Advanced Electrochemical Synthesis of Alpha-Methyl Ketones for Commercial Pharmaceutical Production
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to construct complex molecular architectures, particularly for active pharmaceutical ingredients (APIs) and their precursors. A groundbreaking development in this arena is detailed in patent CN111218695B, which discloses a novel method for realizing the methylation reaction at the alpha position of ketones under electrochemical conditions. This technology represents a paradigm shift from traditional stoichiometric or transition-metal-catalyzed processes to a sustainable electrochemical dehydrogenation coupling strategy. By utilizing methanol not merely as a solvent but as a direct C-1 source for methylation, this invention addresses critical pain points in modern synthetic chemistry, including atom economy, environmental impact, and operational simplicity. For R&D directors and process chemists, this patent offers a robust alternative for functionalizing ketone compounds, which are ubiquitous scaffolds in medicinal chemistry, ranging from antispasmodics like Eperisone to mucolytic agents. The ability to perform these transformations under mild electrochemical oxidation conditions without the need for external chemical oxidants marks a significant advancement in green manufacturing capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the methylation of the alpha position of ketones has relied heavily on the use of various transition metal catalysts, often involving complex ligand systems such as heterocyclic carbene metal complexes. These conventional routes typically necessitate harsh reaction conditions, including high temperatures and the use of strong, often hazardous, chemical oxidants to drive the transformation. From a supply chain and safety perspective, the reliance on precious metals like palladium, ruthenium, or rhodium introduces significant volatility in raw material costs and creates stringent downstream processing requirements. The removal of trace metal residues to meet pharmaceutical purity standards (often in the parts-per-million range) requires additional purification steps, such as scavenging treatments or repeated recrystallizations, which inevitably lower the overall yield and increase the cost of goods sold (COGS). Furthermore, the generation of stoichiometric amounts of metal-containing waste poses severe environmental compliance challenges, making these traditional methods increasingly unsustainable for large-scale commercial production in a regulated environment.
The Novel Approach
In stark contrast, the methodology described in CN111218695B leverages electrochemical energy to drive the dehydrogenation coupling between ketones and methanol. This approach fundamentally alters the reaction landscape by replacing chemical oxidants with electrons, thereby generating hydrogen gas or water as the only byproducts depending on the specific cell configuration. The reaction operates under remarkably mild conditions, typically between 60-75°C, using a reticulated vitreous carbon (RVC) anode and a platinum cathode under a constant current. This setup allows for precise control over the oxidation potential, minimizing side reactions and over-oxidation issues common in chemical methods. By employing methanol as both the reaction medium and the methylating agent, the process achieves exceptional atom economy. The absence of transition metal catalysts means the resulting products are inherently free from heavy metal contamination, simplifying the purification workflow and ensuring a cleaner impurity profile. This electrochemical strategy not only aligns with the principles of green chemistry but also offers a streamlined, cost-effective pathway for the synthesis of valuable alpha-methyl ketone intermediates.
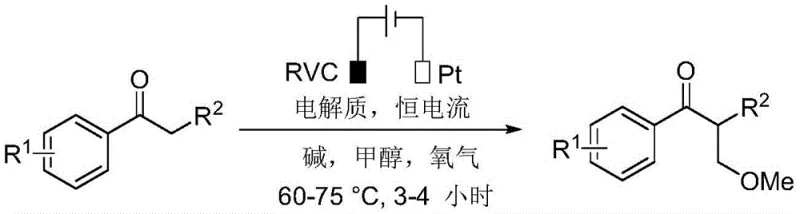
Mechanistic Insights into Electrochemical Dehydrogenation Coupling
The core of this innovation lies in the electrochemical dehydrogenation coupling mechanism, which facilitates the direct functionalization of the ketone alpha-carbon. In this system, the anodic oxidation plays a pivotal role in activating the methanol solvent. Under the applied constant current (typically 10mA in laboratory settings), methanol undergoes oxidation at the RVC anode surface to generate reactive formaldehyde equivalents or methoxy radicals in situ. Simultaneously, the ketone substrate, activated by the alkaline conditions provided by bases such as cesium carbonate or potassium carbonate, forms an enolate species. The electrochemical environment promotes the coupling of these activated species, effectively inserting a methyl group derived from the methanol solvent into the alpha position of the ketone. The presence of an oxygen atmosphere further supports the oxidative cycle, potentially regenerating active species at the electrode interface and ensuring the reaction proceeds to completion with high efficiency. This mechanism avoids the formation of unstable intermediates often seen in thermal aldol-type condensations, providing a more controlled and selective pathway for C-C bond formation.
