Revolutionizing Ethyl α-Cyanocinnamate Production with Advanced Nitrogen-Rich Porous Catalysts
Revolutionizing Ethyl α-Cyanocinnamate Production with Advanced Nitrogen-Rich Porous Catalysts
The landscape of fine chemical synthesis is undergoing a significant transformation, driven by the urgent need for sustainable, metal-free, and highly efficient catalytic systems. A pivotal development in this domain is documented in patent CN113149865B, which introduces a novel organic basic catalyst known as mPMF for the synthesis of ethyl α-cyanocinnamate. This compound serves as a critical building block in the pharmaceutical and agrochemical industries, valued for its electron-deficient olefin structure that facilitates diverse downstream functionalization. The patented technology leverages a nitrogen-rich covalent organic porous material derived from melamine and paraformaldehyde, offering a robust alternative to traditional transition-metal catalysts. By operating under mild conditions with exceptional selectivity exceeding 99.9%, this innovation addresses long-standing challenges regarding catalyst recovery, product purity, and environmental compliance. For global procurement and R&D teams, understanding the mechanistic superiority and commercial viability of this route is essential for securing a competitive edge in the supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ethyl α-cyanocinnamate via Knoevenagel condensation has relied heavily on homogeneous catalysts or supported ionic liquids that present significant operational bottlenecks. Traditional methods often utilize amine-functionalized materials or metal-based systems that suffer from poor stability and low activity, necessitating prolonged reaction times that can extend beyond 12 to 16 hours to achieve moderate yields. Furthermore, many existing protocols require harsh conditions, such as high-temperature autoclave reactions at 100°C, which escalate energy costs and introduce safety hazards during scale-up. A critical drawback of homogeneous catalysis is the difficulty in separating the catalyst from the final product, leading to contamination with residual metals or organic bases that compromise the purity profile required for sensitive pharmaceutical applications. These inefficiencies not only inflate manufacturing costs but also generate substantial waste streams, conflicting with modern green chemistry principles and increasing the burden on wastewater treatment facilities.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113149865B utilizes a nitrogen-rich covalent organic porous framework (mPMF) that fundamentally alters the reaction kinetics and thermodynamics. This heterogeneous catalyst operates efficiently at a mild temperature of 60°C in methanol, drastically reducing the thermal energy input required compared to conventional high-temperature processes. The unique porous structure provides a high specific surface area and abundant basic sites that facilitate the rapid deprotonation of ethyl cyanoacetate, driving the condensation with benzaldehyde to completion within just 3 hours. Crucially, the solid nature of the mPMF catalyst allows for simple filtration, enabling seamless catalyst recovery and reuse without the complex extraction steps associated with liquid catalysts. This approach not only accelerates production cycles but also ensures a cleaner product profile, significantly lowering the cost of goods sold (COGS) by minimizing downstream purification requirements and solvent usage.
Mechanistic Insights into mPMF-Catalyzed Knoevenagel Condensation
The exceptional performance of the mPMF catalyst stems from its unique structural architecture, which is engineered to maximize basicity while maintaining chemical stability. The catalyst is synthesized through a solvothermal process involving melamine and paraformaldehyde, resulting in a cross-linked network rich in triazine rings and amino groups. These nitrogen-containing moieties act as strong Lewis base sites that effectively activate the active methylene group of ethyl cyanoacetate. Upon interaction with the catalyst surface, the acidic protons of the cyanoacetate are abstracted, generating a nucleophilic carbanion species that readily attacks the carbonyl carbon of benzaldehyde. This mechanism bypasses the need for soluble strong bases, thereby preventing side reactions such as hydrolysis or polymerization that often plague homogeneous systems. The porous nature of the material ensures that reactants can diffuse freely to the active sites, while the hydrophobic character of the framework helps in repelling water produced during the condensation, shifting the equilibrium towards product formation.
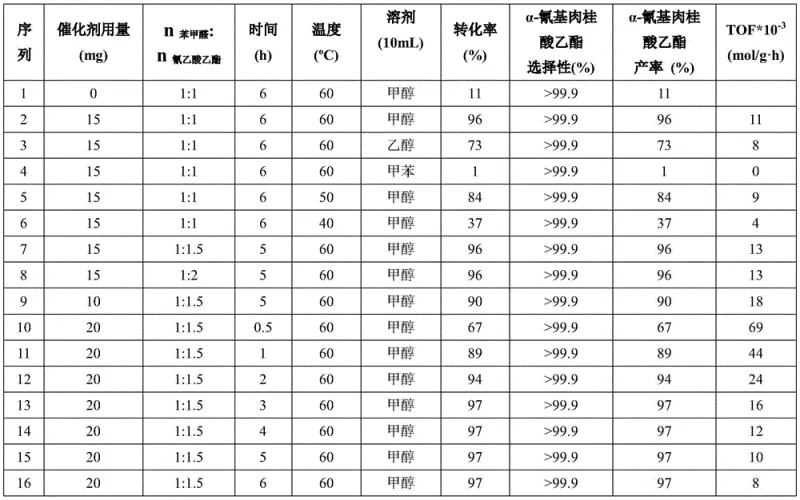
Beyond mere activity, the structural integrity of the mPMF catalyst plays a pivotal role in impurity control and long-term stability. Unlike amorphous polymers that may degrade or leach components into the reaction mixture, the covalent bonding within the mPMF framework ensures that the catalyst remains intact throughout multiple reaction cycles. This stability is crucial for maintaining consistent product quality, as it prevents the introduction of catalyst-derived impurities into the final API intermediate. The high nitrogen content, quantified at approximately 54% by mass, creates a localized alkaline environment that is sufficiently strong to drive the reaction yet mild enough to preserve sensitive functional groups on the substrate. For R&D directors focused on impurity profiles, this means a significantly cleaner crude product with fewer unknown by-products, simplifying the validation process for regulatory filings. The combination of high surface area and defined pore structures allows for precise tuning of the reaction environment, ensuring that the selectivity for the desired E-isomer of ethyl α-cyanocinnamate remains consistently above 99.9%.
