Optimizing Vitamin A Intermediate Production: A Technical Analysis of Novel Chlorohydrin Methodologies
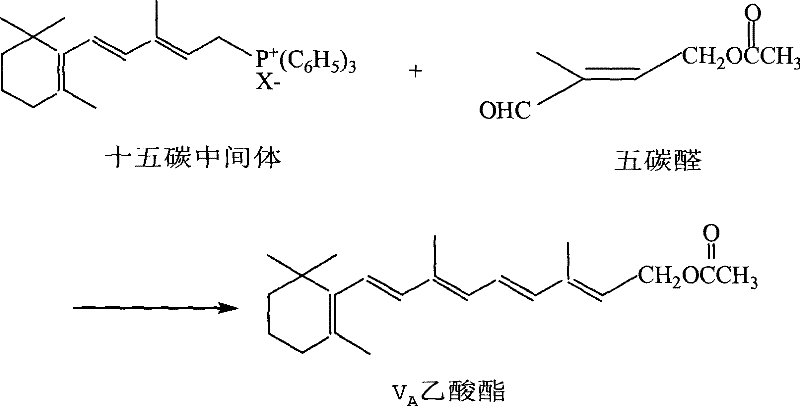
The global demand for Vitamin A acetate continues to drive innovation in the synthesis of its critical precursors, specifically focusing on efficiency and environmental compliance. Patent CN101270048B introduces a transformative approach to producing 1-chloro-2-methyl-4-acetoxy-2-butene, a pivotal C5 building block in the C15+C5 Wittig reaction pathway. Traditional manufacturing methods have long struggled with the limitations of aqueous sodium hypochlorite, which often results in suboptimal yields and significant saline effluent. This new methodology leverages tetrachloroglycoluril as a superior chlorinating agent, offering a reaction system that is both chemically robust and environmentally sustainable. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding this technological shift is essential for securing long-term supply chain stability. The strategic implementation of this chemistry not only enhances product purity but also aligns with modern green chemistry mandates required by top-tier multinational corporations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1-chloro-2-methyl-4-acetoxy-2-butene has relied heavily on the chlorohydrination of isoprene using sodium hypochlorite or calcium hypochlorite. While these reagents are readily available, they suffer from inherently low available chlorine content, typically ranging from 10% to 15% for liquid sodium hypochlorite and around 30% for solid calcium hypochlorite. This inefficiency necessitates the use of large volumes of aqueous solutions, leading to massive generation of inorganic salt waste such as sodium chloride or calcium chloride upon completion of the reaction. Furthermore, the presence of free chlorine gas and various heteroions in these traditional systems often promotes undesirable side reactions, including dichlorination of the olefin double bond. These side reactions complicate downstream purification, reduce overall yield to as low as 52% in some comparative examples, and increase the burden on wastewater treatment facilities. For a procurement manager focused on cost reduction in pharmaceutical intermediate manufacturing, these inefficiencies translate directly into higher raw material consumption and elevated disposal costs.
The Novel Approach
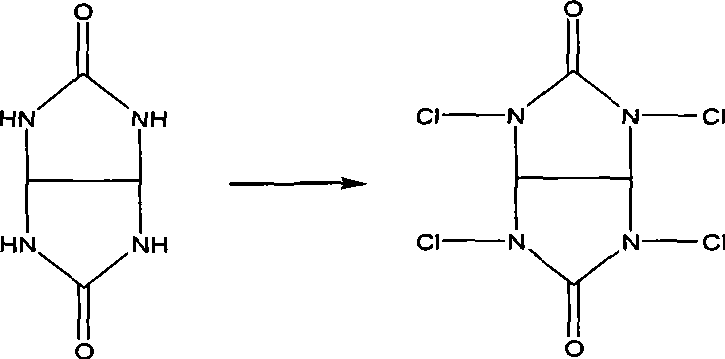
In stark contrast, the novel method described in the patent utilizes tetrachloroglycoluril, a compound with an available chlorine content as high as 95%, to drive the chlorohydrination process. This high concentration of active chlorine allows for a much more stoichiometric and controlled reaction environment, significantly minimizing the volume of water required and eliminating the introduction of extraneous metal ions. The reaction proceeds under mild conditions, typically between -5°C and 10°C, which helps preserve the integrity of the sensitive isoprene substrate while maximizing the formation of the desired 1,4-addition product. By replacing bulk hypochlorite solutions with this solid, high-efficiency reagent, the process fundamentally alters the mass balance of the production line. The result is a dramatic improvement in crude product content, often exceeding 92%, and a substantial reduction in the volume of three-waste emissions. This shift represents a critical upgrade for any facility aiming to scale up complex pharmaceutical intermediates while maintaining rigorous environmental standards.