From an impurity control perspective, this electrochemical method offers distinct advantages over traditional catalytic cycles. Since no exogenous transition metals are introduced, the risk of metal-catalyzed side reactions, such as homocoupling or over-reduction, is significantly mitigated. The primary impurities are likely to be unreacted starting materials or simple over-alkylation products, which are generally easier to separate via standard silica gel chromatography or crystallization compared to metal-ligand complexes. The use of a constant current ensures a steady rate of electron transfer, preventing local hotspots of high oxidation potential that could lead to substrate degradation. This results in a cleaner reaction mixture and higher isolated yields, as evidenced by the patent data showing yields ranging from 68% to 90% across a diverse array of substrates. For quality assurance teams, this translates to a more predictable and robust manufacturing process with reduced batch-to-batch variability.
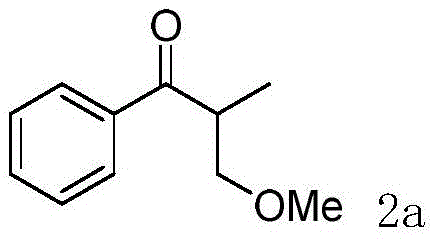
How to Synthesize Alpha-Methyl Ketones Efficiently
The practical implementation of this electrochemical methylation strategy is designed for ease of operation, making it accessible for both laboratory optimization and pilot-scale production. The standard protocol involves charging a three-neck round-bottom flask with the ketone substrate, a suitable base, and a supporting electrolyte such as tetrabutylammonium tetrafluoroborate. The reactor is equipped with the specific electrode configuration—an RVC anode and a platinum cathode—and maintained under an oxygen balloon to facilitate the oxidative cycle. Methanol is added as the solvent, and the mixture is heated to 60-75°C while applying a constant current of 10mA. Reaction progress is monitored via thin-layer chromatography (TLC), with typical reaction times ranging from 3 to 4 hours. Upon completion, the mixture is quenched with water and extracted with ethyl acetate, followed by standard drying and concentration steps. The crude product is then purified by silica gel column chromatography. While the general procedure is straightforward, precise control of current density and electrode surface area is critical for maximizing yield and selectivity. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Prepare the electrochemical cell by installing a reticulated vitreous carbon (RVC) anode and a platinum plate cathode in a three-neck flask equipped with a condenser.
- Charge the reactor with the ketone substrate, a base such as cesium carbonate, and a supporting electrolyte like tetrabutylammonium tetrafluoroborate in methanol solvent.
- Apply a constant current of 10mA under an oxygen atmosphere at 60-75°C until TLC indicates complete consumption of the starting material, then proceed to aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology presents a compelling value proposition centered on cost stability and operational resilience. The most significant economic driver is the elimination of expensive transition metal catalysts. In traditional synthesis, the cost of palladium or rhodium catalysts, along with the specialized ligands required to stabilize them, can constitute a substantial portion of the raw material budget. By removing these components entirely, the direct material costs are drastically reduced. Furthermore, the downstream processing costs associated with metal scavenging and validation testing for residual metals are effectively nullified. This simplification of the purification train not only saves money but also shortens the overall production cycle time, allowing for faster throughput and improved asset utilization. The reliance on commodity chemicals like methanol and common inorganic bases ensures a stable and secure supply chain, insulating the manufacturing process from the geopolitical and market volatilities often associated with rare earth metals and specialty reagents.
