Advanced Red Light Organic Electrophosphorescent Materials for High-Efficiency OLED Displays
The rapid evolution of organic light-emitting diode (OLED) technology has created an urgent demand for materials that can deliver high efficiency and stability, particularly in the red spectrum where concentration quenching has historically been a bottleneck. Patent CN113461739A addresses this critical challenge by disclosing a novel class of red light organic electrophosphorescent materials based on tricyclic metal iridium complexes. Unlike traditional bicyclic complexes that suffer from severe efficiency roll-off at higher driving voltages, these new materials incorporate rigid steric hindrance groups directly onto the phthalazine ligand framework. This strategic molecular design effectively minimizes the interaction between iridium atoms, thereby suppressing the self-quenching of triplet excitons. As a reliable OLED material supplier, understanding the nuances of this patent is essential for developing next-generation displays that require pure red emission without the compromise of luminous efficiency. The invention not only solves the sensitivity to doping concentration but also ensures that the electroluminescent wavelength remains stable regardless of voltage fluctuations, a key requirement for high-end commercial displays.
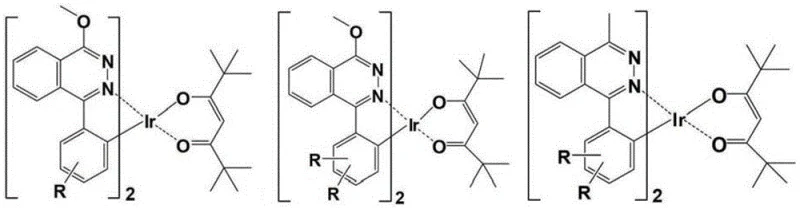
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of red phosphorescent dopants has been plagued by the phenomenon of concentration quenching, where the efficiency of the material drops precipitously as the concentration of the emitter increases. Prior art, including various bicyclic metal iridium complexes, often lacks sufficient steric protection around the metal center. In these conventional structures, the methyl steric hindrance on the ring metal benzene ring is typically very small, and the phthalazine ring often lacks any steric hindrance group entirely. This structural deficiency allows for strong intermolecular interactions, leading to non-radiative energy loss and a significant reduction in device performance. Furthermore, existing methods for introducing steric groups, such as hyperbranched structures or high molecular weight polymers, are often difficult to control, purify, and synthesize, resulting in lower device efficiency and higher production costs. The lack of comprehensive steric hindrance in these older generations of materials means that the optimal doping concentration is usually restricted to very low levels, typically not more than 5%, which limits the flexibility of device engineering and increases the complexity of the manufacturing process.
The Novel Approach
The innovative approach detailed in this patent fundamentally shifts the paradigm by utilizing a tricyclic metal iridium complex architecture equipped with comprehensive steric hindrance groups. By modifying the phthalazine ligand with rigid groups such as di-tert-butyl, pterene, or ortho-position carbazole units, the inventors have created a massive steric hindrance surface that physically isolates the luminescent centers. This design ensures that the direct action among metal atoms is drastically reduced, which in turn minimizes the self-quenching phenomenon of triplet excitons. The result is a material that maintains high luminous efficiency even in a pure solid state, with quantum efficiencies reaching up to 40% in some embodiments, a substantial improvement over the 2.2% external quantum efficiency observed in comparative complexes with insufficient steric hindrance. This novel approach not only enhances the stability of the complex but also simplifies the device fabrication process by allowing for higher doping concentrations without sacrificing performance, representing a significant leap forward in cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Steric Hindrance and Cyclometalation
The superior performance of these materials can be attributed to a sophisticated interplay of electronic and steric effects engineered into the ligand structure. The R1 substituent, often an electron-withdrawing group like a fluoroalkyl or a bulky alkyl group, serves a triple function: it increases the acidity of the hydrogen atom on the cyclometalated benzene ring to facilitate coordination with the iridium atom, enhances the electron transmission capability of the complex, and provides steric density in the direction of the C-Ir bond. Simultaneously, the steric hindrance groups on the phthalazinyl benzene ring, such as the symmetrical di-tert-butyl groups, act as electron-donating groups that increase the coordination capacity of the phthalazine nitrogen atoms. This symmetrical structure is crucial as it avoids the generation of isomers when different nitrogens on the phthalazine coordinate, ensuring a uniform product profile. The rigidity of these groups is paramount; because they are not free to rotate, they significantly reduce the probability of non-radiative transition caused by group vibration, thereby channeling more energy into photon emission rather than heat.
