Scalable Synthesis of 4-(4-Hydroxyphenylmethyleneamino)-1,2,4-triazole-5-thione Derivatives for Antiviral Applications
The pharmaceutical landscape is constantly evolving in the search for potent antiviral agents, particularly those capable of inhibiting influenza virus neuraminidase. A significant advancement in this domain is documented in patent CN109053606B, which discloses a novel class of 4-(4-hydroxyphenylmethyleneamino)-1H-1,2,4-triazole-5(4H)-thiones. These compounds represent a critical breakthrough in the development of next-generation neuraminidase inhibitors, offering a robust structural framework for medicinal chemistry optimization. The patent details a versatile synthetic methodology that allows for extensive structural diversity through the modulation of substituents on both the triazole ring and the phenyl moiety. This flexibility is paramount for tuning biological activity and pharmacokinetic properties in early-stage drug discovery. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented methodologies is essential for providing high-quality building blocks to global R&D teams.
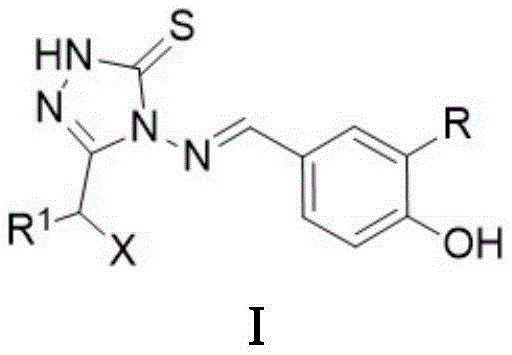
The core innovation lies in the specific arrangement of the 1,2,4-triazole-5-thione scaffold linked via a methyleneamino bridge to a hydroxyphenyl group. This architecture facilitates key interactions with the viral enzyme active site. The patent explicitly defines the scope of substituents, including alkyl chains, halogens, and alkoxy groups, which can be systematically varied to explore structure-activity relationships. For procurement managers and supply chain heads, the clarity of this chemical definition ensures that sourcing strategies can be aligned with specific analog requirements without ambiguity. The ability to access these complex heterocyclic intermediates reliably is a cornerstone for maintaining continuity in antiviral drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized triazole derivatives has often been plagued by multi-step sequences that suffer from low overall yields and poor atom economy. Traditional routes frequently require harsh reaction conditions, such as strong acids or bases at elevated temperatures, which can lead to the degradation of sensitive functional groups like hydroxyls or thiones. Furthermore, conventional methods often necessitate the use of expensive transition metal catalysts or hazardous reagents that complicate downstream purification and waste management. The presence of difficult-to-remove impurities often requires extensive chromatographic separation, which is neither cost-effective nor scalable for industrial production. These inefficiencies create significant bottlenecks in cost reduction in antiviral drug manufacturing, driving up the price of goods and extending lead times for critical API precursors.
The Novel Approach
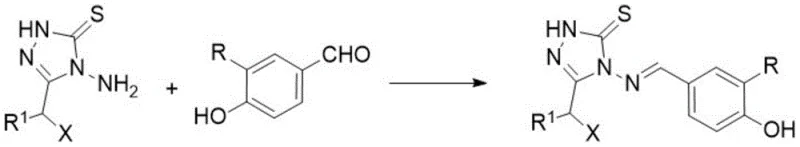
In stark contrast, the methodology presented in the patent employs a direct condensation reaction that significantly streamlines the production process. By reacting 4-amino-3-substituted-1H-1,2,4-triazole-5(4H)-thione directly with substituted benzaldehydes, the synthesis achieves the target Schiff base structure in a single operational step. This approach eliminates the need for intermediate isolation and reduces the consumption of solvents and reagents. The use of glacial acetic acid as both solvent and catalyst provides a mild yet effective medium that promotes the formation of the imine bond while preserving the integrity of the triazole ring. This simplification translates directly into enhanced supply chain reliability, as fewer unit operations mean fewer points of failure and reduced risk of batch-to-batch variability. The robustness of this method supports the commercial scale-up of complex heterocyclic intermediates, making it an attractive option for large-scale manufacturing.
Mechanistic Insights into Schiff Base Condensation
The fundamental chemical transformation driving this synthesis is a classic nucleophilic addition-elimination reaction, commonly known as Schiff base formation. The mechanism initiates with the nucleophilic attack of the exocyclic amino group on the triazole ring onto the electrophilic carbonyl carbon of the benzaldehyde derivative. This step forms a tetrahedral intermediate, which subsequently undergoes dehydration to establish the carbon-nitrogen double bond characteristic of the methyleneamino linkage. The presence of the electron-withdrawing triazole ring enhances the nucleophilicity of the amino group, facilitating the reaction under relatively mild reflux conditions. Acetic acid plays a dual role by protonating the carbonyl oxygen to increase its electrophilicity and by acting as a dehydrating agent to drive the equilibrium towards product formation. Understanding this mechanistic pathway is crucial for R&D directors focusing on purity and impurity profiles, as it highlights the potential for unreacted aldehyde or hydrolysis products as primary impurities.
