Advanced Synthetic Route for 3-Substituted-1H-Pyrrole Intermediates Enabling Commercial Scale-Up
The global demand for high-performance heterocyclic compounds continues to surge, particularly within the pharmaceutical and advanced materials sectors where specific substitution patterns dictate biological activity and electronic properties. Patent CN111116446B introduces a groundbreaking synthetic methodology for producing 3-substituted-1H-pyrrole compounds, addressing a long-standing challenge in organic synthesis regarding regioselectivity. Unlike conventional approaches that struggle to differentiate between the 2- and 3-positions on the pyrrole ring, this novel process utilizes a linear construction strategy starting from inexpensive and readily available raw materials such as diethylamine hydrochloride and glycine ethyl ester hydrochloride. The technology represents a significant leap forward for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering complex heterocycles with precision. By integrating a sequence of Mannich reaction, sulfonamidation, cyclization, dehydration, aromatization, and decarboxylation, the process ensures high structural fidelity and operational safety.
This comprehensive technical insight explores the mechanistic depth and commercial viability of this synthesis, providing R&D directors and procurement leaders with the data necessary to evaluate its integration into existing supply chains. The method not only solves the puzzle of 3-position substitution but also aligns with modern green chemistry principles by minimizing toxic waste and energy consumption. For industries ranging from drug discovery to conductive polymer manufacturing, mastering this pathway offers a competitive edge in securing high-purity building blocks essential for next-generation innovations.
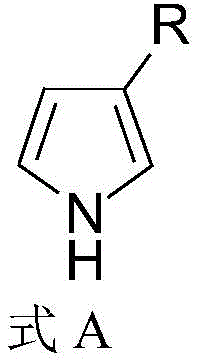
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole derivatives has been dominated by direct electrophilic substitution reactions, such as Friedel-Crafts alkylation or Vilsmeier-Haack formylation, which inherently favor the 2-position due to electronic activation and steric factors. Achieving substitution specifically at the 3-position has remained a persistent puzzle for chemists, often requiring convoluted protection-deprotection strategies or harsh conditions that compromise yield and safety. Traditional routes frequently suffer from poor regioselectivity, resulting in complex mixtures of isomers that are notoriously difficult and costly to separate on an industrial scale. Furthermore, many legacy processes rely on heavy metal catalysts or generate substantial amounts of hazardous wastewater, creating significant environmental liabilities and increasing the cost reduction in API manufacturing efforts. The instability of intermediates in older methods also poses risks during scale-up, limiting the ability to produce these valuable intermediates in the multi-ton quantities required by the global market.
The Novel Approach
In stark contrast, the methodology disclosed in CN111116446B constructs the pyrrole ring de novo from acyclic precursors, effectively bypassing the regioselectivity limitations of direct substitution. By employing a Mannich reaction to functionalize a ketone and a sulfonamidation to activate a glycine derivative, the process sets up a precise cyclization event that locks the substituent into the desired 3-position. This linear assembly line approach ensures that the carbon skeleton is built with atomic precision before the aromatic system is finalized. The route is characterized by mild operating conditions, utilizing common organic solvents and bases like potassium tert-butoxide and DBU, which are easier to handle and recycle than exotic reagents. As illustrated in the comprehensive reaction scheme below, the transformation proceeds through well-defined intermediates, allowing for rigorous quality control at each stage and facilitating the commercial scale-up of complex pharmaceutical intermediates.