Furthermore, the specific choice of the phthalazine core over pyridazine allows for a remarkable red shift in the emission wavelength to the desired 590 nm-650 nm region while maintaining a smaller intramolecular repulsion action. The electronic action of the benzo ring in the phthalazine structure makes the ligand easier to cyclometalate compared to pyridazine ligands, facilitating the formation of the stable tricyclic complex. The R2 group acts as a blocking site for the unstable phthalazine position, preventing oxidation to a ketone and regulating the coordination site to avoid polynuclear complex formation. This precise control over the molecular geometry and electronic environment ensures that the carrier transmission capability of the complex is balanced, which is critical for achieving high efficiency in OLED devices. The combination of these factors results in a material that is not only highly emissive but also robust against the environmental and operational stresses encountered in commercial display applications.
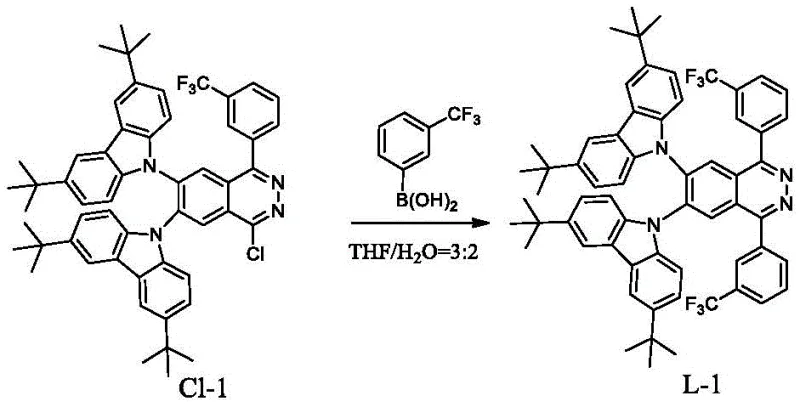
How to Synthesize High-Purity Phthalazine Iridium Complexes Efficiently
The synthesis of these advanced materials follows a robust and scalable two-stage process involving ligand construction followed by metallation. The first stage typically involves a palladium-catalyzed cross-coupling reaction, such as the Suzuki coupling, where a 1-chlorophthalazine derivative reacts with a substituted phenylboronic acid in the presence of a base like potassium carbonate and a catalyst like Pd(dppf)Cl2. Alternatively, for amine-substituted ligands, a nucleophilic substitution route using n-butyllithium and substituted diphenylamine is employed. The second stage involves the reaction of the purified ligand with an iridium source, such as iridium tris(acetylacetonate) or iridium trichloride, in high-boiling solvents like glycerol or ethoxyethanol under reflux conditions. This streamlined process avoids the costly and complex multi-step Diels-Alder reactions required by previous generations of sterically hindered materials. For detailed standardized synthesis steps and specific reaction parameters, please refer to the guide below.
- Synthesize the phthalazine ligand by reacting a 1-chlorophthalazine derivative with substituted phenylboronic acid using a palladium catalyst and base in a THF/water solvent system at 50-150°C.
- Alternatively, generate the ligand by reacting substituted diphenylamine or carbazole with a base and solvent, followed by the addition of the chlorophthalazine derivative at 0-180°C.
