Scalable Synthesis of Potent Acetylcholinesterase Inhibitors for Neurodegenerative Therapy
Scalable Synthesis of Potent Acetylcholinesterase Inhibitors for Neurodegenerative Therapy
The escalating global burden of neurodegenerative disorders, particularly Alzheimer's Disease (AD), has intensified the search for novel therapeutic agents capable of halting cognitive decline. Patent CN107382990B discloses a significant advancement in this field by presenting a robust synthetic pathway for 5-(1H-indole-3-methyl)-N-(2-diisopropylaminoethyl)-1,2,4-dioxazole-3-carboxamide, a compound exhibiting potent acetylcholinesterase (AChE) inhibitory properties. This specific molecular architecture, characterized by a 1,2,4-oxadiazole structural fragment linked to an indole core, represents a strategic modification of marine natural products like phidianidine B, aiming to enhance bioavailability and synthetic accessibility. For pharmaceutical developers and procurement specialists, understanding the nuances of this patented methodology is crucial, as it offers a viable route to high-purity intermediates essential for next-generation AD medications. The structural complexity of the target molecule, as illustrated below, demands precise control over reaction conditions to ensure the integrity of the heterocyclic systems.
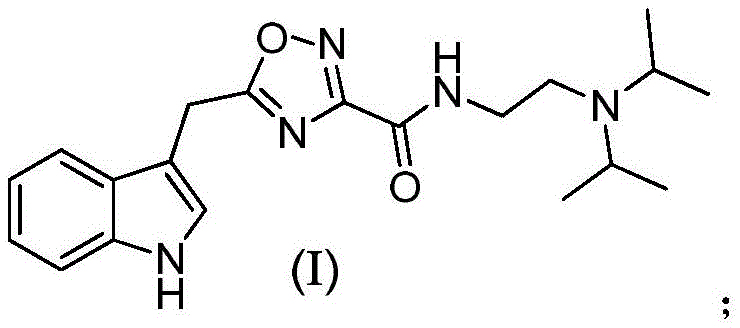
From a commercial perspective, the ability to reliably produce this pharmaceutical intermediate at scale addresses a critical gap in the supply chain for neurology-focused drug discovery programs. The patent outlines a streamlined approach that bypasses the limitations of extracting complex alkaloids from natural sources, which are often plagued by low yields and seasonal variability. By shifting to a fully synthetic route, manufacturers can achieve consistent quality and significantly reduced lead times, which are paramount metrics for supply chain heads managing just-in-time inventory for clinical trials. Furthermore, the detailed reaction conditions provided in the patent allow for rigorous process optimization, ensuring that the final active pharmaceutical ingredient (API) precursors meet the stringent purity specifications required by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of AChE inhibitors has relied heavily on the isolation and semi-synthesis of natural products or the use of multi-step routes involving harsh reagents that compromise atom economy. Traditional methods for constructing the 1,2,4-oxadiazole core often necessitate high-temperature cyclizations or the use of hazardous nitrile oxides, which pose significant safety risks and environmental burdens in a manufacturing setting. Moreover, earlier approaches to functionalizing the indole scaffold frequently resulted in poor regioselectivity, leading to difficult-to-separate impurities that drastically increase downstream purification costs. For procurement managers, these inefficiencies translate into volatile pricing and unreliable delivery schedules, as batch failures become more common when scaling up sensitive natural product extractions. The reliance on scarce marine resources for lead compounds like phidianidine B further exacerbates supply chain fragility, making it nearly impossible to secure the tonnage quantities needed for late-stage clinical development without exorbitant costs.
The Novel Approach
In stark contrast, the methodology described in CN107382990B introduces a modular, three-step synthetic strategy that leverages widely available commodity chemicals to construct the target pharmacophore with high efficiency. The process initiates with the condensation of indole-3-acetic acid and an amino-hydroxyimino ester, utilizing modern peptide coupling technology to drive the formation of the oxadiazole ring under mild conditions. This innovative route eliminates the need for extreme thermal inputs during the critical ring-closing step, thereby preserving the sensitive indole nitrogen and preventing decomposition pathways that typically plague conventional syntheses. As depicted in the comprehensive reaction scheme below, the progression from the initial ester intermediate to the final amide is logical and chemically sound, facilitating easy monitoring via TLC or HPLC at each stage.
