Advanced Synthesis of Furan-Fused Borazadihydropyrene for High-Performance OLED Applications
Advanced Synthesis of Furan-Fused Borazadihydropyrene for High-Performance OLED Applications
The rapid evolution of organic optoelectronics demands materials with precisely tuned electronic properties, driving intense research into boron-nitrogen doped polycyclic aromatic hydrocarbons (PAHs). Patent CN113845538A introduces a groundbreaking design and synthesis method for furan-fused borazadihydropyrene molecules, offering a robust pathway to next-generation organic semiconductor devices. This technology leverages a nitrogen-guided approach to generate specific furan-fused boron-aza-dihydropyrene macroconjugated systems, addressing the critical need for stable, high-efficiency emitters in Organic Light Emitting Diodes (OLEDs) and Organic Field Effect Transistors (OFETs). By replacing carbon-carbon bonds with isoelectric boron-nitrogen bonds, manufacturers can access unique photoelectric characteristics, such as amphoteric pi-bonding and stable free radical cation formation, which are essential for advanced display technologies.
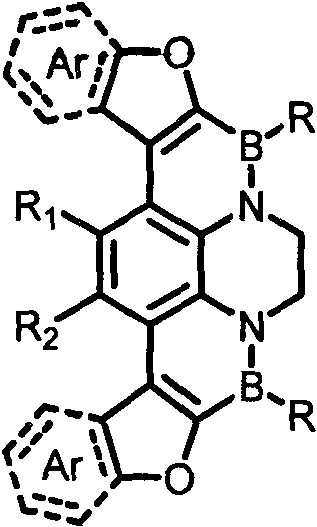
For R&D directors seeking reliable OLED material suppliers, this patent represents a significant leap forward in molecular engineering. The disclosed compounds feature a versatile structural framework where substituents R, R1, R2, and Ar can be independently modified to include alkyl chains, halogens, or complex fused ring systems like pyrene and anthracene. This modularity allows for the fine-tuning of HOMO-LUMO gaps and charge transport properties without compromising the structural integrity of the conjugated backbone. As the industry pushes towards higher resolution and energy-efficient displays, the ability to synthesize these complex heterocycles with high purity and reproducibility becomes a decisive competitive advantage for supply chain leaders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of boron-nitrogen substituted aromatics has been plagued by harsh reaction conditions and limited substrate scope, hindering their commercial adoption in the electronic chemical manufacturing sector. Early methods pioneered by the Dewar group often required extreme temperatures or unstable intermediates, making scale-up hazardous and economically unviable for large-scale production. Furthermore, traditional desulfurization routes or direct borylation strategies frequently suffered from poor regioselectivity, leading to complex mixtures of isomers that were difficult to separate to the stringent purity levels required for semiconductor applications. These legacy processes often relied on stoichiometric amounts of aggressive Lewis acids or sensitive organometallic reagents that degraded rapidly upon exposure to moisture or air, resulting in inconsistent batch quality and increased waste generation.
The Novel Approach
The novel methodology described in the patent overcomes these historical bottlenecks by employing a mild, step-wise synthetic strategy centered on Suzuki coupling and Lewis base-guided electrophilic boron cyclization. This approach utilizes commercially available 3,6-dibromobenzene-1,2-diamine as a robust starting scaffold, ensuring a consistent and cost-effective supply chain foundation. By operating at moderate temperatures ranging from room temperature to 80°C, the process significantly reduces energy consumption and thermal stress on sensitive functional groups, thereby minimizing side reactions and impurity formation. The use of aryl potassium trifluoroborate salts as boron sources offers superior stability compared to traditional boronic acids, enhancing the reliability of the cyclization step and ensuring high yields of the target furan-fused borazadihydropyrene core.
Mechanistic Insights into Electrophilic Boron Cyclization
The core innovation lies in the precise construction of the boron-nitrogen heterocyclic rings through a carefully orchestrated sequence of condensation, reduction, and coupling reactions. Initially, the diamine precursor undergoes condensation with glyoxal followed by reduction with sodium borohydride to establish the central dihydroquinoxaline motif, which serves as the nitrogen donor for subsequent cyclization. The critical step involves the reaction of the furan-substituted intermediate with aryl potassium trifluoroborate in the presence of silicon tetrachloride and triethylamine. In this mechanism, silicon tetrachloride acts as a Lewis acid activator, facilitating the cleavage of the B-C bond and promoting the electrophilic attack on the nitrogen lone pair to close the B-N ring. This intramolecular cyclization is highly efficient due to the pre-organization of the furan and amine groups, driven by the rigid planar geometry of the intermediate.
