Advanced Glycocholic Acid Manufacturing: Technical Upgrade and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic routes for critical bile acid derivatives, particularly glycocholic acid, which serves as a vital physiological marker and a functional excipient in drug delivery systems. A significant technological advancement in this domain is documented in patent CN115785184A, which discloses a novel preparation method that fundamentally alters the economic and safety landscape of manufacturing this high-value compound. Unlike traditional extraction methods that rely on limited animal sources, this chemical synthesis approach offers a consistent and scalable supply chain solution. The core innovation lies in a three-step sequence that bypasses the use of highly toxic reagents historically associated with bile acid conjugation. By leveraging a sulfonyl chloride activation strategy, the process achieves exceptional control over reaction kinetics and impurity profiles. This technical breakthrough is not merely a laboratory curiosity but represents a viable pathway for industrial adoption, addressing the growing demand for high-purity pharmaceutical intermediates. As we examine the molecular architecture of the target molecule, it becomes evident why precise synthetic control is paramount for maintaining its biological efficacy and safety profile.
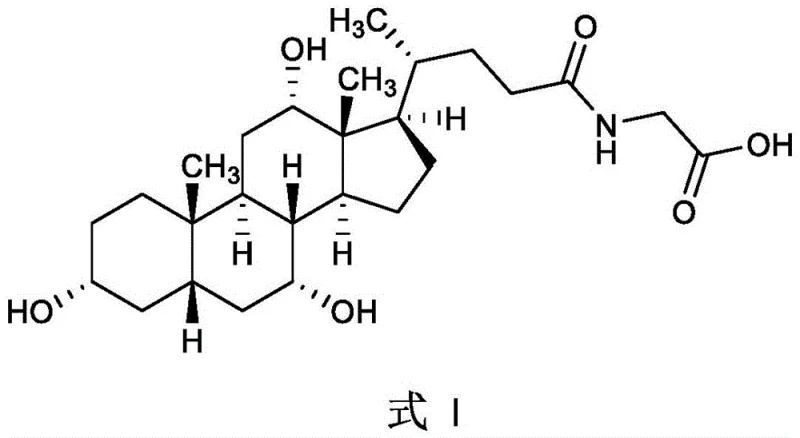
The structural integrity of glycocholic acid, as depicted above, requires meticulous preservation of the steroid nucleus during synthesis, a challenge that older methodologies often failed to address efficiently. The transition from legacy production methods to this modern catalytic approach signifies a major leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediates supplier status. The ability to synthesize this compound from readily available cholic acid without compromising on safety or environmental standards is a key differentiator in the current market. This report delves deep into the mechanistic advantages and commercial implications of adopting this specific synthetic route, providing actionable insights for R&D directors and procurement strategists alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of glycocholic acid has been plagued by significant operational hazards and economic inefficiencies that hinder large-scale adoption. Traditional pathways, such as the azide method, necessitate the use of diphenylphosphoryl azide or similar hazardous reagents that pose severe safety risks due to their potential explosivity and toxicity. Furthermore, the intermediates generated in these processes, such as azidocholic acid, exhibit poor stability, requiring stringent low-temperature controls that drive up energy consumption and equipment costs. Another prevalent method, the mixed anhydride approach, relies on alkyl chloroformates like methyl chloroformate, which are classified as highly toxic chemicals with strict regulatory handling requirements. The use of such substances complicates waste management protocols and increases the overall environmental footprint of the manufacturing process. Additionally, condensing agent methods utilizing reagents like DCC or EDCI introduce substantial material costs, as these coupling agents are expensive and generate difficult-to-remove byproducts that complicate downstream purification. These cumulative factors render conventional methods unsuitable for modern, cost-sensitive industrial production environments where safety and sustainability are non-negotiable priorities.
