Advanced Enzymatic Synthesis of High-Purity Beta-Amino Acids for Commercial Pharmaceutical Manufacturing
Advanced Enzymatic Synthesis of High-Purity Beta-Amino Acids for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes to produce optically active intermediates, particularly beta-amino acids which serve as critical building blocks for antibiotics, alkaloids, and modified peptides. Patent CN1260364C introduces a groundbreaking methodology for the preparation of enantiomerically enriched beta-amino acids through a novel enzymatic hydrolysis process. This technology fundamentally shifts the paradigm from traditional chemical resolution methods, which often rely on hazardous reagents and extreme conditions, to a biocatalytic approach that leverages the specificity of hydrolases. By specifically utilizing N-unprotected beta-amino acid esters with bulky alkyl groups, this invention achieves superior stereoselectivity and space-time efficiency, addressing long-standing challenges in the scalable production of high-purity chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active beta-aminocarboxylic acids has been plagued by significant technical and economic hurdles associated with conventional racemate resolution techniques. Traditional chemical methods often necessitate the use of stoichiometric amounts of expensive chiral reagents to form diastereomeric salts, a process that is inherently atom-inefficient and generates substantial chemical waste. Furthermore, certain established routes require the activation of reagents using dangerous substances such as n-butyllithium, posing severe safety risks and requiring specialized handling protocols that increase operational costs. Perhaps most critically, achieving satisfactory stereoselectivity in these chemical processes frequently demands cryogenic reaction conditions, often around -70°C, which imposes rigorous requirements on reactor materials and results in exorbitant energy consumption for cooling, thereby drastically inflating the manufacturing overhead for large-scale production.
The Novel Approach
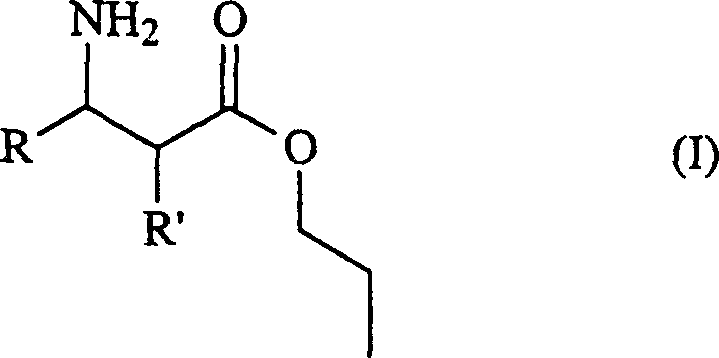
In stark contrast to these legacy methods, the process disclosed in CN1260364C utilizes a biocatalytic strategy that eliminates the need for stoichiometric chiral auxiliaries and extreme temperatures. The core innovation lies in the specific selection of the ester moiety attached to the beta-amino acid; rather than using common methyl or ethyl esters which have shown limited success in enzymatic resolutions, this method employs esters derived from bulkier alcohols such as n-propanol or n-butanol. This structural modification surprisingly enhances the interaction with the enzyme's active site, leading to markedly improved enantioselectivity and reaction rates. The process operates under mild physiological conditions, typically between 15°C and 40°C, and utilizes environmentally friendly hydrolases like Lipase PS, which can be easily separated from the reaction mixture, ensuring a cleaner product profile and simplifying downstream processing significantly.
Mechanistic Insights into Lipase-Catalyzed Ester Hydrolysis
The success of this enzymatic transformation hinges on the precise steric and electronic interactions between the substrate and the biocatalyst, specifically within the context of Lipase PS from Pseudomonas cepacia. In traditional enzymatic resolutions using methyl or ethyl esters, the small size of the ester group often fails to provide sufficient differentiation between the enantiomers within the enzyme's binding pocket, leading to mediocre enantiomeric excess values often hovering around 70% to 85%. However, by introducing a bulkier alkyl chain, such as a propyl or butyl group, the substrate adopts a conformation that fits more selectively into the chiral environment of the enzyme's active site. This steric bulk acts as a discriminatory handle, allowing the lipase to distinguish more effectively between the (R) and (S) enantiomers, thereby accelerating the hydrolysis of the desired enantiomer while leaving the other largely untouched, which is the fundamental driver for the observed high optical purity.
Furthermore, the mechanism benefits from the ability to conduct the reaction in a homogeneous or semi-homogeneous medium, which is crucial for industrial scalability and enzyme stability. The patent highlights that the addition of water-soluble organic solvents like acetone can be employed to dissolve the ester substrates without denaturing the enzyme, ensuring that the reaction proceeds in a single phase where mass transfer limitations are minimized. This homogeneity facilitates better contact between the biocatalyst and the substrate, leading to improved space-time yields. Additionally, the use of immobilized enzymes or specific formulations allows for the easy recovery and reuse of the biocatalyst, further enhancing the economic viability of the process. The result is a robust system capable of consistently delivering products with enantiomeric excess values exceeding 98%, and in optimized examples reaching as high as 99.6% ee, which is a critical threshold for pharmaceutical grade intermediates.
