Scalable Enzymatic Synthesis of Chiral (2S,3R)-p-Methylsulfonylphenylserine Ethyl Ester for Antibiotic Production
Scalable Enzymatic Synthesis of Chiral (2S,3R)-p-Methylsulfonylphenylserine Ethyl Ester for Antibiotic Production
The pharmaceutical industry continuously seeks robust and sustainable pathways for producing key antibiotic intermediates, particularly for broad-spectrum agents like Florfenicol and Thiamphenicol. Patent CN109836362B introduces a transformative biocatalytic methodology for synthesizing (2S,3R)-p-methylsulfonylphenylserine ethyl ester, a critical chiral building block. This innovation shifts the paradigm from traditional heavy-metal-catalyzed chemical resolution to a highly selective enzymatic process utilizing transaldolase. By leveraging the chiral pool of L-threonine, this method achieves superior stereocontrol and operational simplicity. The target molecule, characterized by its specific (2S,3R) configuration, is essential for the biological activity of the final drug substances, making the purity and optical integrity of this intermediate paramount for regulatory compliance and therapeutic efficacy.
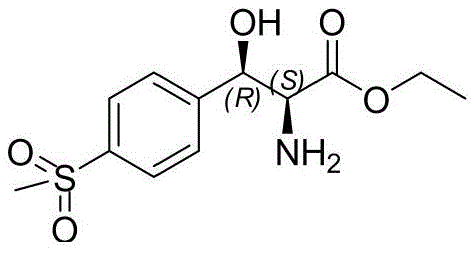
This technical breakthrough addresses the longstanding inefficiencies associated with the industrial manufacture of phenylserine derivatives. The patented process not only enhances the stereoselectivity to levels exceeding 99% ee and dr but also streamlines the downstream processing by eliminating complex resolution steps. For R&D directors and process chemists, this represents a significant opportunity to optimize the impurity profile of the final API. Furthermore, the use of readily available bio-based starting materials aligns with modern green chemistry principles, offering a compelling value proposition for procurement teams focused on long-term sustainability and cost stability in the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of (2S,3R)-p-methylsulfonylphenylserine ethyl ester has relied on a chemically intensive route involving the condensation of p-methylsulfonylbenzaldehyde with glycine. This traditional pathway is fraught with significant technical and environmental drawbacks that hinder efficient large-scale production. The primary bottleneck is the necessity for chiral resolution to separate the desired enantiomer from the racemic mixture, a step that inherently caps the theoretical maximum yield at 50% and often results in practical yields below 35%. Additionally, the condensation reaction requires stoichiometric or catalytic amounts of copper salts, which introduces severe contamination issues.
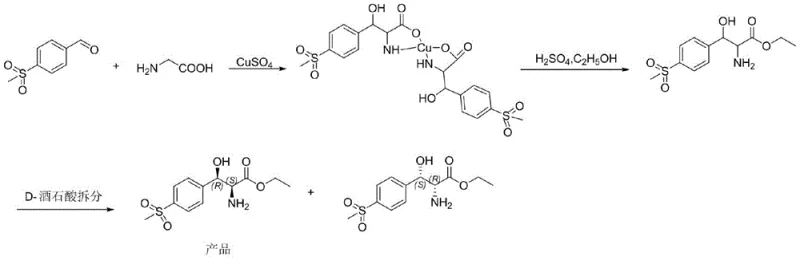
The removal of residual copper ions from the product stream is a particularly hazardous and costly operation, typically requiring the use of malodorous and toxic hydrogen sulfide gas. This generates substantial quantities of heavy metal-containing wastewater and solid waste, posing serious environmental compliance challenges and increasing waste disposal costs. The complexity of the splitting step further complicates the manufacturing process, requiring additional solvents, reagents, and unit operations. These factors collectively contribute to a high production cost and a fragile supply chain that is vulnerable to regulatory scrutiny regarding environmental emissions and worker safety.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a biocatalytic strategy that fundamentally redesigns the synthetic logic. By employing transaldolase (LTTA) as the catalyst and L-threonine as the chiral substrate, the process bypasses the need for racemic synthesis and subsequent resolution. The reaction proceeds in a mild aqueous phosphate buffer system, utilizing pyridoxal phosphate as a coenzyme and magnesium chloride as an activator to facilitate the transformation. This enzymatic condensation exhibits exceptional stereoselectivity, directly yielding the desired (2S,3R) configuration with high fidelity.
