Advanced Synthesis of Rhodamine B Cystamine Derivatives for Commercial Scale-up
Advanced Synthesis of Rhodamine B Cystamine Derivatives for Commercial Scale-up
The development of functionalized fluorescent dyes is a cornerstone of modern bio-imaging and diagnostic technologies, yet achieving precise chemical modification on complex scaffolds remains a significant hurdle for industrial chemists. Patent CN103012419A introduces a robust methodology for synthesizing Rhodamine B derivatives linked to cystamine, a critical intermediate for creating thiol-sensitive fluorescent probes. This innovation addresses the longstanding challenge of controlling regioselectivity when reacting polyfunctional amines like cystamine with carboxylic acid dyes. By implementing a strategic Boc-protection protocol followed by controlled amidation, this process ensures the preservation of a single free amino group essential for downstream conjugation while maintaining the structural integrity of the disulfide bond. For R&D teams seeking reliable rhodamine B derivative supplier partnerships, understanding this mechanistic breakthrough is vital for developing next-generation imaging agents with superior signal-to-noise ratios.
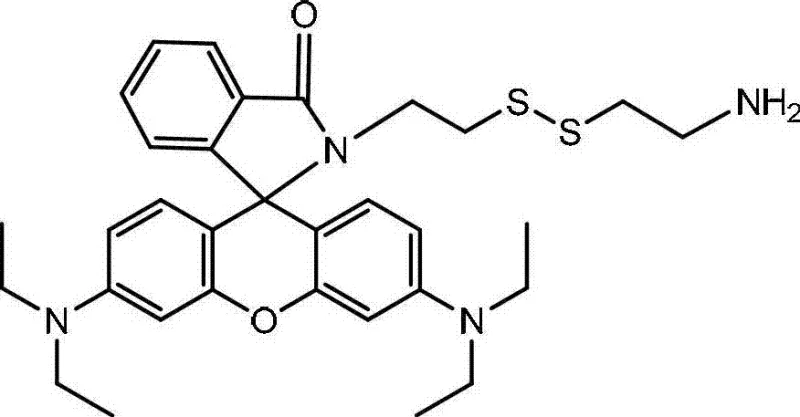
The significance of this synthesis extends beyond academic curiosity; it represents a viable pathway for the cost reduction in fluorescent probe manufacturing where purity and batch consistency are paramount. Traditional methods often struggle with the formation of bis-substituted byproducts or the cleavage of the sensitive disulfide linkage, leading to complex purification nightmares and reduced overall throughput. This patented approach circumvents these issues through a logical sequence of protection, coupling, and deprotection, offering a streamlined route that is highly amenable to scale-up. As we delve into the technical specifics, it becomes clear why this methodology is preferred for producing high-purity OLED material precursors and biological staining agents alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct condensation of Rhodamine B with diamines such as cystamine has been plagued by poor selectivity and unpredictable side reactions. In standard amidation protocols using activating agents like EDC or direct thermal condensation, the presence of two primary amino groups on the cystamine molecule creates a statistical probability of double substitution. This results in the formation of bis-Rhodamine adducts which are not only useless for the intended application but are also structurally similar enough to the target product to make separation via standard chromatography extremely difficult and costly. Furthermore, the harsh conditions often required to drive these unselective reactions can lead to the reduction or scrambling of the disulfide bond, effectively destroying the redox-sensitive functionality that makes cystamine valuable in the first place. These factors combined lead to low effective yields, excessive solvent consumption for purification, and a final product profile that fails to meet the stringent specifications required for pharmaceutical intermediates.
The Novel Approach
The methodology outlined in the patent data revolutionizes this synthesis by introducing a temporary masking strategy that fundamentally alters the reactivity profile of the cystamine substrate. By first treating cystamine dihydrochloride with di-tert-butyl dicarbonate (Boc2O) in the presence of triethylamine, chemists can selectively protect one of the two amino groups, creating a monoprotected intermediate that acts as a mono-functional nucleophile. This intermediate is then subjected to coupling with Rhodamine B using DCC and DMAP in dichloromethane, a condition that favors the formation of the desired amide bond without attacking the protected amine or the disulfide bridge. The final step involves the removal of the Boc group using trifluoroacetic acid, revealing the free primary amine ready for further bioconjugation. This stepwise logic ensures that the final product possesses exactly one reactive handle and one fluorophore, eliminating the need for difficult separations of bis-substituted impurities.
