Scalable Production of Optically Pure N-Boc-Difluorophenylalanine for Advanced Pharmaceutical Intermediates
Scalable Production of Optically Pure N-Boc-Difluorophenylalanine for Advanced Pharmaceutical Intermediates
The pharmaceutical industry's relentless pursuit of metabolic stability and enhanced bioavailability has placed fluorinated amino acids at the forefront of modern drug design. Patent CN100336803C introduces a robust and industrially viable methodology for the preparation of optically pure N-tertiary butyl oxycarbonyl-beta-difluorophenylalanine, a critical building block for bioactive peptides. This technology addresses the longstanding challenges associated with chiral synthesis by leveraging classical diastereomeric salt formation, a technique that offers superior scalability compared to transition metal-catalyzed routes. For R&D directors and process chemists, this patent represents a pivotal shift towards more manageable and cost-effective manufacturing protocols for complex fluorinated intermediates. By utilizing readily available chiral amines such as R-(+)-alpha-phenylethylamine or S-(-)-alpha-phenylethylamine, the process achieves high enantiomeric excess without the need for exotic catalysts. This report analyzes the technical merits and commercial implications of this resolution strategy, providing a comprehensive roadmap for integrating this high-value intermediate into global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral fluorinated phenylalanine derivatives has relied heavily on asymmetric hydrogenation, a method that, while elegant in theory, presents significant hurdles for large-scale industrial application. As illustrated in the prior art, conventional routes often necessitate the use of precious metal catalysts, specifically Rhodium (I) complexes, which are not only prohibitively expensive but also pose severe contamination risks that require rigorous downstream purification. Furthermore, these hydrogenation reactions typically demand high-pressure conditions, often ranging between 30 to 40 Psi, requiring specialized reactor infrastructure that increases capital expenditure and operational safety risks. Another critical limitation is the inherent restriction of asymmetric catalysis to producing a single enantiomer per batch, leaving the unwanted isomer as waste unless a dynamic kinetic resolution is employed, which adds further complexity. The difficulty in preparing and stabilizing these sensitive catalysts, combined with the harsh reaction conditions, creates a bottleneck for manufacturers aiming to produce kilogram-to-ton quantities of pharmaceutical intermediates efficiently.
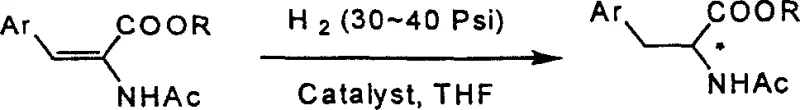
The Novel Approach
In stark contrast to the high-pressure catalytic routes, the methodology disclosed in CN100336803C employs a classical yet highly optimized chiral resolution strategy that transforms the production landscape for difluorophenylalanine derivatives. This novel approach utilizes the formation of diastereomeric salts between the racemic N-Boc-difluorophenylalanine and inexpensive, commercially available chiral resolving agents like alpha-phenylethylamine. The process operates under mild conditions, typically ranging from room temperature to reflux in common solvents like ethyl acetate, eliminating the need for high-pressure vessels and sensitive inert atmospheres. By exploiting the subtle solubility differences between the diastereomeric salts, the desired enantiomer can be selectively crystallized out of the solution with high purity. This method not only simplifies the operational workflow but also allows for the recovery and recycling of the resolving agent, thereby drastically reducing raw material costs. The ability to access both R and S configurations simply by switching the enantiomer of the resolving agent provides unparalleled flexibility for synthesizing diverse peptide libraries.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this technology lies in the precise molecular recognition and thermodynamic stability differences between the diastereomeric salts formed during the reaction. When the racemic N-tertiary butyl oxycarbonyl-beta-difluorophenylalanine interacts with a chiral amine such as S-(-)-alpha-phenylethylamine, two distinct diastereomeric salts are generated in situ. Due to the specific spatial arrangement of the fluorine atoms on the phenyl ring and the steric bulk of the Boc group, one diastereomer exhibits significantly lower solubility in the chosen solvent system, typically ethyl acetate, compared to its counterpart. This solubility disparity drives the selective crystallization of the less soluble salt, effectively separating the enantiomers. The presence of fluorine atoms enhances the lipophilicity and electronic properties of the molecule, which can influence the crystal lattice energy and packing efficiency, further aiding in the separation process. Understanding these intermolecular forces is crucial for optimizing the crystallization kinetics and maximizing the yield of the target optical isomer.
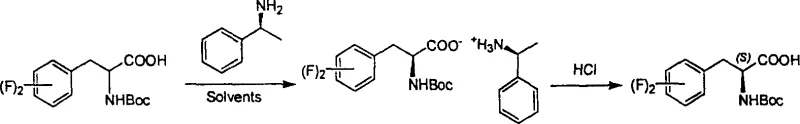
Following the initial crystallization, the process involves a straightforward acid-base workup to liberate the free amino acid derivative. The isolated diastereomeric salt is treated with a mild acid, such as aqueous citric acid, which protonates the amine resolving agent and releases the optically pure N-Boc-difluorophenylalanine into the organic phase. This step is critical for ensuring that no trace of the resolving agent remains in the final product, which is essential for meeting stringent pharmaceutical purity specifications. The mother liquor, which contains the enriched opposite enantiomer, can be subjected to subsequent resolution cycles using the opposite chiral amine, thereby maximizing the overall atom economy of the process. For instance, the mother liquor from the S-amine resolution can be treated with R-(+)-alpha-phenylethylamine to recover the R-enantiomer, demonstrating the versatility and efficiency of this dual-resolution capability. This mechanistic pathway ensures that both enantiomers are accessible from a single racemic starting material, minimizing waste and enhancing the sustainability profile of the manufacturing process.