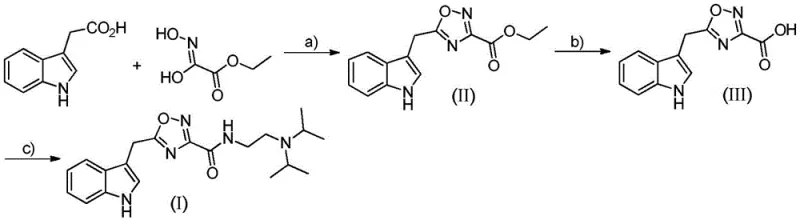
This novel approach not only simplifies the operational workflow but also enhances the overall sustainability profile of the manufacturing process by reducing solvent consumption and waste generation. For a reliable pharmaceutical intermediate supplier, adopting this route means offering clients a product with a superior impurity profile, as the selective nature of the coupling reagents minimizes side reactions. The final amidation step, which introduces the diisopropylaminoethyl side chain crucial for blood-brain barrier penetration, is executed with high conversion rates, ensuring that the costly upstream intermediates are not lost to incomplete reactions. Consequently, this method represents a paradigm shift towards greener, more cost-effective production of complex heterocyclic drugs, aligning perfectly with the industry's push for sustainable chemistry practices.
Mechanistic Insights into Amide Coupling and Oxadiazole Cyclization
The cornerstone of this synthesis lies in the sophisticated interplay between carbodiimide-mediated activation and nucleophilic attack, which drives the formation of the 1,2,4-oxadiazole ring in the first step. Mechanistically, the carboxylic acid of indole-3-acetic acid is activated by reagents such as HATU or DIC to form a highly reactive O-acylisourea or active ester intermediate. This activated species is then susceptible to nucleophilic attack by the hydroxylamine moiety of the ethyl (Z)-2-amino-2-hydroxyimino)acetate. The subsequent intramolecular cyclization involves the dehydration of the resulting hydroxamic acid derivative, a process that is thermodynamically favored under the specified reflux conditions in ethanol. Understanding this mechanism is vital for R&D directors, as it highlights the importance of water removal to drive the equilibrium towards the desired oxadiazole product, preventing the hydrolysis of the activated intermediate back to the starting acid.
Furthermore, the control of impurities is inherently built into the choice of reagents and the sequential nature of the synthesis. The use of specific bases like sodium acetate in the cyclization step acts as a mild catalyst that promotes ring closure without inducing racemization or degradation of the chiral centers, although this specific molecule is achiral. In the final amidation step, the activation of the carboxylic acid (Formula III) ensures that the bulky N,N-diisopropylethylenediamine can effectively attack the carbonyl carbon despite steric hindrance. This precise control over reactivity prevents the formation of symmetric anhydrides or N-acylurea byproducts, which are common pitfalls in amide bond formation. By maintaining strict stoichiometric control and utilizing high-purity coupling agents, the process ensures that the final high-purity pharmaceutical intermediate meets the rigorous standards required for preclinical toxicology studies.
How to Synthesize 5-(1H-indole-3-methyl)-N-(2-diisopropylaminoethyl)-1,2,4-dioxazole-3-carboxamide Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature and solvent choice, to maximize yield and minimize byproduct formation. The patent details a robust protocol that begins with the activation of indole-3-acetic acid in dichloromethane, followed by the addition of the hydroxylamine component and subsequent heating in ethanol to effect cyclization. This two-pot procedure for the first intermediate allows for the isolation of a stable ester, which can be purified if necessary before proceeding to hydrolysis. The hydrolysis step is straightforward, utilizing potassium hydroxide in ethanol to cleave the ethyl ester, followed by careful acidification to precipitate the free acid. Finally, the acid is coupled with the diamine using standard peptide coupling conditions to afford the target molecule. For detailed operational parameters and safety guidelines, refer to the standardized synthesis steps provided below.
- Condense indole-3-acetic acid with ethyl (Z)-2-amino-2-hydroxyimino)acetate using a coupling agent to form the ethyl ester intermediate.
