Advanced Green Synthesis of Imidazole Ethanol: A Technical Breakthrough for Antifungal API Intermediates
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing critical active pharmaceutical ingredient (API) intermediates. A significant advancement in this domain is detailed in patent CN109111402B, which discloses a novel preparation method for imidazole ethanol, chemically known as alpha-(2,4-Dichlorophenyl)-1H-imidazole-1-ethanol. This compound serves as a pivotal building block for the synthesis of widely used imidazole antifungal medications such as econazole and miconazole. The disclosed technology represents a paradigm shift from traditional liquid-base catalysis to a heterogeneous solid-base system utilizing potassium hydroxide supported on alumina (KOH/Al2O3). By employing hexafluoroisopropanol (HFIP) as a specialized solvent, this process achieves mild reaction conditions, shortened reaction times, and, crucially, the ability to recycle both the catalyst and unreacted raw materials. For R&D directors and procurement managers alike, this innovation addresses the dual challenges of maintaining high product quality while adhering to increasingly stringent environmental regulations regarding solvent waste and heavy metal contamination.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of imidazole ethanol has been plagued by significant operational and environmental drawbacks that hinder large-scale commercial viability. Traditional routes often relied on the reaction of 2,4-dichlorophenyl epoxyethane with imidazole under alkaline conditions, a process notorious for generating substantial three-waste pollution. Alternative methods utilized phase transfer catalysts such as tetrabutylammonium bromide (TBAB) in conjunction with solvents like tetrahydrofuran (THF) or acetone. However, THF presents severe safety hazards due to its tendency to form explosive peroxides upon storage, posing risks of chronic poisoning and equipment damage. Furthermore, other common protocols employed N,N-Dimethylformamide (DMF) as a solvent with sodium hydroxide. DMF is problematic because it is unstable in the presence of strong bases at elevated temperatures, readily hydrolyzing into formic acid and dimethylamine, which complicates purification and degrades product quality. Additionally, the use of liquid alkali solutions necessitates complex aqueous workups, leading to equipment corrosion and difficult wastewater treatment, while the phase transfer catalysts used to enhance yield are often expensive and cannot be effectively recycled, thereby inflating production costs and environmental footprints.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes a supported reagent system that fundamentally alters the reaction landscape. By employing KOH/Al2O3 as a solid base catalyst, the process operates under anhydrous conditions, effectively eliminating the corrosion and wastewater issues associated with aqueous alkali systems. The use of HFIP as a solvent is particularly transformative; it is a high-polarity solvent with low viscosity and exceptional thermal stability, allowing for efficient mixing and heat transfer without the degradation issues seen with DMF. The reaction proceeds smoothly at mild temperatures between 45-50°C, significantly reducing energy consumption and minimizing the formation of thermal byproducts. Perhaps most importantly for supply chain sustainability, the solid nature of the catalyst allows for simple filtration to separate it from the reaction mixture, enabling its regeneration and reuse. Similarly, the solvent HFIP can be easily recovered via distillation, creating a closed-loop system that drastically reduces raw material consumption and waste disposal costs.
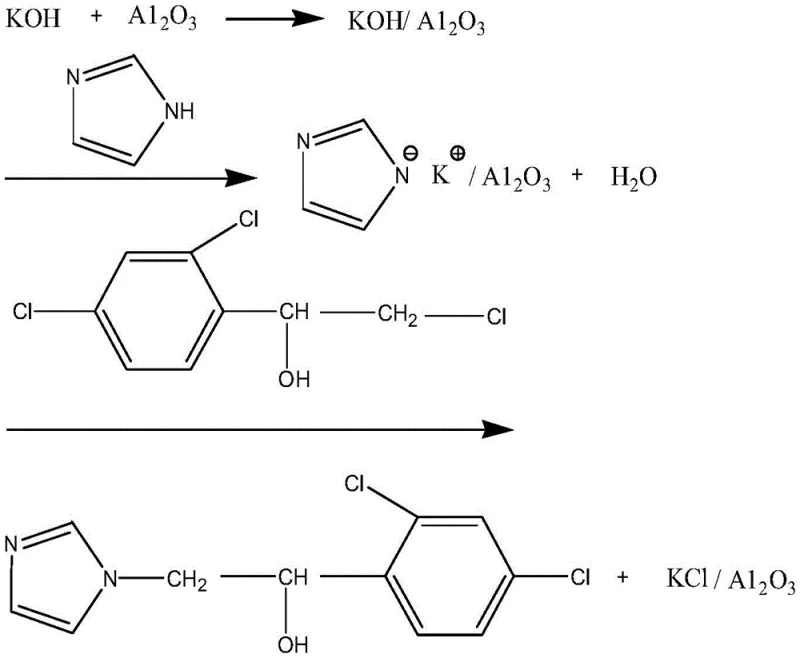
Mechanistic Insights into KOH/Al2O3 Catalyzed N-Alkylation
The efficacy of this synthesis route lies in the unique surface chemistry of the supported catalyst. The preparation involves impregnating neutral alumina (Al2O3) with potassium hydroxide, creating a heterogeneous basic material with high Hammett constant and mechanical strength. When introduced into the HFIP solvent, the KOH/Al2O3 acts as a potent base capable of abstracting the acidic proton from the N-1 position of the imidazole molecule. This deprotonation step generates a highly reactive imidazolyl anion species which remains adsorbed on the surface of the alumina support. This surface confinement is critical as it increases the local concentration of the nucleophile and stabilizes the anionic intermediate, preventing unwanted side reactions that typically occur in homogeneous solution. The alumina support itself possesses both acidic and basic active sites, which may further facilitate the orientation of the reactants, ensuring high regioselectivity for the N-1 alkylation over potential C-alkylation or O-alkylation pathways.
