the Z-isomer in this specific substrate combination is attributed to the steric constraints within the titanacycle intermediate, which favors a geometry that leads to the cis-alkene upon collapse. This mechanistic preference is crucial for the commercial scale-up of complex pharmaceutical intermediates, as it bypasses the thermodynamic equilibration that often favors the more stable E-isomer in acid or base-catalyzed eliminations.
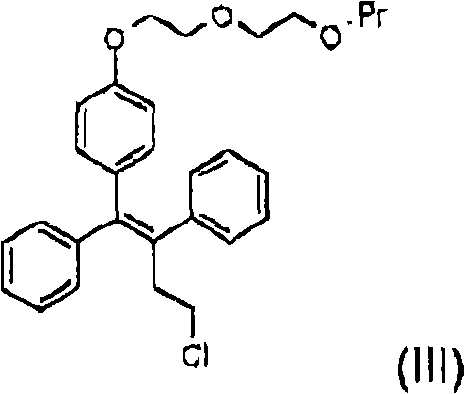
Following the formation of the phenolic core, the subsequent alkylation step is equally critical for installing the side chains required for biological activity. The process employs sodium hydride as a strong, non-nucleophilic base to deprotonate the phenolic hydroxyl group, generating a reactive phenoxide anion in situ. This anion then attacks the alkylating agent, such as a tetrahydropyranyl-protected iodo-ethoxy derivative, via an SN2 mechanism. The use of tetrahydrofuran as the solvent ensures excellent solubility for both the organic substrate and the ionic intermediates, promoting rapid reaction kinetics at moderate temperatures. The choice of the THP protecting group is mechanistically sound; it is stable to the basic conditions of the alkylation but labile to acid, allowing for orthogonal deprotection strategies. This precise control over reactivity ensures that the final product, whether it be the mono-ether ospemifene or the di-ether fispemifene, is obtained with high regioselectivity and minimal formation of over-alkylated byproducts.
How to Synthesize Triphenylbutene Derivatives Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters to maximize yield and purity. The initial coupling step is typically conducted by cooling a mixture of zinc and THF to approximately -10°C before the slow addition of titanium tetrachloride, followed by reflux to activate the metal surface. Once the low-valent titanium slurry is prepared, the ketone substrates are introduced, and the mixture is heated to drive the reductive coupling to completion. The resulting phenolic intermediate is then isolated and subjected to alkylation using sodium hydride in THF at room temperature or under reflux, depending on the reactivity of the specific alkylating agent. Finally, the protecting group is cleaved using aqueous acid or other mild methods to reveal the free hydroxyl or ethoxy functionalities. Detailed standardized synthetic steps see the guide below.
- Perform McMurry reductive coupling of 4-hydroxyphenylbenzophenone and 3-chloropropiophenone using Zn and TiCl4 in THF to generate the Z-isomer enriched phenol intermediate.
- Conduct alkylation of the phenol intermediate using sodium hydride in THF with a protected alkylating agent such as tetrahydropyranyl-protected ethoxy chains.
- Remove the protecting group under acidic conditions or via hydrogenation to yield the final high-purity triphenylbutene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant simplification of the supply chain for raw materials. By utilizing commodity chemicals such as 4-hydroxyphenylbenzophenone and 3-chloropropiophenone as starting materials, manufacturers can mitigate the risk of supply disruptions associated with exotic or custom-synthesized reagents. Furthermore, the elimination of difficult-to-remove protecting groups like benzyl reduces the dependency on precious metal catalysts such as palladium, which are subject to volatile market pricing and supply constraints. This shift towards base-metal catalysis (zinc/titanium) and simple acid/base chemistry creates a more resilient and cost-effective manufacturing framework that is less susceptible to geopolitical fluctuations in the availability of critical mining outputs.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the reduction of processing steps and the avoidance of expensive purification technologies. By favoring the Z-isomer directly during the coupling reaction, the need for extensive recrystallization or preparative chromatography to remove the E-isomer is drastically minimized, leading to higher overall throughput and reduced solvent consumption. Additionally, the use of tetrahydropyranyl protection avoids the costly hydrogenation equipment and catalyst recovery systems required for benzyl deprotection, resulting in substantial capital expenditure savings and lower operating costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: The reliance on standard industrial solvents like tetrahydrofuran, ethanol, and ethyl acetate ensures that the manufacturing process can be easily replicated across multiple geographic locations without requiring specialized infrastructure. The robustness of the reaction conditions, which tolerate standard laboratory and plant-grade reagents, means that quality variations in raw materials are less likely to cause batch failures. This reliability translates into shorter lead times for high-purity pharmaceutical intermediates, allowing downstream drug product manufacturers to maintain leaner inventory levels and respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is designed for scalability with a reduced ecological footprint. The avoidance of heavy metal catalysts in the deprotection step simplifies waste stream management and reduces the burden of heavy metal residue testing in the final API, a critical regulatory requirement. The reaction byproducts, primarily zinc and titanium salts, are manageable within standard wastewater treatment protocols, and the use of recyclable solvents aligns with green chemistry principles. This compliance readiness facilitates faster regulatory approvals and ensures long-term operational continuity in increasingly stringent environmental jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these SERM intermediates. The answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: Why is the McMurry reaction preferred for synthesizing ospemifene intermediates?
A: The McMurry reaction utilizing low-valent titanium species inherently favors the formation of the Z-isomer during the reductive coupling of ketones. This stereoselectivity eliminates the cumbersome crystallization steps required in conventional methods to separate E and Z isomers, significantly simplifying the downstream purification process.
Q: What are the advantages of using tetrahydropyranyl (THP) protection groups?
A: Unlike benzyl protecting groups which require harsh hydrogenation or difficult chemical cleavage, tetrahydropyranyl groups are acid-labile. They can be removed efficiently under mild acidic conditions, such as using aqueous HCl, which preserves the integrity of the sensitive triphenylbutene backbone and improves overall process yield.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available starting materials like 4-hydroxyphenylbenzophenone and standard industrial solvents such as tetrahydrofuran and ethanol. The reaction conditions, including the use of sodium hydride for alkylation and standard workup procedures, are fully adaptable to multi-kilogram and metric ton scales without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylbutene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising synthetic routes disclosed in patents like CN101636372A can be successfully translated into reliable supply streams. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of triphenylbutene derivative meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance extends throughout the entire production lifecycle, from raw material sourcing to final packaging.
We invite global pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their pipeline projects. By engaging with our technical procurement team, clients can request a Customized Cost-Saving Analysis tailored to their specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your supply chain security and drive down the total cost of ownership for your critical drug substances.