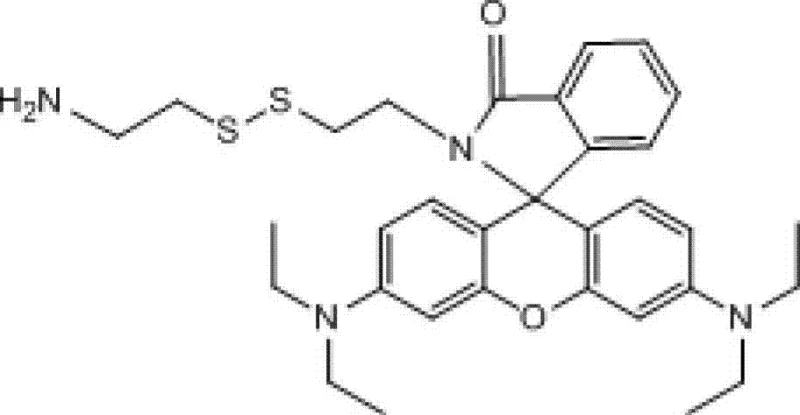
Mechanistic Insights into DCC/DMAP Catalyzed Amidation
The core of this synthesis relies on the efficient activation of the Rhodamine B carboxyl group using N,N'-dicyclohexylcarbodiimide (DCC) in the presence of 4-dimethylaminopyridine (DMAP). Mechanistically, DCC reacts with the carboxylic acid to form an O-acylisourea intermediate, which is highly electrophilic but prone to rearrangement into an unreactive N-acylurea if not intercepted quickly. DMAP serves as a potent nucleophilic catalyst that attacks this intermediate to form an even more reactive acylpyridinium species. This activated ester is then rapidly attacked by the free amino group of the monoprotected cystamine, driving the formation of the amide bond with high kinetic favorability. The use of dichloromethane as the solvent ensures that all organic components remain in solution during the reflux period of 6-14 hours, maximizing molecular collisions and conversion rates while preventing the precipitation of reactants that could stall the reaction kinetics.
Impurity control in this system is achieved primarily through the steric and electronic differentiation provided by the Boc group. Without this protection, the statistical likelihood of the second amine reacting is high, especially given the excess of activating agent often used to drive reactions to completion. However, the tert-butoxycarbonyl group significantly reduces the nucleophilicity of the protected nitrogen and adds steric bulk that hinders its approach to the activated carbonyl of the dye. Additionally, the workup procedures described, involving specific pH adjustments and extractions, are designed to remove the urea byproduct (dicyclohexylurea) and any unreacted starting materials efficiently. The final purification via silica gel column chromatography yields a white to light red powder, indicating a high degree of chemical homogeneity essential for consistent fluorescence performance in downstream applications.
How to Synthesize Rhodamine B Cystamine Derivative Efficiently
Executing this synthesis requires careful attention to stoichiometry and temperature control to maximize the yield of the monoprotected intermediate, which is the bottleneck of the entire process. The patent details a specific protocol where cystamine dihydrochloride is neutralized with three equivalents of triethylamine before the slow addition of Boc2O, ensuring that the reaction environment remains basic enough to facilitate nucleophilic attack on the carbonate without promoting hydrolysis. Following the isolation of the protected amine, the coupling step utilizes a slight molar excess of the amine relative to Rhodamine B to ensure complete consumption of the valuable dye. The final deprotection is conducted under mild acidic conditions to avoid damaging the xanthene core of the fluorophore. For a detailed breakdown of the standardized operating procedures and safety parameters, please refer to the technical guide below.
