Advanced Copper-Catalyzed Synthesis of 1,4-Diaryl-1,3-Butadiynes for Commercial Scale Manufacturing
The landscape of organic synthesis for conjugated diynes has evolved significantly with the introduction of patent CN101948365A, which details a robust and economically viable preparation method for 1,4-diaryl-1,3-butadiynes. These compounds serve as critical building blocks in the fabrication of advanced materials, ranging from photo-activated agrochemicals to sophisticated liquid crystal displays and pharmaceutical intermediates. Traditionally, the construction of the 1,3-diyne motif relied heavily on noble metal catalysis or harsh stoichiometric conditions, creating bottlenecks in supply chain efficiency and cost management for downstream manufacturers. This innovative approach leverages a ligand-free copper catalytic system that operates under remarkably mild thermal conditions, effectively addressing the long-standing challenges of toxicity and expense associated with previous methodologies. By utilizing easily accessible beta,beta-dibromo arylethylene precursors, this technology opens new avenues for the reliable supply of high-purity fine chemical intermediates essential for the global electronics and life sciences sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-diaryl-1,3-butadiynes has been plagued by significant operational and economic drawbacks that hinder large-scale adoption. Conventional routes often depend on palladium catalysts, which are not only prohibitively expensive due to the scarcity of the metal but also introduce severe toxicity concerns that complicate regulatory compliance and waste disposal. Alternative methods utilizing nickel catalysts require toxic ligands like dipyridine and strong bases such as sodium hydride, posing safety risks in an industrial setting. Furthermore, earlier copper-mediated approaches frequently necessitated stoichiometric amounts of copper salts rather than catalytic quantities, leading to excessive metal waste and difficult purification processes. Some prior art methods also demanded extreme reaction temperatures exceeding 120°C in volatile solvents like toluene, increasing energy consumption and the risk of thermal runaway incidents. These cumulative factors result in a fragmented supply chain where cost reduction in electronic chemical manufacturing remains elusive due to the inherent inefficiencies of the legacy synthetic pathways.
The Novel Approach
In stark contrast to these cumbersome traditional techniques, the novel methodology described in the patent introduces a streamlined catalytic cycle using cuprous iodide (CuI) in conjunction with 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU). This system operates efficiently in dimethyl sulfoxide (DMSO), a polar aprotic solvent that facilitates the reaction without the need for additional, air-sensitive ligands. The elimination of exogenous ligands simplifies the reaction mixture, thereby reducing the complexity of downstream purification and minimizing the generation of hazardous byproducts. Operating at a moderate temperature of 80°C, this process significantly lowers energy requirements compared to high-temperature alternatives while maintaining high conversion rates. The use of catalytic rather than stoichiometric copper further enhances the atom economy of the transformation, aligning with green chemistry principles. This breakthrough represents a paradigm shift for any reliable agrochemical intermediate supplier seeking to optimize their production capabilities for conjugated diyne structures.
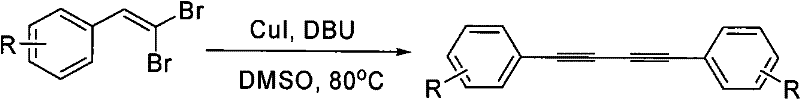
Mechanistic Insights into CuI-Catalyzed Homocoupling
The mechanistic underpinning of this transformation relies on the unique ability of the CuI-DBU system to activate the carbon-bromine bonds of the beta,beta-dibromo arylethylene substrate without external stabilization. In this catalytic cycle, DBU acts as a non-nucleophilic base that facilitates the dehydrohalogenation steps necessary to generate the reactive acetylenic species in situ. The copper center coordinates with the alkyne intermediates, promoting the oxidative coupling that forms the central 1,3-diyne linkage. Unlike palladium cycles that often require phosphine ligands to stabilize low-valent metal states, this copper system leverages the solvent and substrate interactions to maintain catalytic turnover. This ligand-free environment reduces the potential for ligand-derived impurities, which is a critical consideration for R&D directors focused on the purity profiles of API intermediates. The robustness of the mechanism allows for the tolerance of various electronic environments on the aromatic ring, ensuring consistent performance across a diverse range of substrates.
Impurity control is inherently enhanced by the mild reaction conditions and the specific selectivity of the copper catalyst. High-temperature methods often lead to polymerization or decomposition of the sensitive diyne product, resulting in complex mixtures that are difficult to separate. By maintaining the reaction at 80°C, the thermal stress on the product is minimized, preserving the integrity of the conjugated system. Furthermore, the absence of heavy metal contaminants like palladium simplifies the metal scavenging process, a crucial step in meeting stringent pharmaceutical specifications. The use of DMSO as a solvent also aids in solubilizing polar intermediates, preventing precipitation that could lead to incomplete reactions or localized hot spots. This level of control over the reaction environment ensures that the commercial scale-up of complex polymer additives or liquid crystal precursors can proceed with predictable quality outcomes and minimal batch-to-batch variation.