Mechanistic Insights into Tetrachloroglycoluril-Mediated Chlorohydrination
The core innovation of this synthesis lies in the in situ generation of hypochlorous acid through the hydrolysis of tetrachloroglycoluril. Unlike the direct addition of unstable hypochlorite solutions, tetrachloroglycoluril reacts with water to release hypochlorous acid steadily and completely. This controlled release mechanism ensures that the concentration of active chlorinating species remains optimal throughout the reaction, preventing local excesses that could lead to over-chlorination or oxidative degradation of the diene system. The absence of free chlorine gas is particularly beneficial, as it mitigates the risk of radical-mediated side reactions that typically plague conventional chlorohydrination processes. From a technical perspective, this provides R&D teams with a much cleaner impurity profile, simplifying the analytical validation required for regulatory filings. The precision of this chemical transformation underscores the importance of selecting advanced reagents over commodity chemicals when targeting high-value fine chemical intermediates.
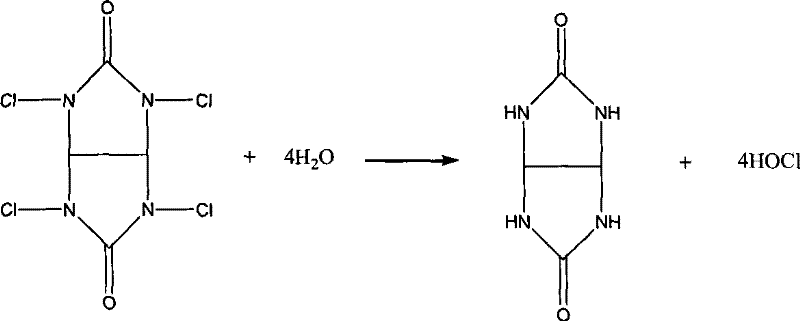
Furthermore, the impurity control mechanism is reinforced by the recyclability of the organic by-product, glycoluril. In traditional methods, the by-products are inorganic salts that accumulate in the wastewater stream, requiring energy-intensive treatment or disposal. In this novel system, the glycoluril framework remains intact after releasing its chlorine atoms and can be separated from the reaction mixture. This structural stability allows for the potential regeneration of the chlorinating agent, creating a semi-closed loop that drastically reduces raw material turnover. For supply chain heads concerned with the continuity of supply, this self-sustaining aspect of the chemistry reduces dependency on fluctuating markets for bulk oxidants. It ensures that the production of high-purity pharmaceutical intermediates remains resilient against external supply shocks, providing a strategic advantage in a competitive global market.
How to Synthesize 1-Chloro-2-Methyl-4-Acetoxy-2-Butene Efficiently
The operational protocol for this synthesis is designed to maximize safety and yield while minimizing environmental impact. The process begins with the preparation of a biphasic system containing isoprene, water, and a polymerization inhibitor such as hydroquinone, maintained at a controlled low temperature to prevent exothermic runaway. The key step involves the gradual addition of tetrachloroglycoluril, which ensures that the hydrolysis rate matches the consumption rate of the olefin, thereby maintaining a steady state of hypochlorous acid concentration. Following the chlorohydrination, the organic layer is separated and subjected to acetylation using acetic anhydride under acid catalysis to yield the final target molecule. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Prepare the reaction system by mixing isoprene with water and a polymerization inhibitor in a cooled vessel maintained between -5°C and 10°C.