- Protect one amino group of cystamine dihydrochloride using di-tert-butyl dicarbonate (Boc2O) and triethylamine in methanol.
- Condense the monoprotected cystamine with Rhodamine B carboxyl group using DCC and catalytic DMAP in dichloromethane under reflux.
- Remove the Boc protecting group using trifluoroacetic acid (TFA) in dichloromethane to yield the final active amine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by utilizing commodity chemicals that are readily available in the global market, thereby reducing supply chain risk and volatility. Reagents such as Boc2O, DCC, and trifluoroacetic acid are standard inventory items for most fine chemical manufacturers, meaning there is no reliance on exotic or single-source catalysts that could disrupt production schedules. The elimination of complex transition metal catalysts also simplifies the regulatory landscape, as there is no need for extensive heavy metal testing or specialized removal processes that often add significant cost and time to the manufacturing of pharmaceutical intermediates. This simplicity translates directly into a more resilient supply chain capable of meeting fluctuating demand without the bottlenecks associated with specialized reagent sourcing.
- Cost Reduction in Manufacturing: The implementation of a protection-deprotection strategy significantly lowers the cost burden associated with purification. By preventing the formation of difficult-to-separate bis-substituted byproducts, the process reduces the volume of silica gel and solvents required for column chromatography, which are major cost drivers in fine chemical production. Furthermore, the high yields reported in the condensation and deprotection steps mean that less raw material is wasted, improving the overall atom economy of the process. This efficiency allows for a more competitive pricing structure for the final high-purity fluorescent probe precursors, making it an attractive option for large-scale commercial projects.
- Enhanced Supply Chain Reliability: The robustness of this chemistry ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream users in the diagnostics and imaging sectors. Because the reaction conditions are relatively mild and do not require specialized high-pressure or cryogenic equipment, the process can be easily transferred between different manufacturing sites or scaled up in existing facilities without significant capital expenditure. This flexibility enhances supply continuity, ensuring that customers receive their orders on time regardless of minor fluctuations in production capacity at any single location.
- Scalability and Environmental Compliance: The waste profile of this synthesis is manageable and aligns well with modern environmental standards, as the primary byproducts are organic compounds that can be incinerated or treated through standard wastewater protocols. The absence of heavy metals simplifies the disposal of spent catalyst beds and reduces the environmental footprint of the manufacturing process. Additionally, the scalability is proven by the use of standard unit operations like reflux and extraction, which are easily adapted from laboratory glassware to industrial reactors, facilitating the commercial scale-up of complex fluorescent intermediates without encountering unexpected engineering challenges.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, purity, and application of this specific Rhodamine B derivative. Understanding these nuances is essential for integrating this intermediate into your own research and development pipelines effectively. The answers provided are derived directly from the experimental data and technical specifications outlined in the source patent documentation.
Q: How does this method prevent double substitution on cystamine?
A: By selectively protecting one amino group with a Boc group prior to condensation, the method sterically and electronically blocks the second amino group from reacting with Rhodamine B, ensuring mono-substitution.
Q: What is the expected yield for the condensation step?
A: According to the patent data, the condensation step utilizing DCC/DMAP coupling achieves a high yield of approximately 90% after silica gel column chromatography purification.
Q: Is the disulfide bond stable during the synthesis?
A: Yes, the mild reaction conditions employed, specifically avoiding strong reducing agents or extreme pH shifts during the coupling phase, preserve the integrity of the critical disulfide bridge required for biological applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rhodamine B Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from pilot batches to full industrial supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch of Rhodamine B cystamine derivative we produce. Our commitment to quality assurance means that you can rely on us to deliver materials that meet the exacting standards required for sensitive biological applications and electronic material fabrication.
We invite you to engage with our technical procurement team to discuss how our optimized synthesis routes can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our manufacturing efficiencies can lower your overall material costs. We encourage potential partners to contact us directly to obtain specific COA data for our current inventory and to request route feasibility assessments for any custom modifications you may require. Let us help you accelerate your development timeline with our reliable supply of high-performance chemical intermediates.