- Cost Reduction in Manufacturing: The economic benefits extend beyond simple raw material substitution. By utilizing methanol as both the solvent and the reagent, the process achieves a high degree of atom economy, meaning less waste is generated per unit of product. This reduction in waste volume directly lowers the costs associated with waste disposal and environmental compliance, which are increasingly significant factors in chemical manufacturing. Additionally, the mild reaction conditions (60-75°C) reduce energy consumption compared to high-temperature thermal processes. The absence of complex catalyst recovery systems further simplifies the plant infrastructure requirements, leading to lower capital expenditure (CAPEX) for new facilities or retrofits. Collectively, these factors contribute to a leaner, more cost-efficient manufacturing model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical production, where interruptions can have cascading effects on drug availability. This electrochemical method relies on widely available, bulk commodity chemicals that are produced globally in massive quantities. Methanol, potassium carbonate, and standard electrolytes are not subject to the same supply constraints as specialized organometallic catalysts. This ubiquity ensures that production schedules can be maintained even during periods of market disruption. Moreover, the simplicity of the reaction setup—requiring only a power supply and standard glassware or electrochemical flow cells—means that the technology can be easily deployed across multiple manufacturing sites. This flexibility allows for distributed manufacturing strategies, reducing the risk associated with single-source dependencies and enhancing the overall resilience of the supply network against logistical bottlenecks.
- Scalability and Environmental Compliance: Scaling electrochemical processes is a well-understood engineering challenge with established solutions, such as parallel plate reactors or flow cells, which allow for precise control of mass transfer and current distribution. The linear relationship between current and reaction rate in electrochemistry makes scale-up predictable and manageable, unlike some batch chemical reactions where heat and mass transfer limitations can cause yield drops at larger scales. From an environmental standpoint, the process aligns perfectly with modern sustainability goals. It avoids the use of toxic alkylating agents like methyl iodide and eliminates heavy metal waste streams. This "green" profile simplifies the regulatory approval process for new drug applications (NDAs) and reduces the environmental footprint of the manufacturing site. Companies adopting this technology can leverage these sustainability credentials to meet corporate social responsibility (CSR) targets and appeal to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical methylation technology. These insights are derived directly from the experimental data and technical specifications provided in patent CN111218695B, offering a clear understanding of the process capabilities and limitations. Understanding these details is crucial for project managers evaluating the feasibility of integrating this method into existing production lines or for R&D teams designing new synthetic routes for complex intermediates.
Q: What are the primary advantages of this electrochemical method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of expensive and toxic transition metal catalysts. Traditional methods often require palladium, ruthenium, or rhodium complexes which leave difficult-to-remove metal residues in the final API. This electrochemical approach uses electricity as the clean reagent, ensuring high purity profiles essential for pharmaceutical regulatory compliance.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable. The use of constant current electrolysis with robust electrode materials like RVC and Platinum allows for straightforward translation from laboratory milligram scales to kilogram and tonne-scale flow chemistry reactors. The mild reaction temperatures (60-75°C) and use of commodity chemicals like methanol further support large-scale manufacturing feasibility.
Q: How does the use of methanol as both solvent and reagent impact cost efficiency?
A: Using methanol as both the solvent and the C-1 methyl source drastically simplifies the supply chain and reduces raw material costs. It eliminates the need for specialized, hazardous methylating agents like methyl iodide or dimethyl sulfate. This dual role enhances atom economy and reduces the volume of organic waste generated during the synthesis, leading to substantial operational cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Methyl Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111218695B can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We understand that the transition to metal-free synthesis is not just a technical upgrade but a strategic move towards sustainability and cost efficiency, and we are committed to supporting our partners through every stage of this evolution.
We invite procurement leaders and technical directors to engage with us to explore how this electrochemical methylation technology can optimize your specific supply chain. By leveraging our expertise, you can achieve significant operational improvements without compromising on quality or compliance. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecules. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can drive value for your organization and secure your supply of critical alpha-methyl ketone intermediates.