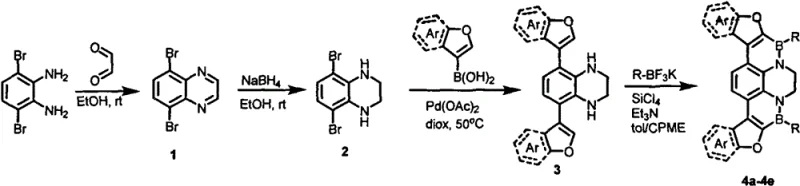
Impurity control is inherently built into this mechanistic pathway through the use of selective palladium-catalyzed Suzuki coupling prior to the cyclization event. By installing the bulky furan-aromatic groups first, the steric environment around the reactive amine sites is optimized, preventing unwanted polymerization or oligomerization during the final boron incorporation step. The reaction conditions, specifically the use of toluene and cyclopentylmethyl ether as solvents, provide an ideal medium for solubilizing the organic intermediates while maintaining the stability of the boron species. This careful balance of reactivity and stability ensures that the final products, such as compounds 4a through 4e, are obtained as high-purity white solids after standard silica gel chromatography, meeting the rigorous specifications demanded by the display and optoelectronic materials industry.
How to Synthesize Furan-Fused Borazadihydropyrene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these advanced materials, emphasizing operational simplicity and reproducibility. The process begins with the preparation of the dihydroquinoxaline core, followed by the installation of the furan arms via palladium catalysis, and concludes with the ring-closing borylation. Each step is designed to maximize yield while minimizing the need for exotic reagents or extreme conditions, making it highly suitable for transfer from laboratory discovery to pilot plant operations. For technical teams looking to implement this chemistry, the detailed procedural notes regarding solvent choices, stoichiometry, and workup procedures offer valuable guidance for optimizing the process further. The standardized nature of the reactions allows for seamless integration into existing fine chemical manufacturing workflows.
- Condense 3,6-dibromobenzene-1,2-diamine with glyoxal followed by reduction with NaBH4 to form the dihydroquinoxaline core.
- Perform Suzuki coupling with arylfuranboronic acid using Pd(OAc)2 catalyst at 50°C to install the furan moieties.
- Execute Lewis base-guided electrophilic boron cyclization using aryl potassium trifluoroborate and SiCl4 at 80°C to close the boron-nitrogen rings.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial opportunities for cost reduction in electronic chemical manufacturing by leveraging widely available commodity chemicals. The reliance on 3,6-dibromobenzene-1,2-diamine and glyoxal as primary feedstocks ensures a stable supply base, mitigating the risks associated with sourcing specialized or monopolized precursors. Additionally, the elimination of cryogenic conditions and the use of ambient pressure reactions simplify the engineering requirements for production vessels, allowing for the utilization of standard glass-lined or stainless steel reactors without the need for specialized low-temperature infrastructure. This operational flexibility translates directly into lower capital expenditure and reduced overhead costs for manufacturing partners.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing aryl potassium trifluoroborate salts, which are generally more stable and easier to handle than volatile boron halides, reducing material loss and safety containment costs. The mild reaction temperatures eliminate the need for extensive heating or cooling utilities, drastically lowering the energy footprint per kilogram of product produced. Furthermore, the high selectivity of the cyclization step minimizes the formation of difficult-to-remove byproducts, reducing the burden on downstream purification units and increasing the overall throughput of the facility.
- Enhanced Supply Chain Reliability: By avoiding the use of air-sensitive reagents that require strict inert atmosphere handling throughout the entire process, the supply chain becomes more resilient to logistical disruptions. The intermediates generated in the early stages, such as the furan-substituted diamines, possess sufficient stability to be isolated and stored if necessary, providing buffer capacity against demand fluctuations. This robustness ensures consistent delivery schedules for downstream device manufacturers who rely on just-in-time inventory models for their production lines.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, as demonstrated by the use of common organic solvents like ethanol, toluene, and dioxane which are easily recovered and recycled in industrial distillation columns. The absence of heavy metal contaminants in the final cyclization step, aside from the trace palladium from the earlier coupling which is removed during workup, simplifies waste treatment and disposal protocols. This alignment with green chemistry principles facilitates regulatory compliance in jurisdictions with strict environmental standards, smoothing the path for global market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these furan-fused borazadihydropyrene derivatives. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their product portfolios. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of this boron-nitrogen doping strategy?
A: The boron-nitrogen doping strategy allows for significant tuning of optical and electronic properties while maintaining the conjugated backbone, creating stable free radical cations suitable for OLED applications.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes mild reaction conditions (50°C to 80°C) and commercially available reagents like 3,6-dibromobenzene-1,2-diamine, facilitating straightforward scale-up from lab to commercial tonnage.
Q: What purification methods are recommended for the final product?
A: The patent specifies silica gel column chromatography using ethyl acetate and petroleum ether eluents to achieve high-purity white solid products suitable for electronic grade applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furan-Fused Borazadihydropyrene Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is essential for securing a competitive edge in the market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to mass manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of furan-fused borazadihydropyrene meets the exacting standards required for OLED and OFET applications. Our commitment to quality assurance means that you can rely on us for consistent material performance in your final devices.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how our capabilities align with your project goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this synthetic route can improve your margins. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a successful and profitable collaboration.
