Advanced Sulfur Ylide Strategy for Scalable Paxlovid Intermediate Production
Advanced Sulfur Ylide Strategy for Scalable Paxlovid Intermediate Production
The global pharmaceutical landscape has witnessed an unprecedented demand for antiviral therapeutics, particularly following the emergence of SARS-CoV-2. At the heart of this medical response lies the critical need for efficient, safe, and scalable synthesis of key intermediates such as (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3,1,0]hexyl-2-carboxylic acid methyl ester hydrochloride, commonly referred to as Formula 1. This compound serves as the foundational scaffold for both PF-07321332, the active ingredient in Paxlovid, and Boceprevir, a established treatment for hepatitis C. Recent intellectual property developments, specifically patent CN115784968A, have unveiled a transformative synthetic methodology that replaces hazardous phosphorus-based reagents with a significantly safer sulfur ylide approach. This technological leap not only addresses long-standing safety concerns associated with pyrophoric reagents but also delivers substantial improvements in reaction yield and atom economy, positioning it as a preferred route for reliable pharmaceutical intermediate supplier networks aiming to secure the global supply chain.
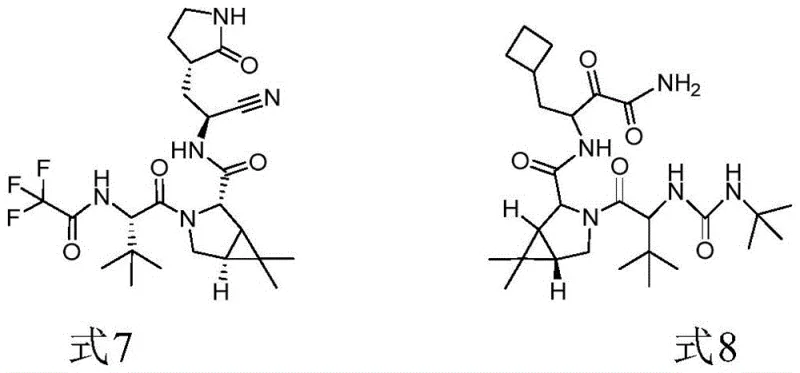
The strategic importance of Formula 1 cannot be overstated, as it represents the chiral core required for the biological activity of these potent protease inhibitors. The ability to manufacture this intermediate with high stereochemical purity and minimal impurity profiles is paramount for regulatory compliance and drug efficacy. The new method described in the patent leverages the unique reactivity of sulfur ylides generated in situ from diphenyl sulfide and 2-bromopropane. By shifting away from traditional organolithium chemistry, manufacturers can mitigate the risks of thermal runaways and fire hazards, thereby ensuring continuous operation and reducing the burden on safety infrastructure. This innovation underscores a broader industry trend towards greener, more sustainable chemical manufacturing processes that do not compromise on performance or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the 3-azabicyclo[3.1.0]hexane core has relied heavily on phosphorus ylide chemistry, a pathway fraught with significant operational and economic challenges. Conventional routes typically necessitate the use of n-butyllithium to generate the reactive ylide species, a reagent known for its spontaneous combustion upon exposure to air and violent reaction with moisture. This inherent danger mandates the use of specialized inert atmosphere equipment and rigorous safety protocols, which invariably drive up capital expenditure and operational complexity. Furthermore, alternative methods utilizing transition metal catalysts, such as cobalt dibromide with complex ligands, introduce issues related to heavy metal contamination and the high cost of ligand preparation. These legacy processes often suffer from moderate atom economy due to the use of poly-halogenated reagents like 2,2-dibromopropane, leading to increased waste generation and higher disposal costs that erode profit margins in cost reduction in pharmaceutical intermediate manufacturing initiatives.
