Advanced Biocatalytic Synthesis of 2,2-Dimethylcyclopropanecarboxamide for Commercial Scale-up
Introduction to Green Biocatalysis for Pharmaceutical Intermediates
The pharmaceutical industry is currently undergoing a paradigm shift towards sustainable manufacturing, driven by the urgent need to reduce environmental footprints while maintaining rigorous quality standards for active pharmaceutical ingredients (APIs). A pivotal development in this domain is documented in patent CN101481665B, which details a novel biocatalytic method for preparing 2,2-dimethylcyclopropanecarboxamide, a critical chiral precursor for the synthesis of Cilastatin. This patent introduces a specialized microbial strain, Rhodococcus boritolerans FW815, which exhibits exceptionally high nitrile hydratase activity, enabling the efficient hydration of 2,2-dimethylcyclopropanecarbonitrile under mild aqueous conditions. Unlike traditional chemical processes that rely on hazardous reagents and extreme temperatures, this biological approach leverages the inherent selectivity of enzymes to achieve near-quantitative conversion rates with minimal by-product formation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant advancement in process chemistry, offering a pathway to cleaner, more cost-effective production of complex organic molecules essential for modern medicine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amides from nitriles has been dominated by chemical hydrolysis methods that impose severe constraints on manufacturing efficiency and environmental compliance. Traditional protocols typically necessitate the use of strong mineral acids or concentrated alkalis, coupled with elevated reaction temperatures that demand substantial energy input and specialized corrosion-resistant equipment. These harsh conditions often lead to poor reaction selectivity, resulting in the formation of undesirable carboxylic acid by-products through over-hydrolysis, which complicates downstream purification and reduces overall yield. Furthermore, the reliance on stoichiometric amounts of corrosive reagents generates significant volumes of hazardous wastewater, creating a heavy burden for waste treatment facilities and increasing the total cost of ownership for chemical manufacturers. The presence of residual heavy metal catalysts, often required to accelerate these reactions, introduces additional risks regarding product safety and necessitates expensive removal steps to meet regulatory limits for pharmaceutical applications.
The Novel Approach
In stark contrast, the biocatalytic strategy outlined in the patent utilizes the nitrile hydratase enzyme produced by Rhodococcus boritolerans FW815 to facilitate the transformation in a purely aqueous medium at ambient temperatures ranging from 25°C to 35°C. This enzymatic process operates with remarkable atom economy, converting the nitrile group directly to the corresponding amide without generating stoichiometric salt waste or requiring toxic solvents. The biological system demonstrates exceptional tolerance to the substrate, allowing for high substrate loading concentrations up to 100 mM while maintaining rapid reaction kinetics that complete within minutes. By eliminating the need for extreme pH adjustments and high thermal energy, this novel approach drastically simplifies the reactor design and operational requirements, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates. The inherent specificity of the enzyme ensures that the cyclopropane ring remains intact, preventing ring-opening side reactions that are common in aggressive chemical environments, thereby securing a superior impurity profile for the final product.
Mechanistic Insights into Nitrile Hydratase Catalysis
The core of this technological breakthrough lies in the unique properties of the nitrile hydratase enzyme harbored within the Rhodococcus boritolerans FW815 strain, which functions as an iron-dependent metalloenzyme. The catalytic mechanism involves the coordination of the nitrile substrate to the non-heme iron center at the active site, where it undergoes nucleophilic attack by a metal-bound water molecule to form the amide product. Crucially, the patent highlights that the expression of this enzyme is inducible, requiring the presence of specific organic compounds such as caprolactam or the substrate itself during the fermentation phase to trigger high-level protein synthesis. This inducible nature allows for precise control over biocatalyst production, ensuring that the microbial cells are loaded with maximum enzymatic activity prior to the bioconversion step. The enzyme's stability in the presence of organic substrates is another key feature, permitting the reaction to proceed in a biphasic or high-concentration system without rapid denaturation, which is a common failure point for many industrial biocatalysts.
From a quality control perspective, the mechanistic pathway ensures a clean impurity profile because the enzyme strictly targets the cyano group without affecting other sensitive functional groups on the molecule. The patent data indicates that the conversion rate approaches 100%, leaving only trace amounts of unreacted nitrile, which simplifies the analytical burden for QC laboratories. The reaction medium consists primarily of water, buffer salts, and the biocatalyst cells, meaning that the post-reaction mixture is free from organic solvent residues that often plague chemically synthesized intermediates. This purity advantage is critical for downstream applications where residual solvents must be kept below strict ppm thresholds. The ability to recycle the whole-cell biocatalyst for multiple batches further underscores the robustness of the enzyme system, as the cellular matrix protects the active sites from shear stress and inhibitory effects, maintaining consistent performance over repeated cycles of operation.