The Novel Approach
In stark contrast to the perilous legacy techniques, the novel method disclosed in the patent utilizes a sulfonyl chloride-mediated activation strategy that dramatically simplifies the synthetic workflow while enhancing safety. By reacting cholic acid with accessible sulfonyl chlorides, such as p-toluenesulfonyl chloride, in the presence of a mild organic base, the process generates a stable active intermediate that is far less prone to decomposition. This shift eliminates the need for cryogenic conditions and hazardous azide chemistry, thereby reducing the barrier to entry for commercial scale-up of complex bile acids. The subsequent coupling with glycine esters proceeds smoothly under moderate thermal conditions, ensuring high conversion rates without the formation of stubborn side products. Moreover, the final hydrolysis step employs standard aqueous base treatment, which is easily manageable in standard stainless steel reactors commonly found in fine chemical facilities. This methodological overhaul translates directly into cost reduction in API manufacturing by lowering raw material expenses and minimizing the need for specialized safety infrastructure. The result is a streamlined process that aligns perfectly with the operational requirements of a reliable pharmaceutical intermediates supplier seeking to optimize their production portfolio.
Mechanistic Insights into Sulfonyl Chloride-Mediated Activation
The success of this synthetic route hinges on the precise activation of the carboxylic acid moiety on the cholic acid side chain, a transformation that is elegantly achieved through sulfonylation. In the initial step, the carboxyl group of cholic acid reacts with the sulfonyl chloride in the presence of a tertiary amine base, such as N-methylmorpholine or triethylamine, to form a mixed anhydride-like sulfonate intermediate. This activation is crucial because it converts the relatively unreactive carboxylic acid into a highly electrophilic species capable of undergoing nucleophilic attack by the amino group of glycine ester. The choice of solvent, typically acetonitrile or acetone, plays a pivotal role in solubilizing the bulky steroid substrate while maintaining the reactivity of the sulfonylating agent. Temperature control during this exothermic activation phase is critical to prevent over-reaction or degradation of the sensitive steroid backbone. The formation of this active intermediate sets the stage for the subsequent amide bond formation, which is the defining structural feature of glycocholic acid.
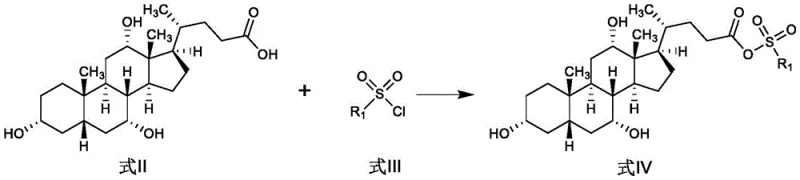
Following the activation, the coupling reaction with glycine ester proceeds via a nucleophilic substitution mechanism where the amine attacks the activated carbonyl carbon. This step is designed to minimize racemization and ensure the stereochemical integrity of the final product is maintained throughout the synthesis. The use of glycine esters rather than free glycine enhances solubility in the organic phase, driving the equilibrium towards the desired amide product. The reaction conditions are optimized to balance reaction rate with selectivity, ensuring that the hydroxyl groups on the steroid ring remain untouched. This selectivity is vital for preserving the biological activity of the molecule, as modification of the ring hydroxyls would render the product ineffective for its intended pharmaceutical applications. The mechanistic clarity of this pathway allows for precise troubleshooting and optimization during technology transfer.
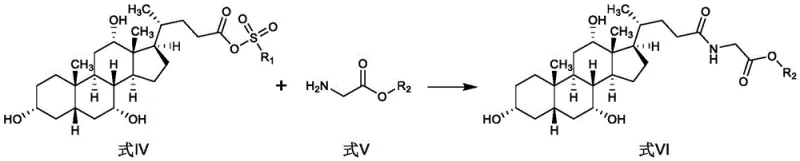
Impurity control is inherently built into this mechanism due to the clean nature of the byproducts formed during the reaction. The sulfonate leaving group and the amine salt byproducts are generally water-soluble or easily removable during the aqueous workup phases. This contrasts sharply with carbodiimide-mediated couplings which generate urea byproducts that are notoriously difficult to separate from the product. The final hydrolysis step cleaves the ester protecting group under basic conditions, releasing the free carboxylic acid of the glycine moiety to yield the final glycocholic acid. The simplicity of this hydrolysis ensures that no complex deprotection strategies are needed, further reducing the potential for impurity generation. By understanding these mechanistic nuances, R&D teams can better appreciate the robustness of the process and its suitability for producing high-purity glycocholic acid consistently.