How to Synthesize Ethyl α-Cyanocinnamate Efficiently
Implementing this advanced catalytic system requires adherence to specific preparation and reaction protocols to maximize yield and catalyst longevity. The synthesis begins with the careful solvothermal assembly of the catalyst itself, followed by its application in the Knoevenagel condensation under optimized solvent and temperature conditions. The process is designed to be straightforward, utilizing standard laboratory glassware and commonly available reagents, which facilitates easy translation from bench-scale discovery to pilot plant operations. Detailed standard operating procedures regarding the precise stoichiometry, mixing rates, and workup techniques are critical for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Prepare the mPMF catalyst by dispersing melamine and paraformaldehyde in anhydrous DMSO, heating at 120°C, followed by solvothermal treatment at 160°C for 48 hours to form the nitrogen-rich porous structure.
- In a round-bottom flask, combine benzaldehyde and ethyl cyanoacetate in a 1: 1.5 molar ratio with methanol as the solvent and add 20mg of the mPMF catalyst per mmol of substrate.
- Heat the reaction mixture to 60°C with magnetic stirring for 3 hours, then filter the catalyst and analyze the filtrate via gas chromatography to confirm >97% conversion and >99.9% selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the mPMF catalytic technology represents a strategic opportunity to optimize cost structures and enhance supply reliability. The shift from metal-based or complex ionic liquid catalysts to a simple organic polymer derived from commodity chemicals like melamine significantly reduces raw material volatility and cost. Since the catalyst does not contain precious or toxic metals, the supply chain is insulated from the geopolitical and market fluctuations often associated with palladium, platinum, or rare earth elements. Furthermore, the elimination of heavy metals removes the necessity for expensive scavenging resins or complex purification trains, directly translating to substantial cost savings in the manufacturing process. The mild reaction conditions also imply lower energy consumption, contributing to a reduced carbon footprint and aligning with corporate sustainability goals without compromising output efficiency.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplicity of the catalyst synthesis and the elimination of costly downstream processing. Because the mPMF catalyst is heterogeneous, it can be removed via simple filtration, avoiding the need for extensive aqueous workups or chromatographic purification often required to remove homogeneous catalyst residues. This streamlined workflow reduces solvent consumption and labor hours, leading to a significantly lower cost per kilogram of the final intermediate. Additionally, the high conversion rates achieved in shorter timeframes increase reactor throughput, allowing manufacturers to produce more batches within the same timeframe, effectively amortizing fixed operational costs over a larger volume of product.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, non-toxic raw materials that are widely available in the global chemical market. Unlike specialized catalysts that may have single-source suppliers or long lead times, the precursors for mPMF are commodity chemicals with stable supply chains. The stability of the catalyst itself ensures that it can be stored for extended periods without degradation, allowing for strategic stockpiling to buffer against market disruptions. Moreover, the reproducibility of the reaction minimizes the risk of batch failures, ensuring that delivery schedules to downstream pharmaceutical clients are met consistently. This reliability is paramount for maintaining just-in-time inventory systems and avoiding production stoppages in the broader value chain.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the absence of high-pressure or high-temperature requirements, reducing the engineering complexity and capital expenditure needed for reactor design. The metal-free nature of the catalyst simplifies waste management, as the spent catalyst and effluent streams do not require hazardous waste disposal protocols associated with heavy metal contamination. This ease of compliance with environmental regulations accelerates the permitting process for new production lines and reduces the liability associated with industrial waste. The ability to operate safely at atmospheric pressure and moderate temperatures further enhances the safety profile of the facility, lowering insurance premiums and operational risks associated with high-energy chemical processes.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology, we have compiled answers to common inquiries regarding the catalyst's performance, stability, and application scope. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industrial implementation. Understanding these nuances is vital for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the advantages of the mPMF catalyst over traditional homogeneous catalysts?
A: Unlike homogeneous catalysts which are difficult to separate and often contain toxic metals, the mPMF catalyst is a heterogeneous, metal-free organic polymer. It offers excellent stability, can be easily filtered from the reaction mixture, and achieves high selectivity (>99.9%) without requiring complex downstream purification processes to remove heavy metal residues.
Q: What are the optimal reaction conditions for this synthesis?
A: According to patent CN113149865B, the optimal conditions involve using methanol as the solvent at a reaction temperature of 60°C. The molar ratio of benzaldehyde to ethyl cyanoacetate should be maintained at 1:1.5, with a catalyst loading of approximately 20mg per mmol of substrate, achieving maximum yield within 3 hours.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. The catalyst precursors (melamine and paraformaldehyde) are inexpensive and industrially available. The synthesis avoids high-pressure autoclaves and extreme temperatures (operating at mild 60°C), significantly reducing energy consumption and safety risks associated with scaling up fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl α-Cyanocinnamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the mPMF system in delivering high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required by global regulatory bodies. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are equipped to handle complex organic syntheses with the precision and safety necessary for commercial success.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of high-purity ethyl α-cyanocinnamate for your downstream applications.