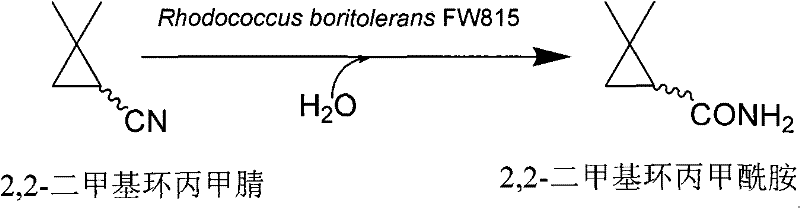
How to Synthesize 2,2-Dimethylcyclopropanecarboxamide Efficiently
Implementing this biocatalytic route requires a structured approach to fermentation and bioconversion to maximize yield and operational efficiency. The process begins with the cultivation of the Rhodococcus boritolerans FW815 strain in a defined fermentation medium supplemented with iron ions and an inducer such as caprolactam to stimulate nitrile hydratase production. Once the cells reach the desired density and enzymatic activity, they are harvested via centrifugation and washed to remove residual nutrients that could interfere with the subsequent conversion step. The prepared biocatalyst is then suspended in water or a phosphate buffer, and the substrate, 2,2-dimethylcyclopropanecarbonitrile, is introduced, often in a fed-batch manner to manage solubility and reaction exotherms. The detailed standardized synthesis steps for optimizing this pathway are provided in the guide below.
- Cultivate Rhodococcus boritolerans FW815 in an iron-supplemented fermentation medium with caprolactam as an inducer at 28-32°C for 36-96 hours to generate high-activity nitrile hydratase cells.
- Harvest the enzyme-containing cells via centrifugation and wash them thoroughly with physiological saline or phosphate buffer to remove residual media components.
- Suspend the washed cells in water or buffer, add 2,2-dimethylcyclopropanecarbonitrile substrate in batches at 25-35°C, and react until conversion is complete before purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition from chemical synthesis to this biocatalytic method offers profound strategic benefits that extend beyond simple technical metrics. The elimination of hazardous reagents and the reduction in energy consumption directly translate to lower operational expenditures, as the process does not require expensive corrosion-resistant reactors or complex waste neutralization systems. The simplicity of the downstream processing, which involves basic filtration and crystallization rather than multi-step extractions and chromatography, significantly shortens the production cycle time, allowing for faster turnaround on customer orders. Furthermore, the ability to reuse the biocatalyst cells for multiple batches enhances the overall productivity of the fermentation suite, effectively increasing capacity without the need for capital investment in new infrastructure. These factors combine to create a more resilient and cost-efficient supply chain capable of meeting the fluctuating demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The biocatalytic process fundamentally alters the cost structure by removing the need for precious metal catalysts and stoichiometric chemical reagents that drive up raw material expenses. Since the reaction proceeds in water at mild temperatures, the energy load for heating and cooling is drastically reduced compared to traditional high-temperature hydrolysis, leading to substantial utility savings. Additionally, the high selectivity of the enzyme minimizes the formation of by-products, which means less material is lost to waste streams and more of the starting material is converted into saleable product, improving the overall mass balance. The simplified purification protocol reduces the consumption of organic solvents and adsorbents, further lowering the variable costs associated with each kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Relying on a robust microbial strain like Rhodococcus boritolerans FW815 ensures a consistent and renewable source of catalytic activity, mitigating the risks associated with the supply volatility of specialized chemical catalysts. The fermentation-based production of the biocatalyst can be scaled independently of the conversion step, allowing manufacturers to stockpile active cells to buffer against potential disruptions in raw material availability. The mild reaction conditions also reduce the risk of equipment failure or safety incidents related to high-pressure or high-temperature operations, ensuring uninterrupted production schedules. This reliability is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates, enabling suppliers to respond swiftly to urgent client requirements without compromising on quality or safety standards.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system aligns perfectly with modern environmental regulations, as it generates minimal hazardous waste and avoids the emission of volatile organic compounds (VOCs). Scaling this process from laboratory to industrial levels is straightforward because the biological parameters, such as oxygen transfer and mixing, are well-understood and easily controlled in standard fermenters. The absence of toxic heavy metals in the process stream simplifies wastewater treatment and disposal, reducing the regulatory burden and associated compliance costs for the manufacturing facility. This environmental compatibility not only future-proofs the production line against tightening legislation but also enhances the brand reputation of the supplier as a partner committed to sustainable and responsible chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology, based on the specific data and findings presented in the patent literature. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the proven capabilities of the Rhodococcus boritolerans FW815 strain and its associated process parameters.
Q: What are the advantages of using Rhodococcus boritolerans FW815 over chemical synthesis?
A: The biocatalytic method using Rhodococcus boritolerans FW815 operates under mild conditions (25-35°C, neutral pH) compared to harsh chemical hydrolysis requiring strong acids or bases. It offers higher selectivity, eliminates heavy metal catalysts, simplifies downstream purification, and allows for cell reuse across multiple batches, significantly reducing environmental impact and operational costs.
Q: Can the biocatalyst cells be reused for multiple production cycles?
A: Yes, stability experiments indicate that the Rhodococcus boritolerans FW815 cells maintain high nitrile hydratase activity and can be recovered and reused for approximately 4 to 6 consecutive batches without significant loss of catalytic efficiency, enhancing process economics.
Q: What is the typical purity and conversion rate achieved with this method?
A: The process achieves near-quantitative substrate conversion rates, often exceeding 99%, with minimal by-product formation. The resulting 2,2-dimethylcyclopropanecarboxamide can be isolated with high purity through simple vacuum distillation and crystallization, meeting stringent specifications for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Dimethylcyclopropanecarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in delivering high-value pharmaceutical intermediates with superior quality and sustainability profiles. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN101481665B can be seamlessly translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2,2-dimethylcyclopropanecarboxamide meets the exacting standards required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of biological manufacturing, from strain optimization to downstream purification, delivering a product that supports your drug development timelines.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this biocatalytic route for your supply chain. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and value for your organization.