- Perform the final metallation by mixing the obtained phthalazine ligand with an iridium raw material such as Ir(acac)3 or IrCl3 in a high-boiling solvent like glycerol or ethoxyethanol at 50-200°C for 10-36 hours.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this technology offers distinct advantages rooted in process simplification and material stability. The elimination of complex synthetic pathways, specifically the avoidance of difficult Diels-Alder cycloadditions used in prior art to introduce steric groups, translates directly into a more streamlined manufacturing workflow. This simplification reduces the number of unit operations required, lowers the consumption of specialized reagents, and minimizes the generation of hazardous waste, aligning with modern green chemistry principles. For supply chain heads, this means a more predictable production timeline and reduced dependency on exotic starting materials, as the key precursors like chlorophthalazine derivatives and substituted boronic acids are commercially available and cost-effective. The robustness of the final tricyclic complex also implies better shelf-life and handling characteristics, reducing the risk of material degradation during storage and transport.
- Cost Reduction in Manufacturing: The synthetic route described in the patent utilizes standard organic transformations that are well-understood and easily optimized for large-scale production. By replacing expensive and low-yielding multi-step sequences with direct coupling and metallation reactions, the overall cost of goods sold is significantly lowered. The use of common solvents such as tetrahydrofuran, water, and glycerol further reduces solvent recovery costs and environmental compliance burdens. Additionally, the high purity achievable through standard recrystallization and column chromatography techniques minimizes the need for expensive preparative HPLC purification, leading to substantial cost savings in the final processing stages.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials ensures a stable supply chain that is less susceptible to market volatility. The starting materials, including various substituted phenylboronic acids and chlorophthalazine derivatives, are produced by multiple global suppliers, mitigating the risk of single-source dependency. Furthermore, the reaction conditions are relatively mild and tolerant, ranging from 50°C to 200°C, which allows for flexibility in reactor selection and operation. This flexibility enhances the resilience of the supply chain, enabling rapid scale-up from pilot to commercial production without the need for specialized high-pressure or cryogenic equipment.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction times ranging from 2 to 36 hours depending on the specific step, which fits well within standard batch processing schedules. The solvents used, such as ethoxyethanol and glycerol, have established recycling protocols, facilitating waste reduction and adherence to strict environmental regulations. The high yield of the ligand synthesis steps, often exceeding 75%, combined with the efficient metallation process, ensures high atom economy. This efficiency not only reduces the carbon footprint of the manufacturing process but also lowers the disposal costs associated with chemical waste, making it an environmentally responsible choice for sustainable electronic chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these red light organic electrophosphorescent materials. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for R&D and procurement teams evaluating this technology for integration into their product lines.
Q: How does the steric hindrance group improve the performance of red phosphorescent materials?
A: The introduction of rigid steric hindrance groups, such as tert-butyl or triptycene moieties, physically separates the iridium complex molecules. This spatial separation significantly reduces intermolecular interactions that lead to concentration quenching, thereby maintaining high luminous efficiency even in pure solid states or at high doping concentrations.
Q: What is the emission wavelength range of these new iridium complexes?
A: The disclosed red light organic electrophosphorescent materials exhibit a light-emitting wavelength range of 590 nm to 650 nm. This specific range is achieved through the phthalazine core structure which facilitates a significant red shift compared to pyridazine analogues, making them ideal for pure red emission in display applications.
Q: Are the synthesis methods suitable for large-scale production?
A: Yes, the preparation methods utilize standard organic synthesis techniques such as Suzuki coupling and nucleophilic substitution followed by reflux metallation. These processes avoid complex multi-step reactions like Diels-Alder cycloadditions found in prior art, using readily available solvents and catalysts that are conducive to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phthalazine Iridium Complex Supplier
As the demand for high-performance OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is crucial for navigating the complexities of commercialization. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to mass market availability is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade materials, guaranteeing that every batch of phthalazine iridium complex delivers consistent performance. We understand the critical nature of impurity profiles in OLED emitters and employ advanced analytical techniques to ensure that our products meet the highest standards of quality and reliability.
We invite you to engage with our technical procurement team to discuss how this patented technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to these sterically hindered complexes. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your specific application requirements. Whether you are developing next-generation smartphones, televisions, or lighting solutions, our expertise in the commercial scale-up of complex iridium complexes positions us as the ideal partner to drive your project forward with confidence and efficiency.