Controlling the impurity profile in this reaction is largely dependent on the stoichiometry and reaction time. Since the reaction is reversible, using a slight excess of the aldehyde can drive the conversion to completion, minimizing the residual amine starting material. Conversely, careful control of water removal during the reflux period ensures that the equilibrium favors the product. The subsequent workup involving filtration and washing effectively removes soluble impurities, such as unreacted aldehydes and acetic acid residues. Recrystallization from ethanol further purifies the solid product by exploiting differences in solubility between the target compound and potential side products. This rigorous control over the reaction parameters ensures the delivery of high-purity triazole thione derivatives that meet the stringent specifications required for pharmaceutical applications.
How to Synthesize 4-(4-Hydroxyphenylmethyleneamino)-1,2,4-triazole-5-thione Efficiently
The practical execution of this synthesis is remarkably straightforward, making it highly accessible for laboratory and pilot-scale production. The process involves mixing the amine and aldehyde components in acetic acid and heating the mixture to reflux. The simplicity of the operation reduces the need for specialized equipment, allowing for rapid technology transfer between sites. Detailed procedural specifics regarding molar ratios, exact reflux times for different substituents, and crystallization parameters are critical for optimizing yield and quality. For a comprehensive guide on the standardized operating procedures, please refer to the technical steps outlined below.
- Mix 4-amino-3-substituted-1H-1,2,4-triazole-5(4H)-thione with the corresponding substituted benzaldehyde in glacial acetic acid.
- Heat the reaction mixture to reflux temperature and maintain stirring for 2.5 to 6 hours depending on the specific substituents.
- Cool the reaction, filter the precipitated solid, wash with dichloromethane or ethanol, and recrystallize from ethanol to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages that align with the strategic goals of cost efficiency and supply security. The reliance on commodity chemicals such as vanillin, hydroxybenzaldehyde, and acetic acid ensures that raw material costs remain low and stable. The elimination of precious metal catalysts removes a significant cost driver and simplifies the regulatory burden associated with heavy metal residuals in the final product. This inherent efficiency supports significant cost savings in the overall manufacturing budget, allowing for more competitive pricing of the final intermediates. Additionally, the high yields reported in the patent examples demonstrate the material efficiency of the process, reducing waste generation and disposal costs.
- Cost Reduction in Manufacturing: The process utilizes inexpensive, readily available starting materials and avoids the need for costly catalysts or anhydrous conditions. The one-pot nature of the reaction minimizes labor and utility costs associated with multiple isolation steps. By eliminating complex purification techniques like column chromatography in favor of simple recrystallization, the operational expenditure is drastically reduced. This economic efficiency makes the production of these antiviral intermediates financially viable even at smaller scales, fostering innovation in drug discovery.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis are bulk chemicals with established global supply chains, mitigating the risk of shortages. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or reagent quality. This stability ensures consistent output and reducing lead time for high-purity API precursors, which is critical for maintaining just-in-time inventory levels. Suppliers can confidently commit to delivery schedules knowing that the process is resilient and reproducible.
- Scalability and Environmental Compliance: The use of acetic acid as a solvent is advantageous for scale-up due to its favorable safety profile and ease of recovery. The reaction generates water as the only byproduct, aligning with green chemistry principles and simplifying waste treatment protocols. The solid product precipitates directly from the reaction mixture, facilitating easy filtration and reducing solvent consumption during workup. These factors collectively support the commercial scale-up of complex heterocyclic intermediates while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triazole derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details helps stakeholders make informed decisions regarding sourcing and development strategies. For further clarification on specific custom synthesis requirements, our technical team is available to provide detailed assessments.
Q: What is the primary application of these triazole thione derivatives?
A: These compounds are specifically designed as influenza virus neuraminidase inhibitors, showing significant potential as antiviral pharmaceutical intermediates.
Q: What are the typical reaction conditions for this synthesis?
A: The synthesis utilizes a simple reflux condition in glacial acetic acid, typically ranging from 2.5 to 6 hours, avoiding the need for complex catalysts or anhydrous environments.
Q: How is the purity of the final product ensured?
A: High purity is achieved through a straightforward workup involving filtration, washing with organic solvents like dichloromethane, and final recrystallization from ethanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Hydroxyphenylmethyleneamino)-1,2,4-triazole-5-thione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral therapies. Our expertise in heterocyclic chemistry positions us as a strategic partner for scaling these patented pathways from gram-scale research to multi-ton commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch meets the highest industry standards. We are committed to supporting your R&D efforts with materials that enable rapid progress in your drug discovery programs.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. Our team can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By leveraging our manufacturing capabilities, you can accelerate your timeline to market while maintaining control over costs and quality. Let us be your partner in bringing innovative antiviral solutions to the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