- Hydrolyze the ethyl ester intermediate under basic catalysis followed by acidification to obtain the carboxylic acid derivative.
- Perform amidation of the carboxylic acid with N,N-diisopropylethylenediamine using a condensing agent to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthetic route offers substantial strategic advantages for organizations looking to optimize their supply chain resilience and reduce overall manufacturing expenditures. The primary benefit stems from the utilization of commodity-grade starting materials, such as indole-3-acetic acid, which are produced on a massive industrial scale for the agricultural and fragrance sectors, ensuring a stable and low-cost supply base. Unlike routes dependent on exotic natural extracts, this fully synthetic pathway decouples production from biological variables, allowing for consistent year-round manufacturing regardless of seasonal fluctuations. For procurement managers, this translates into predictable pricing models and the ability to negotiate long-term contracts with confidence, knowing that the raw material risk is negligible. Additionally, the elimination of transition metal catalysts removes the need for expensive scavenging resins and extensive heavy metal testing, further streamlining the quality control budget.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by employing efficient coupling reagents that drive reactions to completion with minimal excess, thereby reducing raw material waste. The avoidance of cryogenic conditions or high-pressure equipment lowers the capital expenditure required for reactor infrastructure, making the process accessible to a wider range of contract manufacturing organizations. Furthermore, the high yields reported in the patent examples suggest that the throughput per batch can be maximized, effectively lowering the cost per kilogram of the final active ingredient. By simplifying the purification steps to standard silica gel chromatography or crystallization, the process avoids the need for preparative HPLC, which is a major cost driver in fine chemical synthesis.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable intermediates allows for the strategic stocking of key precursors, buffering the supply chain against sudden disruptions in logistics or raw material availability. Since the synthesis does not depend on single-source biological extracts, the risk of supply interruption due to environmental factors or geopolitical issues is virtually eliminated. This reliability is critical for pharmaceutical companies managing tight clinical trial timelines, where any delay in API supply can have cascading effects on regulatory filings. The robustness of the chemistry also means that technology transfer between different manufacturing sites can be accomplished with minimal re-optimization, ensuring global supply continuity.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot plant operations without fundamental changes to the chemistry. The use of common organic solvents like dichloromethane, ethanol, and ethyl acetate facilitates solvent recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. Regulatory compliance is simplified as the process avoids genotoxic reagents and persistent organic pollutants, easing the burden on environmental health and safety teams. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet market demand as the drug candidate progresses through clinical phases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this oxadiazole derivative, based on the data provided in the patent documentation. These insights are designed to clarify the feasibility of the synthesis and its potential impact on drug development pipelines. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader portfolio of neurodegenerative disease therapeutics.
Q: What is the biological activity of the synthesized oxadiazole compound?
A: The compound exhibits significant acetylcholinesterase (AChE) inhibitory activity with an IC50 value of 6.62 μM, indicating strong potential for treating Alzheimer's Disease.
Q: Are the raw materials for this synthesis readily available?
A: Yes, the synthesis utilizes commercially accessible starting materials such as indole-3-acetic acid and standard coupling reagents, ensuring supply chain stability.
Q: Does this process require transition metal catalysts?
A: No, the described methodology relies on organic coupling agents like HATU or DIC, eliminating the need for expensive and toxic transition metal removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(1H-indole-3-methyl)-N-(2-diisopropylaminoethyl)-1,2,4-dioxazole-3-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable supply of high-quality intermediates for the development of life-saving Alzheimer's therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project never stalls due to capacity constraints. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-(1H-indole-3-methyl)-N-(2-diisopropylaminoethyl)-1,2,4-dioxazole-3-carboxamide meets your exact requirements. Our commitment to excellence extends beyond mere compliance; we actively collaborate with our partners to optimize processes for maximum efficiency and cost-effectiveness.
We invite you to contact our technical procurement team to discuss your specific needs and request a Customized Cost-Saving Analysis tailored to your project volume. Whether you require specific COA data for regulatory submissions or detailed route feasibility assessments for process validation, our experts are ready to provide the support you need. Partner with us to accelerate your drug development timeline and secure a competitive advantage in the rapidly evolving landscape of neurodegenerative disease treatment.
