Advanced Six-Step Route for High-Purity Alpha-Methyl-Amino Acid Intermediates
Advanced Six-Step Route for High-Purity Alpha-Methyl-Amino Acid Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and scalable synthetic routes for complex molecules. A pivotal development in this field is detailed in patent CN101565390A, which discloses a robust methodology for preparing Alpha-methyl-Alpha, Alpha-disubstituted-Alpha-aminophenol and its derivatives. These compounds serve as critical building blocks for alpha-non-natural amino acids, which are increasingly vital in the design of protease inhibitors, stable peptides, and advanced bio-pharmaceuticals. By leveraging a novel six-step organic chemistry sequence, this technology addresses the longstanding challenges of optical purity and production costs associated with traditional synthesis methods. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for securing a competitive edge in drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure Alpha-Methyl-α, α-disubstituted-amino acids has relied heavily on cyclic chiral template molecules such as oxazolidinones, imidazolones, or diketopiperazines. While effective in laboratory settings, these conventional approaches suffer from significant drawbacks that hinder industrial application. The synthesis of the chiral templates themselves often involves lengthy multi-step sequences requiring uncommon reagents, which drastically inflates the raw material costs and complicates the supply chain logistics. Furthermore, the key alkylation steps in these traditional routes frequently demand harsh reaction conditions, such as the use of strong bases like n-butyllithium or lithium diisopropylamide at cryogenic temperatures below -70°C. These extreme conditions not only pose severe safety risks in a manufacturing environment but also require specialized, energy-intensive equipment, making the commercial scale-up of complex polymer additives or pharmaceutical intermediates economically unviable.
The Novel Approach
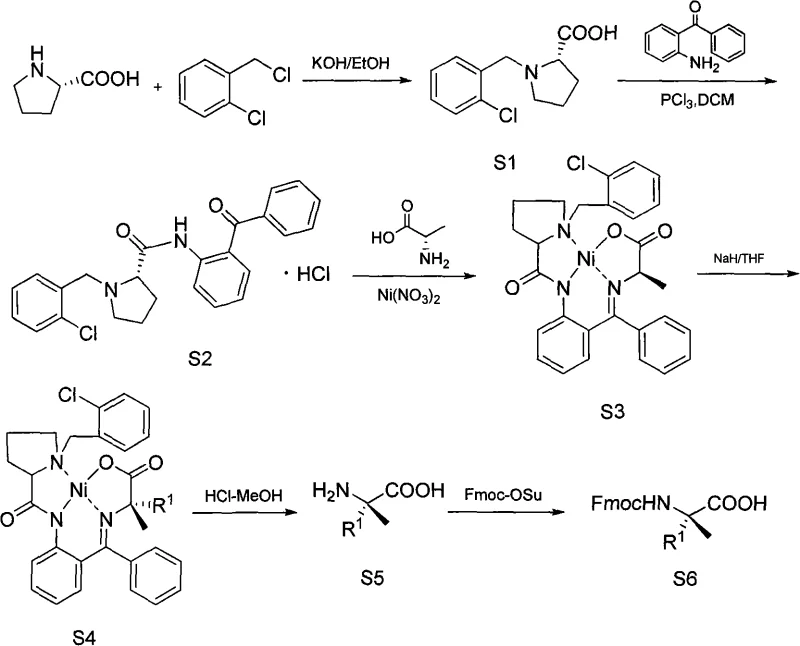
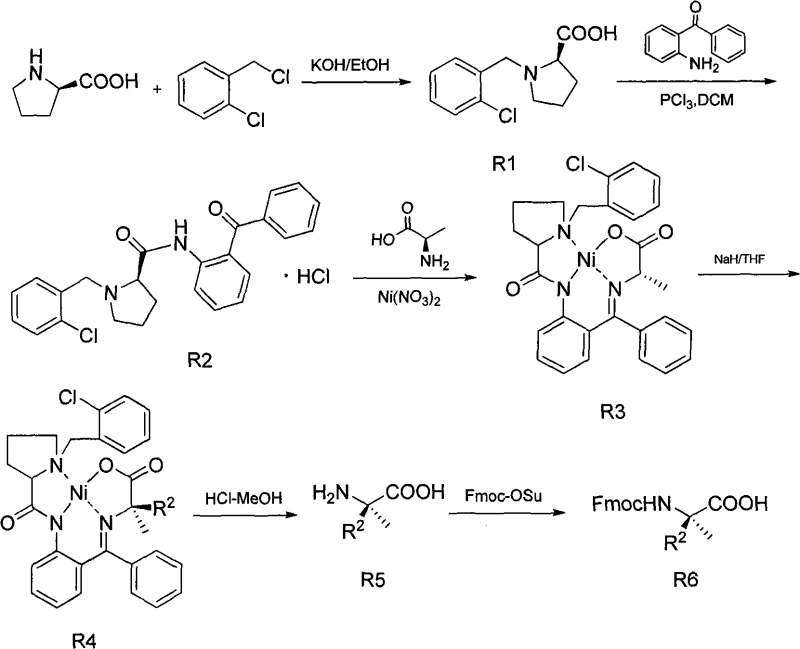
In stark contrast, the methodology outlined in the referenced patent introduces a streamlined pathway that circumvents these bottlenecks by utilizing a nickel-complex mediated strategy. This novel approach initiates with readily available and low-cost starting materials, specifically L-proline or D-proline, reacting with o-chlorobenzyl chloride to form the foundational intermediates S1 or R1. The subsequent steps involve the formation of a Schiff base with 2-aminobenzophenone and the crucial complexation with nickelous nitrate and alanine. This creates a rigid chiral environment that facilitates highly selective alkylation under much milder conditions, typically between 0°C and 10°C for the alkylation step and 40-60°C for earlier condensations. By eliminating the need for expensive chiral catalysts like palladium or hazardous cryogenic reagents, this process offers a substantial cost reduction in pharmaceutical intermediate manufacturing while ensuring the high optical purity required for downstream biological applications.