Following the formation of the surface-bound imidazolyl anion, the nucleophilic attack on the electrophilic carbon of 1-(2,4-dichlorophenyl)-2-chloro-ethanol proceeds efficiently. The chloride leaving group is displaced, forming the desired C-N bond and resulting in the production of imidazole ethanol. A key advantage of this mechanism is the generation of potassium chloride (KCl) which remains associated with the alumina support or precipitates out, rather than dissolving into a complex aqueous phase. This simplifies the downstream purification process immensely. The reaction byproduct, water, is generated in situ during the initial deprotonation but is managed effectively within the anhydrous HFIP system, preventing the hydrolysis of the chloro-ethanol substrate. The overall mechanism ensures high selectivity and yield, as evidenced by the consistent melting points of the product across multiple batches, indicating a robust and reproducible chemical transformation that minimizes impurity profiles.
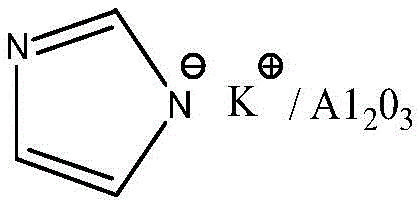
How to Synthesize Imidazole Ethanol Efficiently
The operational procedure for this synthesis is designed for scalability and ease of execution in a standard chemical plant environment. The process begins with the preparation of the catalyst, followed by the main alkylation reaction in HFIP, and concludes with a streamlined workup involving filtration and recrystallization. The detailed standardized synthesis steps, including specific mass ratios, temperature controls, and vacuum distillation parameters required for GMP-compliant manufacturing, are outlined below.
- Preparation of the solid base catalyst by loading KOH onto alumina (Al2O3) followed by drying.
- N-alkylation reaction of imidazole and 1-(2,4-dichlorophenyl)-2-chloro-ethanol in HFIP solvent at 45-50°C.
- Workup involving filtration, solvent recovery, neutralization, and recrystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology offers tangible economic and logistical benefits that extend beyond simple yield improvements. The elimination of hazardous solvents like THF and unstable solvents like DMF removes significant regulatory burdens and safety compliance costs associated with storage and disposal. The ability to recover and reuse the expensive HFIP solvent multiple times without loss of performance translates directly into substantial cost savings on raw materials. Furthermore, the heterogeneous nature of the catalyst means that filtration replaces complex extraction and washing steps, reducing processing time and labor costs. This efficiency gain allows for faster batch turnover times, enhancing the overall responsiveness of the supply chain to market demands for antifungal intermediates.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the recyclability of key inputs. Unlike traditional methods where phase transfer catalysts are consumed and lost in the aqueous waste stream, the KOH/Al2O3 catalyst can be regenerated by simple washing and drying, allowing for multiple cycles of use. This drastically reduces the cost per kilogram of the catalyst. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the need for extensive chromatographic purification or multiple recrystallizations, which are often the most expensive steps in API intermediate manufacturing. The avoidance of equipment corrosion also extends the lifespan of reactor vessels and piping, lowering capital expenditure on maintenance and replacement.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the volatility of raw material markets and regulatory restrictions on solvents. By utilizing a robust solid catalyst and a stable solvent system, this method mitigates the risk of production stoppages due to reagent instability or supply shortages of specialized phase transfer agents. The simplified post-treatment process, which avoids the generation of large volumes of saline wastewater, ensures that production is not bottlenecked by wastewater treatment capacity limits. This reliability makes the supplier a more dependable partner for long-term contracts, ensuring consistent delivery of high-purity imidazole ethanol even during periods of high market demand.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new engineering challenges, particularly regarding heat transfer and mixing in viscous or heterogeneous systems. The low viscosity of HFIP combined with the solid catalyst ensures excellent mixing characteristics even in large-scale reactors, facilitating a smooth transition from pilot plant to commercial production. From an environmental perspective, the process aligns with green chemistry principles by reducing waste generation at the source. The absence of heavy metal catalysts and the minimization of organic solvent waste simplify the environmental impact assessment and permitting process, allowing for faster approval of new production lines and ensuring compliance with increasingly strict global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical implications of adopting this technology for industrial manufacturing.
Q: What are the advantages of using KOH/Al2O3 over traditional liquid bases?
A: The supported solid base eliminates the need for aqueous workups and phase transfer catalysts, simplifying separation and allowing for catalyst recycling, which significantly reduces waste generation.
Q: Why is Hexafluoroisopropanol (HFIP) preferred as a solvent in this process?
A: HFIP offers high polarity and low viscosity, facilitating better mixing and reaction kinetics. Crucially, it has excellent thermal stability and can be easily recovered and reused via distillation, unlike DMF which degrades under basic conditions.
Q: How does this method improve the purity of Imidazole Ethanol?
A: By avoiding liquid-liquid heterogeneous catalysis and minimizing side reactions associated with high temperatures or unstable solvents, this method yields a product with a sharp melting point range of 132.0-133.6°C, indicating superior purity compared to older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for key pharmaceutical intermediates like imidazole ethanol. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented process are fully realized in a commercial setting. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of imidazole ethanol meets the exacting standards required for the synthesis of antifungal APIs. We are committed to leveraging advanced catalytic technologies to deliver products that offer both superior quality and competitive pricing.
We invite global partners to collaborate with us to optimize their supply chains for antifungal intermediates. By adopting this green synthesis route, we can jointly achieve significant efficiency gains and cost reductions. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
