Advanced Purification of Etoricin Intermediates for Commercial Scale-Up and High Purity Standards
Advanced Purification of Etoricin Intermediates for Commercial Scale-Up and High Purity Standards
The chemical industry constantly seeks methods to enhance the purity of critical intermediates used in pharmaceuticals and specialty coatings. Patent CN114456088A introduces a groundbreaking approach to purifying Etoricin (ETO), a vital compound serving as a UV absorber and pharmaceutical intermediate. This technology addresses the persistent challenge of insoluble impurities that traditionally plague the Knoevenagel condensation synthesis of Etoricin. By implementing a specialized ethanol-based refinement process, manufacturers can now achieve purity levels exceeding 99.5%, effectively isolating the problematic byproduct 2-cyano-3,3-diphenylacrylamide. For R&D directors and procurement specialists, this represents a significant leap forward in ensuring consistent quality for high-purity pharmaceutical intermediates and reducing the variability often seen in bulk chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Etoricin often suffer from the formation of stubborn byproducts that are difficult to separate using standard crystallization techniques. In conventional processes, the crude product typically contains insoluble solid impurities that co-precipitate with the desired ester, leading to reduced yields and compromised optical properties in final applications like automotive glass or cosmetics. These impurities, often amide derivatives formed through side reactions with ammonia sources, resist removal by simple solvent washing, forcing manufacturers to accept lower purity grades or engage in costly, multi-step chromatographic separations that are not feasible for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the accumulation of these impurities in recycled mother liquors can poison subsequent batches, creating a cycle of diminishing returns that impacts both cost and supply continuity.
The Novel Approach
The patented method revolutionizes this landscape by leveraging the differential solubility of the target compound versus its impurities in specific solvent systems. The core innovation lies in the strategic use of hot ethanol treatment to selectively dissolve the Etoricin while leaving the insoluble 2-cyano-3,3-diphenylacrylamide behind as a filterable solid. This process is integrated into a streamlined workflow where cyclohexane serves as an efficient water-carrying agent during the initial condensation, facilitating high conversion rates. 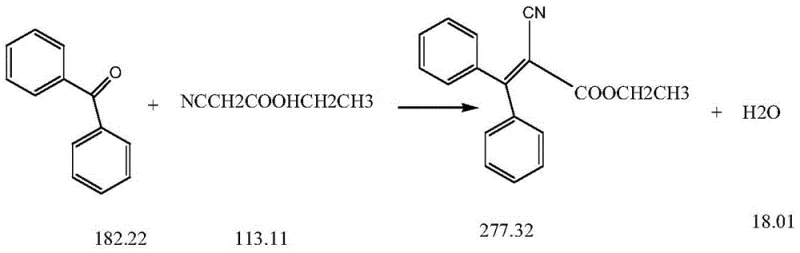 Following the reaction, a sophisticated extraction and recycling protocol allows for the recovery of unreacted raw materials and solvents, drastically minimizing waste. This approach not only simplifies the purification train but also ensures that the final product meets stringent specifications required for reliable agrochemical intermediate and pharma applications without the need for expensive metal catalysts or complex downstream processing.
Following the reaction, a sophisticated extraction and recycling protocol allows for the recovery of unreacted raw materials and solvents, drastically minimizing waste. This approach not only simplifies the purification train but also ensures that the final product meets stringent specifications required for reliable agrochemical intermediate and pharma applications without the need for expensive metal catalysts or complex downstream processing.
Mechanistic Insights into Impurity Formation and Control
Understanding the genesis of impurities is crucial for R&D teams aiming to optimize reaction parameters. Detailed structural analysis using NMR and Mass Spectrometry has confirmed that the primary contaminant is 2-cyano-3,3-diphenylacrylamide, with a molecular weight of 248.29 and a melting point of approximately 240.5°C. 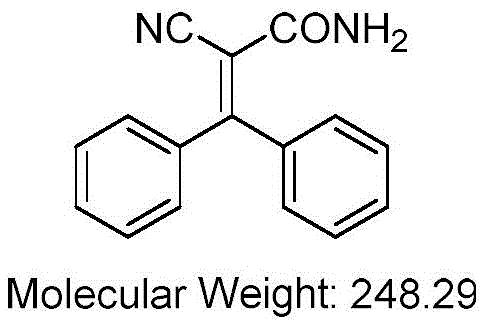 This species arises primarily through the ammonolysis of the ethyl ester intermediate or the direct reaction of raw materials with ammonia generated in situ from ammonium acetate decomposition. The mechanism involves the nucleophilic attack of ammonia on the ester carbonyl, converting the ethoxy group into an amide functionality which drastically alters the solubility profile of the molecule. By identifying this pathway, process chemists can better control the concentration of free ammonia and adjust the acidity of the reaction medium to suppress this side reaction, thereby enhancing the overall efficiency of cost reduction in electronic chemical manufacturing and related sectors.