Mechanistic Insights into Nickel-Complex Mediated Alkylation
The core innovation of this synthesis lies in the formation and utilization of the chiral nickel(II) complex, designated as S3 or R3 in the patent documentation. This complex acts as a glycine equivalent, where the alanine moiety is coordinated to the nickel center within a rigid planar geometry enforced by the benzophenone imine ligand. When treated with a base such as sodium hydride in tetrahydrofuran (THF), the alpha-proton of the alanine residue is abstracted to form a nucleophilic enolate. The steric bulk of the proline-derived auxiliary and the coordination geometry of the nickel atom direct the incoming electrophile, such as allyl bromide or benzyl bromide, to attack from a specific face. This stereocontrol is paramount, as it ensures the formation of the desired diastereomer with high fidelity, thereby preserving the optical integrity of the final alpha-non-natural amino acid product without the need for difficult chiral separations later in the process.
Following the alkylation to form intermediates S4 or R4, the nickel template is efficiently removed through acid hydrolysis using hydrochloric acid in methanol. This step cleaves the Schiff base and releases the nickel ions, yielding the free amino acid S5 or R5. The final step involves the protection of the amino group with 9-fluorenylmethoxycarbonyl (Fmoc) using Fmoc-Osu under mild basic conditions. This sequence is particularly advantageous because the nickel complex can be recycled or disposed of more easily than precious metal catalysts, and the reaction conditions are compatible with a wide range of functional groups. The ability to tune the side chain by simply varying the alkyl halide (R1X or R2X) allows for the rapid generation of diverse libraries of alpha-methyl amino acids, providing R&D teams with the flexibility needed for structure-activity relationship (SAR) studies in drug discovery.
How to Synthesize Alpha-Methyl-Amino Acid Derivatives Efficiently
The practical implementation of this synthesis route involves a carefully orchestrated six-step sequence that balances yield, purity, and operational simplicity. The process begins with the N-alkylation of proline, followed by amidation and complexation to set the stereochemical stage. The critical alkylation step is performed under inert atmosphere to prevent oxidation, ensuring consistent results. Subsequent hydrolysis and protection steps are designed to be robust, utilizing standard workup procedures like extraction and crystallization that are familiar to process chemists. For a detailed breakdown of the specific reaction parameters, stoichiometry, and purification techniques required to replicate this high-yielding pathway, please refer to the standardized synthesis guide provided below.
- Condense L-proline or D-proline with o-chlorobenzyl chloride under alkaline conditions to form the initial proline derivative (S1/R1).