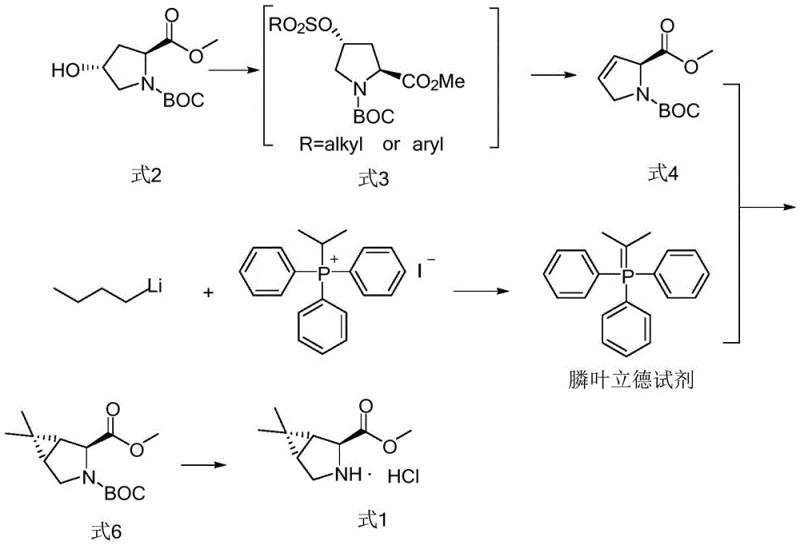
The Novel Approach
In stark contrast to these hazardous legacy methods, the novel approach detailed in patent CN115784968A introduces a robust sulfur ylide strategy that fundamentally alters the risk profile of the synthesis. By utilizing diphenyl sulfide and 2-bromopropane as precursors, the process generates the necessary ylide species under much milder conditions using potassium tert-butoxide as the base. This substitution eliminates the need for pyrophoric organolithium reagents entirely, allowing reactions to proceed safely at temperatures ranging from -25°C to -20°C without the threat of spontaneous ignition. The reaction sequence is streamlined into three distinct steps: the formation of the sulfonium salt, the generation of the ylide, and the subsequent cyclopropanation with the alkene precursor (Formula 4). This method not only simplifies the operational workflow but also achieves exceptional yields, with the key cyclopropanation step consistently exceeding 96% efficiency, thereby maximizing material throughput and minimizing waste.
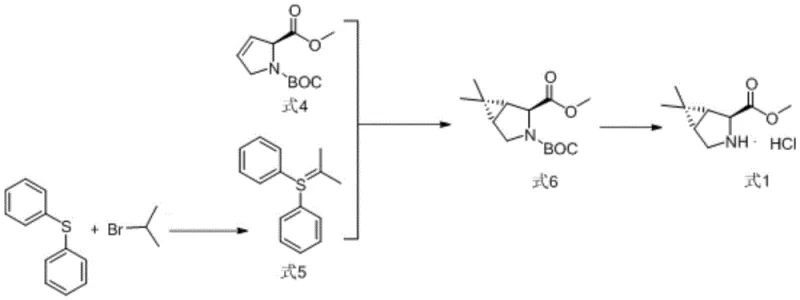
Mechanistic Insights into Sulfur Ylide Cyclopropanation
The core of this technological advancement lies in the precise generation and reactivity of the sulfur ylide intermediate. The mechanism initiates with the alkylation of diphenyl sulfide by 2-bromopropane to form a sulfonium salt, which is subsequently deprotonated by a strong base like potassium tert-butoxide to generate the reactive sulfur ylide species. Unlike phosphorus ylides which often require cryogenic conditions to maintain stability, the sulfur ylide generated in this system exhibits sufficient stability to react efficiently at moderately low temperatures. When introduced to the electron-deficient alkene of Formula 4, the ylide undergoes a concerted [2+1] cycloaddition. This process involves the nucleophilic attack of the ylide carbon on the alkene double bond, followed by the displacement of the sulfide leaving group, resulting in the formation of the strained cyclopropane ring fused to the pyrrolidine backbone. The stereochemical outcome is rigorously controlled by the chiral environment of the starting proline derivative, ensuring the exclusive formation of the desired (1R,2S,5S) configuration essential for biological activity.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of metal leaching into the final product, a critical quality attribute for pharmaceutical intermediates that often requires expensive scavenging steps to rectify. Additionally, the byproduct of the reaction is diphenyl sulfide, a stable and easily separable organic compound that does not pose the same environmental hazards as heavy metal salts or phosphine oxides. The reaction conditions are optimized to minimize side reactions such as polymerization or over-alkylation, which are common pitfalls in ylide chemistry. By maintaining strict temperature control between -25°C and -20°C during the addition of the base and the alkene, the process ensures high selectivity, resulting in a crude product profile that is amenable to straightforward purification, thus preserving the overall yield and purity of the high-purity pharmaceutical intermediate.
How to Synthesize (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3,1,0]hexyl-2-carboxylic acid methyl ester hydrochloride Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to replicate the high yields reported in the patent literature. The process begins with the preparation of the sulfur ylide reagent in tetrahydrofuran, followed by its immediate reaction with the alkene substrate in dichloromethane. The final step involves a deprotection and salt formation sequence using hydrogen chloride gas in methyl tert-butyl ether. Each stage has been optimized to balance reaction kinetics with safety, ensuring that the exothermic nature of the ylide formation is managed effectively. For R&D teams looking to adopt this technology, the detailed standardized synthesis steps are provided below to facilitate rapid technology transfer and process validation.