- Gradually add tetrachloroglycoluril to generate hypochlorous acid in situ, ensuring the molar amount of water exceeds the chlorine content for complete hydrolysis.
- Separate the organic layer containing the chlorohydrin mixture and react with acetic anhydride under acid catalysis to yield the final acetoxy product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthesis route offers profound commercial benefits that extend beyond mere chemical yield improvements. For procurement managers, the shift to tetrachloroglycoluril represents a strategic opportunity to optimize the cost structure of Vitamin A intermediate manufacturing. The elimination of large volumes of saline wastewater reduces the operational expenditure associated with effluent treatment and regulatory compliance. Additionally, the higher selectivity of the reaction means that less raw material is wasted on by-products, effectively lowering the cost of goods sold without compromising on quality. These qualitative efficiencies contribute to a more competitive pricing model, allowing partners to secure cost reduction in electronic chemical manufacturing or pharmaceutical sectors through smarter process design rather than simple negotiation.
- Cost Reduction in Manufacturing: The utilization of a high-content chlorinating agent significantly reduces the molar equivalents of reagents required per kilogram of product. By avoiding the dilution effects inherent in liquid hypochlorite solutions, the process minimizes energy consumption related to heating, cooling, and solvent recovery. The ability to recycle the glycoluril by-product further amortizes the cost of the primary reagent over multiple batches. This cumulative effect leads to substantial cost savings over the lifecycle of the product, making it an economically superior choice for large-scale production runs where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: Relying on solid, stable reagents like tetrachloroglycoluril mitigates the risks associated with the transport and storage of hazardous liquid oxidants. The longer shelf life and higher density of the solid reagent simplify logistics, reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the robustness of the reaction against variations in pH and temperature ensures consistent batch-to-batch quality, which is critical for maintaining certification with global regulatory bodies. This reliability fosters stronger partnerships between suppliers and multinational clients who prioritize uninterrupted production schedules.
- Scalability and Environmental Compliance: The reduction in three-waste emissions aligns perfectly with increasingly stringent environmental regulations worldwide. The process generates minimal solid waste and significantly less aqueous waste compared to traditional methods, facilitating easier permitting and expansion of production capacity. This environmental stewardship enhances the corporate social responsibility profile of the manufacturing entity, appealing to end-users who require sustainable sourcing for their own ESG reporting. Scalability is thus achieved not just through equipment size, but through process intensification that respects ecological boundaries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantageous effects and background technical challenges outlined in the patent documentation. Understanding these nuances helps stakeholders make informed decisions about integrating this methodology into their existing production frameworks. The answers reflect a commitment to transparency and technical accuracy, ensuring that all parties have a clear understanding of the process capabilities and limitations.
Q: What are the advantages of using tetrachloroglycoluril over sodium hypochlorite?
A: Tetrachloroglycoluril offers a significantly higher available chlorine content (up to 95%) compared to sodium hypochlorite (10-15%), resulting in higher reaction yields and drastically reduced saline wastewater.
Q: How does this method impact impurity profiles in Vitamin A intermediates?
A: By avoiding free chlorine gas and minimizing heteroions, this method reduces dichlorination side reactions, leading to a cleaner product profile with content exceeding 92%.
Q: Is the by-product glycoluril recyclable in this process?
A: Yes, the glycoluril by-product generated during hydrolysis can be recovered and re-chlorinated, creating a closed-loop system that minimizes solid waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Chloro-2-Methyl-4-Acetoxy-2-Butene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1-chloro-2-methyl-4-acetoxy-2-butene meets the exacting standards required for Vitamin A synthesis. We are committed to delivering high-purity pharmaceutical intermediates that support your downstream processing needs without compromise.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic potential of switching to this greener methodology. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Together, we can build a more efficient, sustainable, and profitable supply chain for the next generation of fine chemicals.