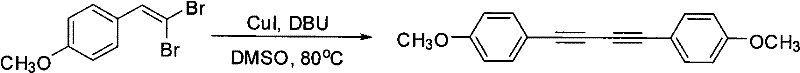
How to Synthesize 1,4-Diaryl-1,3-Butadiyne Efficiently
The practical implementation of this synthesis route is designed for straightforward execution in standard chemical processing equipment, making it highly accessible for contract manufacturing organizations. The process begins with the precise charging of the reactor with the beta,beta-dibromo arylethylene starting material, followed by the addition of the copper catalyst and the organic base in the DMSO solvent matrix. Maintaining the specified molar ratios is essential to drive the reaction to completion while minimizing excess reagent waste. The reaction progress is monitored to ensure full conversion before proceeding to the workup phase, which involves a simple aqueous quench and organic extraction. This operational simplicity reduces the training burden on plant personnel and minimizes the risk of operator error during scale-up. For detailed procedural specifics regarding reagent grades and exact mixing protocols, please refer to the standardized synthesis steps outlined below.
- Charge the reactor with DMSO solvent, beta,beta-dibromo arylethylene substrate, DBU base, and catalytic cuprous iodide.
- Stir the reaction mixture at 80°C for approximately 8 hours to ensure complete conversion.
- Quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology offers substantial opportunities for optimizing the cost structure of diyne-based supply chains. The replacement of precious metal catalysts with abundant copper salts directly translates to a significant reduction in raw material expenditure, freeing up capital for other areas of R&D investment. Additionally, the simplified purification workflow reduces the consumption of silica gel and eluents during chromatography, further driving down the cost of goods sold. For supply chain heads, the reliance on commercially available and stable reagents like DBU and DMSO mitigates the risk of supply disruptions often associated with specialized ligands or sensitive organometallic complexes. The mild operating conditions also extend the lifespan of reactor vessels and reduce maintenance downtime, contributing to overall operational efficiency. These factors collectively enhance the reliability of supply for high-purity OLED material precursors and other value-added intermediates.
- Cost Reduction in Manufacturing: The transition from stoichiometric or noble metal catalysts to a catalytic copper system fundamentally alters the economic model of production. By eliminating the need for expensive palladium and air-sensitive ligands, manufacturers can achieve drastic savings on input costs without compromising yield. The reduced metal loading also means lower costs associated with metal recovery and waste treatment facilities, which are often significant overheads in fine chemical manufacturing. Furthermore, the energy efficiency gained from operating at 80°C rather than higher temperatures contributes to a lower carbon footprint and reduced utility bills. These cumulative savings allow for more competitive pricing strategies in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: The robustness of the reagent profile ensures that production schedules are not held hostage by the availability of exotic or unstable chemicals. Beta,beta-dibromo arylethylene precursors are readily synthesized from common feedstocks, ensuring a steady upstream supply. The stability of the CuI/DBU system allows for longer shelf-life of prepared catalyst solutions, reducing waste from expired reagents. This reliability is paramount for reducing lead time for high-purity pharmaceutical intermediates, where just-in-time delivery is often a contractual requirement. Manufacturers can maintain leaner inventory levels of critical reagents, knowing that the supply chain is resilient to minor fluctuations in the market.
- Scalability and Environmental Compliance: The environmental profile of this process is superior to many legacy methods, facilitating easier regulatory approval for new manufacturing sites. The absence of toxic nickel or palladium residues simplifies the effluent treatment process, ensuring compliance with increasingly strict environmental discharge standards. The use of DMSO, while requiring proper handling, is a well-understood solvent in industrial hygiene, unlike some volatile organic solvents used in older protocols. This ease of compliance accelerates the timeline for technology transfer from lab to pilot to full commercial scale. Consequently, companies can bring new products to market faster, capitalizing on emerging opportunities in the liquid crystal and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis platform. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers reflect the balance between chemical efficiency and practical manufacturability that defines modern process chemistry.
Q: What are the primary advantages of this CuI-catalyzed method over palladium-based routes?
A: This method eliminates the need for expensive and toxic palladium catalysts and air-sensitive nitrogen ligands, significantly reducing raw material costs and simplifying waste treatment protocols.
Q: Can this synthesis protocol be scaled for industrial production of liquid crystal intermediates?
A: Yes, the use of mild reaction conditions (80°C) and readily available solvents like DMSO makes the process highly amenable to large-scale commercial manufacturing without requiring specialized high-pressure equipment.
Q: What types of substituents are tolerated on the aryl ring during this coupling reaction?
A: The protocol demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups such as methoxy, methyl, amino, and benzyloxy substituents at various positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Diaryl-1,3-Butadiyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in delivering high-value intermediates for the global marketplace. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to industrial reality. Our state-of-the-art facilities are equipped to handle the specific solvent and temperature requirements of this process, guaranteeing stringent purity specifications through our rigorous QC labs. We understand that consistency is key in the supply of electronic materials and pharmaceutical building blocks, and our quality management systems are designed to deliver exactly that level of assurance to our partners.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your next generation of products. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Together, we can optimize your supply chain and accelerate your time to market with reliable, high-quality chemical solutions.