- Preparation of Sulfur Ylide Reagent: React diphenyl sulfide with 2-bromopropane followed by deprotonation with potassium tert-butoxide in tetrahydrofuran at -25°C to -20°C.
- Cyclopropanation Reaction: React the generated sulfur ylide with the alkene precursor (Formula 4) in dichloromethane at -25°C to -20°C to form the bicyclic structure.
- Deprotection and Salt Formation: Treat the protected intermediate with hydrogen chloride gas in methyl tert-butyl ether at 40°C to 50°C to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfur ylide methodology translates into tangible strategic benefits that extend beyond mere chemical elegance. The elimination of hazardous reagents like n-butyllithium removes a significant bottleneck in logistics and storage, as these materials often require specialized transport and dedicated containment facilities. By switching to stable, commercially available starting materials such as diphenyl sulfide and 2-bromopropane, organizations can streamline their supply chains and reduce the lead time for high-purity pharmaceutical intermediates. Furthermore, the simplified safety profile allows for manufacturing in a broader range of facilities, increasing supply redundancy and mitigating the risk of production stoppages due to safety audits or regulatory inspections. This resilience is crucial for maintaining the continuity of supply for life-saving medications like Paxlovid.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of expensive and dangerous reagents. Traditional methods relying on phosphorus ylides often incur high costs associated with the purchase of specialized phosphonium salts and the handling of pyrophoric bases. In contrast, the sulfur ylide route utilizes commodity chemicals that are significantly cheaper and easier to source in bulk quantities. Additionally, the high reaction yields exceeding 96% mean that less raw material is wasted, directly improving the cost of goods sold (COGS). The avoidance of heavy metal catalysts also eliminates the need for costly metal scavenging resins and the associated waste disposal fees, leading to substantial cost savings throughout the production lifecycle without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain stability is often compromised by the reliance on niche reagents with limited suppliers. The reagents used in this novel method, specifically diphenyl sulfide and 2-bromopropane, are produced by multiple global manufacturers, ensuring a competitive market and consistent availability. This diversification of the supply base reduces the risk of shortages that can plague specialized chemical markets. Moreover, the mild reaction conditions reduce the strain on utility systems, such as cryogenic cooling, making the process less susceptible to infrastructure failures. This robustness ensures that production schedules can be met reliably, providing downstream partners with the confidence needed for long-term planning and inventory management.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this sulfur ylide route is inherently designed for scalability. The absence of pyrophoric materials means that larger reactors can be operated with standard safety protocols rather than requiring bespoke engineering controls. From an environmental standpoint, the process generates less hazardous waste compared to metal-catalyzed routes, aligning with increasingly stringent global environmental regulations. The atom economy is superior, and the byproducts are non-toxic organic sulfides that can be potentially recycled or disposed of with lower environmental impact. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its potential for integration into existing manufacturing portfolios. The following questions address common inquiries regarding safety, yield, and scalability, drawing directly from the experimental data and technical specifications outlined in the patent documentation. These insights are intended to provide clarity on how this technology compares to incumbent methods and what operational changes might be required for adoption.
Q: Why is the sulfur ylide method safer than the traditional phosphorus ylide route?
A: The traditional route relies on n-butyllithium, a pyrophoric reagent that poses significant fire risks and requires specialized handling equipment. The new sulfur ylide method utilizes potassium tert-butoxide under mild conditions, drastically reducing operational hazards.
Q: What yields can be expected from this new synthesis method?
A: The patent data indicates that the cyclopropanation step achieves yields exceeding 96%, and the final deprotection step reaches over 97%, resulting in a total yield greater than 93%, which is superior to many conventional methods.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates at moderate temperatures (-25°C to 50°C) and avoids cryogenic conditions or expensive transition metal catalysts like cobalt dibromide, making it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3,1,0]hexyl-2-carboxylic acid methyl ester hydrochloride Supplier
As the demand for antiviral therapeutics continues to evolve, partnering with a manufacturer that possesses both the technical expertise and the capacity to deliver complex intermediates is critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the sulfur ylide route to ensure the consistent supply of high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We operate state-of-the-art facilities equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest standards of quality and safety required for API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this safer, more efficient process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your production costs and supply chain resilience. Together, we can drive the next generation of pharmaceutical manufacturing forward.