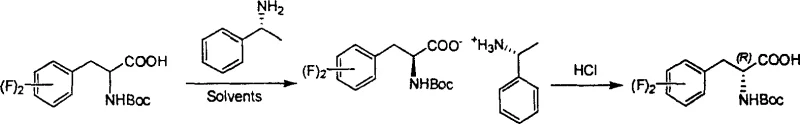
How to Synthesize N-Boc-Difluorophenylalanine Efficiently
The synthesis of this high-value intermediate follows a streamlined protocol that balances high optical purity with operational simplicity, making it ideal for both pilot and commercial scale production. The process begins with the dissolution of the racemic starting material in a suitable solvent, followed by the controlled addition of the chiral resolving agent to induce selective precipitation. Detailed standard operating procedures regarding temperature gradients, stirring rates, and filtration techniques are essential to reproduce the high e.e. values reported in the patent literature. Operators must pay close attention to the cooling rates and seeding strategies to ensure the formation of high-quality crystals that trap minimal impurities. The following guide outlines the critical stages of this resolution process, serving as a foundational reference for process engineers looking to implement this technology.
- Dissolve racemic N-tertiary butyl oxycarbonyl-beta-difluorophenylalanine in ethyl acetate and heat to reflux to ensure complete solubility before adding the resolving agent.
- Slowly add the chiral resolving agent, either S-(-)-alpha-phenylethylamine or R-(+)-alpha-phenylethylamine, in a 1: 1 molar ratio while maintaining controlled cooling to induce crystallization.
- Filter the resulting diastereomeric salt crystals, wash with cold solvent, and subsequently treat with aqueous citric acid to liberate the optically pure amino acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this resolution technology offers transformative benefits that directly impact the bottom line and operational resilience. By shifting away from precious metal catalysis, manufacturers can eliminate the volatility associated with Rhodium prices and the complex logistics of handling hazardous high-pressure gases. The reliance on commodity chemicals like ethyl acetate and alpha-phenylethylamine ensures a stable and diversified supply base, reducing the risk of production stoppages due to raw material shortages. Furthermore, the ambient pressure conditions allow for the utilization of standard glass-lined or stainless steel reactors, significantly lowering the barrier to entry for contract manufacturing organizations. This accessibility translates into a more competitive pricing structure for the final API intermediate, enabling pharmaceutical companies to optimize their cost of goods sold without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of expensive Rhodium catalysts and high-pressure equipment results in substantial capital and operational expenditure savings. The ability to recover and recycle the chiral resolving agent further drives down the variable cost per kilogram, making the process economically superior to asymmetric hydrogenation routes. Additionally, the simplified workup procedures reduce solvent consumption and energy usage, contributing to a leaner manufacturing footprint. These cumulative efficiencies allow for a more aggressive pricing strategy in the competitive market for fluorinated amino acids.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials mitigates the risk of supply chain disruptions often associated with specialized catalysts or reagents. The robustness of the crystallization process ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding costly delays in drug development timelines. The scalability of the method means that supply can be rapidly ramped up to meet clinical trial demands or commercial launch volumes without the need for extensive process re-validation. This reliability fosters stronger partnerships between suppliers and pharmaceutical innovators.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to heavy metal-catalyzed reactions, simplifying effluent treatment and disposal protocols. The absence of toxic metals in the product stream reduces the burden on downstream purification steps, aligning with green chemistry principles and increasingly strict environmental regulations. The straightforward nature of the unit operations facilitates easy scale-up from laboratory grams to multi-ton commercial production, ensuring that the technology remains viable as market demand grows. This environmental and operational compatibility positions the method as a sustainable choice for long-term manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of optically pure N-Boc-difluorophenylalanine. These insights are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing clarity on the feasibility and performance of this resolution method. Understanding these nuances is vital for stakeholders evaluating the integration of this intermediate into their synthetic pathways.
Q: What optical purity can be achieved using this resolution method?
A: According to the patent data, the initial crystallization yields enantiomers with an e.e. value of up to 98.8%. Through recrystallization and recycling of mother liquors, the total recovery of optically pure material can reach approximately 84%.
Q: Is the chiral resolving agent recoverable in this process?
A: Yes, the process is designed for sustainability and cost efficiency. After the acidification and extraction of the product, the aqueous layer containing the resolving agent can be alkalized to recover the alpha-phenylethylamine for reuse.
Q: Why is this method preferred over asymmetric hydrogenation for large-scale production?
A: Unlike asymmetric hydrogenation which requires expensive Rhodium catalysts and high-pressure equipment (30-40 Psi), this resolution method operates at atmospheric pressure using standard crystallization techniques, significantly reducing capital expenditure and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Boc-Difluorophenylalanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity fluorinated amino acids play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-Boc-difluorophenylalanine meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of chiral resolution, delivering products that facilitate smoother regulatory filings and faster time-to-market for our clients.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your specific drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency. Let us be your partner in transforming complex chemical challenges into commercial successes.