This species arises primarily through the ammonolysis of the ethyl ester intermediate or the direct reaction of raw materials with ammonia generated in situ from ammonium acetate decomposition. The mechanism involves the nucleophilic attack of ammonia on the ester carbonyl, converting the ethoxy group into an amide functionality which drastically alters the solubility profile of the molecule. By identifying this pathway, process chemists can better control the concentration of free ammonia and adjust the acidity of the reaction medium to suppress this side reaction, thereby enhancing the overall efficiency of cost reduction in electronic chemical manufacturing and related sectors.
Furthermore, the patent elucidates two distinct pathways for this impurity generation, providing a comprehensive map for mitigation. One source is the direct ammonolysis of the synthesized Etoricin ester, while another potential source involves the ammonolysis of the starting ethyl cyanoacetate prior to condensation.  This dual-source understanding allows for precise tuning of the addition order and temperature profiles during the synthesis. For instance, maintaining strict temperature controls below 10°C during the crystallization phase helps prevent the thermal degradation that might accelerate amide formation. Such mechanistic clarity is invaluable for scaling up processes where heat transfer limitations can otherwise lead to hot spots and increased impurity loads, ensuring that the commercial scale-up of complex polymer additives or UV stabilizers remains robust and predictable.
This dual-source understanding allows for precise tuning of the addition order and temperature profiles during the synthesis. For instance, maintaining strict temperature controls below 10°C during the crystallization phase helps prevent the thermal degradation that might accelerate amide formation. Such mechanistic clarity is invaluable for scaling up processes where heat transfer limitations can otherwise lead to hot spots and increased impurity loads, ensuring that the commercial scale-up of complex polymer additives or UV stabilizers remains robust and predictable.
How to Synthesize Etoricin Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-quality Etoricin suitable for sensitive applications. It begins with the precise charging of ethyl cyanoacetate, glacial acetic acid, ammonium acetate, benzophenone, and cyclohexane into a reactor equipped for reflux and water separation. The detailed standardized synthesis steps involve careful monitoring of the water separation rate to drive the equilibrium forward, followed by a controlled cooling and freezing sequence to maximize crystal yield. For the complete operational procedure, including specific stirring rates, vacuum levels for solvent recovery, and the exact number of ethanol washing cycles required to achieve >99.5% purity, please refer to the step-by-step guide below which encapsulates the critical process parameters for successful implementation.
- Conduct the condensation reaction between benzophenone and ethyl cyanoacetate using glacial acetic acid and ammonium acetate catalysts with cyclohexane as a water carrier.
- Perform extraction and separation using cyclohexane to isolate the crude product, followed by solvent recovery and recycling of mother liquors.
- Purify the crude solid by multiple hot ethanol washes to remove insoluble 2-cyano-3,3-diphenylacrylamide impurities, yielding high-purity Etoricin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates directly into enhanced operational efficiency and risk mitigation. The ability to recycle mother liquors up to three times without significant loss of quality means a substantial reduction in raw material consumption per kilogram of finished product. This closed-loop system minimizes the dependency on volatile raw material markets and reduces the volume of hazardous waste requiring disposal, aligning with increasingly strict environmental regulations. Moreover, the simplified purification train, which avoids complex chromatography or exotic reagents, lowers the barrier for manufacturing entry and ensures a more stable supply of high-purity OLED material precursors or pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps and the ability to recover and reuse solvents and unreacted starting materials significantly lowers the overall cost of goods sold. By avoiding the need for transition metal catalysts which often require costly removal steps to meet ppm-level specifications, the process achieves cost optimization through chemical simplicity. The high yield obtained from the optimized crystallization and washing protocol ensures that less feedstock is wasted, directly improving the margin profile for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against impurity accumulation ensures consistent batch-to-batch quality, which is critical for long-term supply contracts in the pharmaceutical sector. The use of common, commercially available solvents like ethanol and cyclohexane mitigates the risk of supply disruptions associated with specialized reagents. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream customers receive materials that consistently meet their rigorous quality standards.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as reflux, filtration, and distillation that are easily transferred from pilot plant to multi-ton production facilities. The reduction in waste generation through solvent recycling and the avoidance of heavy metals contribute to a greener manufacturing footprint, facilitating easier regulatory approval in environmentally sensitive markets. This alignment with green chemistry principles not only reduces compliance costs but also enhances the brand value of the final product in markets that prioritize sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity on how this method resolves traditional pain points in Etoricin production. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing lines for improved efficiency and product quality.
Q: What is the primary impurity found in Etoricin synthesis?
A: The primary impurity identified is 2-cyano-3,3-diphenylacrylamide, formed via the ammonolysis of the ester intermediate or raw materials during the reaction process.
Q: How does the new purification method improve yield?
A: By utilizing hot ethanol washing to selectively dissolve the product while leaving the insoluble amide impurity behind, the method significantly increases the single-pass conversion rate and overall yield.
Q: Can the mother liquor be recycled in this process?
A: Yes, the patent describes a robust recycling protocol where mother liquors and separated lower layers are reused up to three times, minimizing waste and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etoricin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of advanced chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN114456088A are fully realized in practical manufacturing environments. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Etoricin meets the exacting demands of the global pharmaceutical and specialty chemical industries.
We invite you to collaborate with us to leverage this advanced purification technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for reliable fine chemical intermediates and sustainable manufacturing solutions.