- React the proline derivative with phosphorus trichloride and 2-aminobenzophenone to construct the benzophenone imine intermediate (S2/R2).
- Form the chiral nickel complex by reacting the imine with alanine and nickelous nitrate hexahydrate, followed by alkylation with halohydrocarbons to introduce the side chain.
- Hydrolyze the nickel complex using hydrochloric acid to release the free amino acid, followed by Fmoc protection to yield the final protected product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for critical amino acid intermediates. Traditional methods often rely on bespoke chiral reagents that are sourced from limited suppliers, creating single points of failure and price volatility. By shifting to a process based on commodity chemicals like proline, alanine, and simple alkyl halides, manufacturers can decouple their production from these volatile markets. The elimination of cryogenic steps and noble metal catalysts further reduces the capital expenditure required for reactor infrastructure and waste treatment, leading to significant operational savings that can be passed down the supply chain.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive chiral templates and catalysts with inexpensive, bulk-available starting materials. The avoidance of n-butyllithium and similar pyrophoric reagents eliminates the need for specialized low-temperature reactors and the associated high energy costs for cooling. Furthermore, the simplified purification protocols, which rely on standard recrystallization and chromatography rather than complex distillation or preparative HPLC, reduce solvent consumption and processing time. These factors combine to lower the overall cost of goods sold (COGS), making the final high-purity pharmaceutical intermediates more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of raw materials that are produced on a massive industrial scale globally. Proline and alanine are fermentation products with stable, diversified supply bases, unlike specialized chiral auxiliaries which may have long lead times. The robustness of the reaction conditions, which tolerate moderate temperature fluctuations and do not require ultra-dry or oxygen-free environments for every step, reduces the risk of batch failures due to minor operational deviations. This reliability ensures consistent delivery schedules, allowing downstream drug manufacturers to maintain lean inventory levels without the fear of production stoppages.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route offers distinct advantages for commercial expansion. The reaction solvents, such as methanol, ethanol, and ethyl acetate, are relatively benign and easier to recover and recycle compared to the chlorinated solvents often required in harsher traditional methods. The absence of heavy metal residues like palladium in the final product simplifies the purification burden and ensures compliance with strict regulatory limits for elemental impurities in drug substances. The process is inherently safer, reducing the potential for thermal runaways or hazardous spills, which facilitates easier permitting and faster scale-up from pilot plant to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries regarding the process specifics and quality attributes. These insights are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for process development teams. Understanding these technical nuances is crucial for assessing the fit of this synthetic route within your specific project timelines and quality requirements.
Q: What are the primary advantages of this nickel-complex method over traditional chiral templates?
A: Unlike traditional methods requiring expensive chiral auxiliaries like oxazolidinones or harsh cryogenic conditions with n-butyllithium, this patented process utilizes inexpensive, commercially available raw materials such as proline and alanine. It operates under moderate temperatures (40-60°C) and avoids noble metal catalysts, significantly simplifying the supply chain and reducing production costs while maintaining high optical purity.
Q: What is the expected overall yield and optical purity of the final Fmoc-protected amino acids?
A: The six-step process described in patent CN101565390A achieves an overall yield ranging from 15% to 50%, depending on the specific alkyl group introduced. The method is specifically designed to produce derivatives with extremely high optical activity, ensuring the stereochemical integrity required for sensitive peptide drug applications without the need for complex resolution steps.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for scalability. The reaction conditions are mild, avoiding dangerous reagents like n-butyllithium, and utilize common solvents like methanol, ethanol, and dichloromethane. The use of robust nickel complexes and standard purification techniques like recrystallization and column chromatography makes the transition from laboratory scale to multi-ton commercial production feasible and safe.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Methyl-Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality non-natural amino acids play in the advancement of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering Alpha-Methyl-Amino Acid derivatives with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify identity, potency, and chiral integrity. Our facility is equipped to handle the specific requirements of this nickel-complex chemistry, including inert atmosphere reactors and specialized filtration systems for metal removal.
We invite you to collaborate with us to leverage this innovative synthesis technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to discuss your needs,索取 specific COA data for our existing inventory, or initiate a dialogue regarding route feasibility assessments for custom derivatives. Let us help you secure a stable, cost-effective supply of these essential building blocks for your pharmaceutical pipeline.