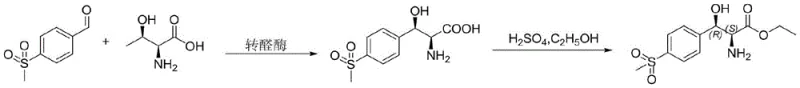
Furthermore, the new method allows for a telescoped process where the amino acid intermediate generated by the enzyme is directly subjected to ethylation without prior purification. This integration of biocatalysis and chemical synthesis significantly reduces the number of isolation steps, solvent usage, and overall processing time. The elimination of heavy metals and toxic gases not only improves the environmental footprint but also simplifies the quality control requirements for metal residues. This streamlined workflow translates directly into improved process economics and a more robust manufacturing capability suitable for meeting the growing global demand for veterinary and human antibiotics.
Mechanistic Insights into Transaldolase-Catalyzed Asymmetric Synthesis
The core of this technological advancement lies in the precise mechanistic action of the transaldolase enzyme, which facilitates the carbon-carbon bond formation with exquisite stereocontrol. Unlike non-enzymatic aldol reactions that often require harsh bases or Lewis acids and result in mixtures of diastereomers, the biocatalyst creates a highly defined chiral environment within its active site. The enzyme likely operates through a Schiff base mechanism involving the pyridoxal phosphate cofactor, which activates the L-threonine substrate. This activation allows for the specific cleavage or rearrangement that generates a nucleophilic species capable of attacking the p-methylsulfonylbenzaldehyde electrophile.
The stereochemical outcome is dictated by the inherent chirality of the L-threonine starting material and the geometric constraints of the enzyme-substrate complex. This "chiral pool" strategy ensures that the newly formed stereocenters at the alpha and beta positions of the serine backbone are established with the correct (2S,3R) configuration from the outset. The high diastereomeric ratio (dr > 99%) and enantiomeric excess (ee > 99%) observed in the examples indicate that the enzyme effectively suppresses the formation of unwanted (2R,3S), (2R,3R), or (2S,3S) isomers. This level of control is difficult to achieve with small-molecule organocatalysts or metal complexes without extensive ligand optimization.
From an impurity control perspective, this mechanism offers distinct advantages. Since the reaction does not proceed through a racemic intermediate, the burden on downstream purification is drastically reduced. The primary impurities are likely unreacted starting materials or minor enzymatic byproducts that are easily removed during the workup, such as the calcium salt precipitation step described in the patent. The absence of metal catalysts eliminates the risk of metal-catalyzed degradation pathways or complexation issues that can plague traditional syntheses. This clean reaction profile is critical for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals must be maintained throughout the synthesis of the final drug substance.
How to Synthesize (2S,3R)-p-Methylsulfonylphenylserine Ethyl Ester Efficiently
The implementation of this biocatalytic route requires careful attention to reaction parameters to maximize the efficiency of the enzyme and the subsequent chemical steps. The process begins with the preparation of a reaction mixture containing p-methylsulfonylbenzaldehyde and L-threonine in a buffered aqueous solution. The addition of magnesium chloride and pyridoxal phosphate is crucial for maintaining enzyme activity and stability during the transformation. Maintaining the temperature at approximately 30°C ensures optimal catalytic turnover while preventing thermal denaturation of the protein catalyst. Following the enzymatic conversion, which can achieve over 95% conversion within 20 hours, the reaction is terminated by heating, and the product is isolated via calcium salt precipitation.