How to Synthesize Glycocholic Acid Efficiently
Implementing this synthesis route requires a systematic approach to reaction engineering and process control to maximize yield and quality. The procedure begins with the careful dissolution of cholic acid in a polar aprotic solvent, followed by the controlled addition of the base and sulfonyl chloride to manage the exotherm. Detailed standardized synthesis steps are essential to ensure reproducibility across different batches and scales. The subsequent addition of the glycine ester solution must be timed precisely to coincide with the peak concentration of the active intermediate. Finally, the hydrolysis and isolation steps require precise pH control to ensure complete precipitation of the product while keeping impurities in the mother liquor. Adhering to these operational parameters is key to unlocking the full potential of this patented technology.
- Activate cholic acid using sulfonyl chloride and a base in an organic solvent to form a reactive intermediate.
- React the active intermediate with glycine ester under controlled temperature conditions to form glycocholate ester.
- Hydrolyze the glycocholate ester using an aqueous base followed by acidification to isolate the final glycocholic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method offers tangible strategic benefits that extend beyond simple unit cost metrics. The elimination of highly toxic reagents such as alkyl chloroformates and azides significantly reduces the regulatory burden and insurance costs associated with chemical manufacturing. This shift towards safer chemistry enhances supply chain reliability by minimizing the risk of production stoppages due to safety incidents or regulatory compliance issues. Furthermore, the use of commodity chemicals like sulfonyl chlorides and common solvents ensures that raw material sourcing is stable and less susceptible to market volatility compared to specialized coupling agents. The simplified post-treatment process, which avoids complex chromatographic purifications, leads to faster batch turnover times and improved asset utilization. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The replacement of expensive condensing agents with low-cost sulfonyl chlorides results in substantial cost savings on raw materials. Additionally, the avoidance of toxic reagents reduces the expenditure on specialized waste disposal and environmental remediation measures. The high yield and purity achieved directly reduce the cost of goods sold by minimizing material loss and reprocessing requirements. This economic efficiency makes the process highly attractive for large-scale production where margin optimization is critical.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials, the risk of supply disruption is significantly mitigated compared to methods requiring niche reagents. The robust nature of the reaction conditions allows for flexible manufacturing scheduling without the need for extreme temperature control infrastructure. This flexibility ensures consistent delivery performance, which is crucial for maintaining trust with downstream pharmaceutical clients. The process stability also reduces the likelihood of batch failures, ensuring a steady flow of product to the market.
- Scalability and Environmental Compliance: The absence of hazardous explosives or highly toxic gases makes this process inherently safer to scale from pilot plant to commercial tonnage. Waste streams are easier to treat due to the lack of persistent organic pollutants associated with older methods. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals. The ease of scale-up ensures that production capacity can be expanded rapidly to meet surging market demand without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glycocholic acid synthesis technology. These answers are derived directly from the patent specifications and practical process understanding to provide clarity on feasibility and performance. Understanding these details is crucial for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios. The information provided here serves as a foundational guide for further technical discussions and feasibility assessments.
Q: Why is the sulfonyl chloride method preferred over the azide method for glycocholic acid?
A: The azide method involves hazardous reagents and unstable intermediates posing safety risks, whereas the sulfonyl chloride route utilizes stable, low-cost materials with higher process safety suitable for industrial scale.
Q: What are the purity levels achievable with this new synthesis route?
A: Patent data indicates that recrystallization of the crude product yields glycocholic acid with purity exceeding 99.5%, meeting stringent pharmaceutical excipient standards.
Q: Is this process scalable for commercial production volumes?
A: Yes, the method avoids toxic reagents like methyl chloroformate and uses common solvents like acetonitrile, simplifying waste treatment and enabling safe commercial scale-up of complex bile acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycocholic Acid Supplier
The technical potential of this sulfonyl chloride-mediated synthesis route is immense, offering a pathway to high-quality bile acid derivatives that meet the rigorous demands of the global pharmaceutical market. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our commitment to stringent purity specifications and the operation of rigorous QC labs guarantees that every batch of glycocholic acid delivered meets the highest international standards. We understand the critical nature of supply continuity for our partners and have optimized our processes to deliver consistent quality and reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can benefit your specific application needs. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this safer, more efficient route. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Partner with us to secure a sustainable and competitive supply of high-purity pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