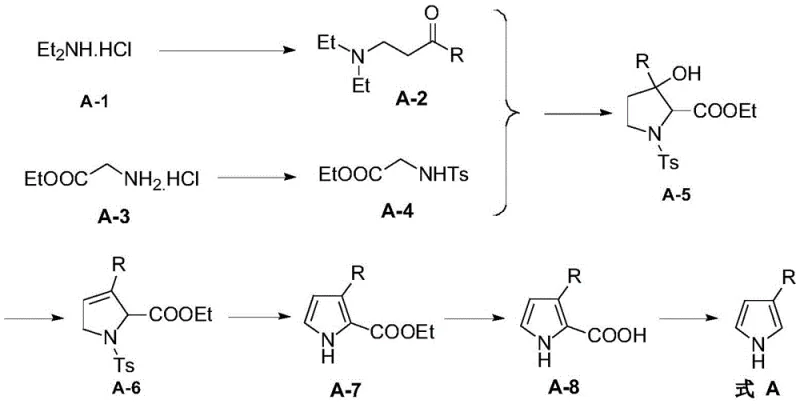
Mechanistic Insights into Base-Mediated Cyclization and Aromatization
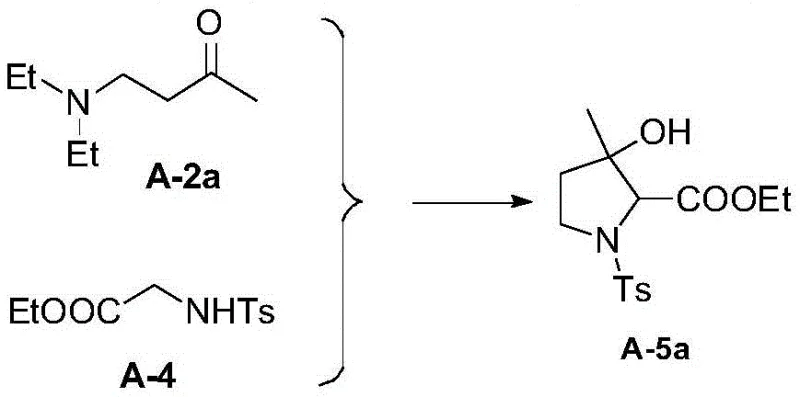
The core innovation of this synthesis lies in the cyclization and subsequent aromatization steps, which transform the linear precursors into the stable heterocyclic core. The cyclization reaction, typically mediated by a strong base such as potassium tert-butoxide in a mixed solvent system of THF and tert-butanol, involves the nucleophilic attack of the sulfonamide nitrogen onto the electrophilic center of the Mannich base derivative. This intramolecular condensation forms the five-membered pyrroline ring with high stereochemical control, establishing the critical 3-substituted architecture. The choice of base is paramount; optimization data indicates that bulky alkoxides like tBuOK provide superior yields compared to smaller hydroxides or amines, likely due to reduced side reactions and better solubility profiles in the organic phase. Following cyclization, the intermediate undergoes a dehydration step using phosphorus oxychloride in pyridine, which eliminates the hydroxyl group to introduce unsaturation, setting the stage for aromatization.
The final conversion to the aromatic pyrrole system is achieved through a base-mediated elimination of the sulfonyl protecting group, often utilizing DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene) in toluene at elevated temperatures. This step is crucial as it restores aromaticity while simultaneously removing the directing group, yielding the free NH pyrrole. The mechanism involves the abstraction of acidic protons and the expulsion of the sulfinate leaving group, driven by the thermodynamic stability of the aromatic product. Impurity control is maintained throughout this sequence by the robustness of the sulfonamide protection, which prevents polymerization or over-alkylation common in unprotected pyrrole chemistry. The detailed mechanism of the cyclization step is depicted below, highlighting the formation of the key pyrroline intermediate A-5.
How to Synthesize 3-Substituted-1H-Pyrrole Efficiently
Implementing this synthesis requires careful attention to stoichiometry and reaction conditions to maximize yield and purity. The process begins with the preparation of the Mannich base and the sulfonamide precursor, both of which are isolated as stable solids or oils suitable for storage. The critical cyclization step demands an inert atmosphere and precise temperature control to prevent degradation of the sensitive intermediates. Subsequent steps involving dehydration and aromatization utilize standard workup procedures such as extraction and crystallization, which are easily adaptable to large-scale reactor setups. The final decarboxylation step, performed in ethylene glycol at high temperatures, cleanly removes the ester functionality to deliver the target molecule. For a detailed breakdown of the standardized operating procedures and specific parameter ranges validated in the patent, please refer to the guide below.