- Condense p-methylsulfonylbenzaldehyde with L-threonine in a phosphate buffer containing magnesium chloride and pyridoxal phosphate, catalyzed by transaldolase at 30°C.
- Terminate the enzymatic reaction by heating, filter off precipitated calcium salts after pH adjustment, and concentrate the filtrate.
- Perform direct ethylation on the crude amino acid intermediate using ethanol and sulfuric acid under reflux to obtain the final ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers substantial strategic benefits beyond mere technical performance. The most significant advantage is the drastic reduction in raw material costs associated with the elimination of the chiral resolution step. In traditional manufacturing, half of the produced material is discarded or requires recycling, representing a massive inefficiency. By utilizing L-threonine, a commodity fermentation product available in large volumes, the process ensures a stable and cost-effective supply of chiral starting material. This shift from a resolution-based model to an asymmetric synthesis model fundamentally alters the cost structure of the intermediate, enabling significant margin improvements.
- Cost Reduction in Manufacturing: The removal of copper salts and the associated wastewater treatment infrastructure leads to direct operational expenditure savings. Traditional methods require expensive effluent treatment to handle heavy metals and the procurement of hazardous reagents like hydrogen sulfide. The new aqueous-based enzymatic process generates benign waste streams that are easier and cheaper to treat. Furthermore, the telescoping of the enzymatic and esterification steps reduces solvent consumption and energy usage by eliminating intermediate drying and purification stages. These cumulative efficiencies result in a leaner manufacturing process with a lower cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Relying on L-threonine and p-methylsulfonylbenzaldehyde diversifies the supply base away from specialized chiral resolving agents or expensive metal catalysts. Both key starting materials are produced on a multi-ton scale globally, ensuring high availability and price stability. The simplified process flow also reduces the risk of production delays caused by complex purification bottlenecks or equipment fouling from metal salts. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: The mild reaction conditions (30°C, atmospheric pressure, aqueous buffer) make this process inherently safer and easier to scale from pilot to commercial production compared to exothermic chemical condensations. The absence of toxic gases and heavy metals simplifies regulatory compliance and reduces the environmental liability of the manufacturing site. This "green" profile aligns with the increasing corporate sustainability goals of major pharmaceutical companies, potentially qualifying the supplier for preferred vendor status based on environmental, social, and governance (ESG) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic process. Understanding these details is essential for evaluating the feasibility of adopting this technology for your specific supply chain needs. The answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: How does the enzymatic route improve yield compared to traditional chemical synthesis?
A: Traditional methods rely on chiral resolution of racemates, which theoretically limits yield to 50% and practically results in yields below 35%. The enzymatic route utilizes L-threonine as a chiral starting material, bypassing the resolution step entirely and achieving conversion rates exceeding 95% with high stereoselectivity.
Q: What are the environmental benefits of replacing copper catalysis with transaldolase?
A: The conventional process requires large amounts of copper salts and toxic hydrogen sulfide gas for metal removal, generating hazardous heavy metal wastewater. The biocatalytic method operates in aqueous phosphate buffer without heavy metals, significantly reducing wastewater treatment costs and eliminating toxic gas emissions.
Q: Is purification required between the enzymatic step and esterification?
A: No, the process allows for a telescoped operation. The amino acid intermediate obtained from the enzymatic reaction can be directly subjected to ethylation without isolation or purification, which simplifies the workflow and reduces solvent consumption and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3R)-p-Methylsulfonylphenylserine Ethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics. Our technical team has thoroughly analyzed the potential of this enzymatic route and is well-positioned to support its industrialization. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to manufacturing reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical purity required for FDA and EMA compliance.
We invite you to collaborate with us to leverage this advanced technology for your antibiotic supply chain. Our experts can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green synthesis can lower your overall procurement costs. Please contact our technical procurement team to request specific COA data and route feasibility assessments. We are committed to delivering reliable supply and technical excellence, serving as your strategic partner in the competitive pharmaceutical intermediates market.