- Perform Mannich reaction using diethylamine hydrochloride, aldehyde, and methyl ketone in alcohol solvent with acid catalysis to form compound A-2.
- Conduct sulfonamidation of glycine ethyl ester hydrochloride with sulfonyl chloride in the presence of base to yield compound A-4.
- Execute cyclization of A-2 and A-4 using strong base (e.g., potassium tert-butoxide) to form the pyrroline ring structure (A-5).
- Carry out dehydration, aromatization using organic base (DBU), followed by hydrolysis and thermal decarboxylation to obtain the final 3-substituted-1H-pyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers profound advantages that directly address the pain points of modern supply chain management. By shifting away from unpredictable direct substitution methods to a controlled linear synthesis, manufacturers can secure a more reliable supply of 3-substituted-1H-pyrrole derivatives. The use of commodity chemicals like diethylamine and glycine esters as starting materials insulates the production cost from the volatility associated with specialized fine chemical feedstocks. This foundational stability allows for consistent pricing models and long-term supply agreements, which are critical for procurement managers planning annual budgets for active pharmaceutical ingredient production. Furthermore, the high atom economy and reduced waste generation translate into lower disposal costs and simplified regulatory compliance, enhancing the overall sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reliance on inexpensive, bulk-available reagents significantly lowers the raw material cost base. The process avoids the need for complex chromatographic purifications, relying instead on crystallization and extraction, which drastically reduces solvent consumption and processing time. This streamlined workflow minimizes labor and utility expenses, driving down the total cost of ownership for the final intermediate. Additionally, the high yields reported in the patent examples suggest that less raw material is wasted per unit of product, further amplifying the economic efficiency of the operation.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch performance. The intermediates generated along the pathway are stable and can be stockpiled if necessary, providing a buffer against supply disruptions. This flexibility allows suppliers to respond more agilely to sudden spikes in demand from downstream clients. The simplicity of the equipment requirements—standard glass-lined or stainless steel reactors—means that production can be easily transferred between different manufacturing sites without extensive requalification, safeguarding continuity of supply.
- Scalability and Environmental Compliance: Designed with industrial application in mind, the process operates under mild conditions that do not require extreme pressures or cryogenic temperatures, reducing energy consumption and safety risks. The avoidance of toxic heavy metals simplifies the wastewater treatment process, aligning with increasingly stringent environmental regulations globally. This green chemistry profile not only reduces the environmental footprint but also mitigates the risk of production shutdowns due to compliance issues. The scalability is evidenced by the successful demonstration of the route in multi-kilogram batches, proving its readiness for ton-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and benefits of adopting this route. Understanding these nuances is essential for technical teams evaluating the fit of this intermediate within their specific development pipelines.
Q: How does this process overcome the regioselectivity issues of traditional pyrrole synthesis?
A: Traditional electrophilic substitution often favors the 2-position. This patented route constructs the ring from linear precursors (Mannich base and sulfonamide), ensuring exclusive substitution at the 3-position through controlled cyclization and subsequent aromatization steps.
Q: What are the scalability advantages of this synthetic method?
A: The process utilizes mild reaction conditions, avoids hazardous transition metal catalysts, and employs common industrial solvents. The high repeatability and robust purification steps (crystallization/extraction) make it highly suitable for kilogram-to-ton scale production.
Q: What purity levels can be achieved with this methodology?
A: Experimental data indicates that the final 3-substituted-1H-pyrrole products achieve purity levels greater than 99%, with low impurity profiles, meeting stringent requirements for pharmaceutical and electronic material applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted-1H-Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medicines and advanced materials. Our team of expert chemists has extensively analyzed the synthetic pathway described in CN111116446B and possesses the technical capability to execute this complex sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from laboratory bench to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-substituted-1H-pyrrole meets the highest industry standards for impurity profiles and physical properties.
We invite you to collaborate with us to leverage this innovative synthesis for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget. Let us be your partner in turning complex chemical challenges into commercial successes.