How to Synthesize N-Unprotected Beta-Amino Acid Esters Efficiently
The synthesis of these high-value chiral intermediates follows a streamlined protocol that prioritizes operational simplicity and safety, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins with the preparation of an aqueous reaction medium where the pH is carefully controlled, typically maintained between 7 and 8.5 using automated metering of base solutions like sodium hydroxide. To ensure complete solubility of the hydrophobic ester substrate, a co-solvent such as acetone is introduced, creating a homogeneous reaction phase that maximizes the interface between the enzyme and the reactant. Once the temperature stabilizes within the optimal range of 20°C to 30°C, the specific lipase catalyst is added to initiate the hydrolysis, triggering the selective cleavage of the ester bond on the target enantiomer.
- Prepare an aqueous reaction medium adjusted to a pH range of 7 to 8.5, optionally adding water-soluble organic solvents like acetone to ensure homogeneity.
- Introduce the racemic N-unprotected beta-amino acid ester substrate, specifically selecting bulky alkyl esters such as n-propyl or n-butyl variants rather than methyl or ethyl esters.
- Add a hydrolase catalyst, preferably Lipase PS from Pseudomonas cepacia, and maintain the reaction temperature between 15°C and 40°C to achieve high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology represents a strategic opportunity to de-risk the supply of critical chiral building blocks while simultaneously driving down the total cost of ownership. By shifting away from chemical resolution methods that rely on stoichiometric chiral reagents and cryogenic infrastructure, manufacturers can eliminate significant capital and operational expenditures associated with low-temperature reactors and hazardous material handling. The transition to a biocatalytic process operating at ambient temperatures drastically reduces energy consumption, as there is no longer a need for energy-intensive cooling systems to maintain -70°C conditions. This reduction in utility usage translates directly into lower variable costs per kilogram of product, enhancing the overall margin profile for the final API or intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric chiral resolving agents and dangerous activators like n-butyllithium removes a major cost driver from the bill of materials. Furthermore, the high enantioselectivity achieved with bulky esters minimizes the formation of unwanted isomers, reducing the need for costly recycling loops or complex purification steps to remove impurities. The ability to use catalytic amounts of enzyme, which can potentially be immobilized and reused, further decouples production costs from the price volatility of chiral chemicals, leading to a more stable and predictable cost structure for long-term supply agreements.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process under mild conditions ensures higher batch-to-batch consistency, which is vital for maintaining uninterrupted supply lines to downstream pharmaceutical customers. Unlike chemical methods that are sensitive to minor fluctuations in temperature or reagent quality, the biocatalytic route offers a wider operating window, reducing the risk of batch failures and subsequent supply shortages. Additionally, the use of commercially available lipases and simple alcohol precursors for the ester formation ensures that raw material sourcing is not dependent on niche or single-source suppliers, thereby diversifying the supply base and mitigating geopolitical or logistical risks.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers substantial advantages by avoiding the generation of heavy metal waste and toxic byproducts associated with traditional chemical synthesis. The aqueous nature of the reaction medium simplifies waste treatment and disposal, aligning with increasingly stringent global environmental regulations. The scalability is further supported by the ability to run the reaction in standard stirred-tank reactors without the need for specialized cryogenic equipment, allowing for rapid scale-up from pilot plant to multi-ton commercial production without significant engineering bottlenecks or regulatory hurdles related to process safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic hydrolysis technology for beta-amino acid production. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for R&D teams planning process validation and for procurement teams assessing supplier capabilities.
Q: Why are bulky esters preferred over methyl or ethyl esters in this enzymatic process?
A: According to patent CN1260364C, using bulky ester groups like n-propyl or n-butyl significantly enhances space-time efficiency and enantioselectivity compared to traditional methyl or ethyl esters, which often yield lower optical purity.
Q: What are the typical reaction conditions for this biocatalytic hydrolysis?
A: The process operates under mild conditions, typically at a pH of 7 to 8.5 and temperatures between 15°C and 40°C, utilizing Lipase PS in an aqueous or aqueous-organic solvent system.
Q: How does this method compare to conventional chemical resolution?
A: Unlike conventional methods requiring stoichiometric chiral reagents and cryogenic temperatures (-70°C), this enzymatic approach uses catalytic amounts of enzyme, avoids dangerous reagents like n-butyllithium, and achieves higher enantiomeric excess (>98% ee).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic hydrolysis route described in CN1260364C for producing high-purity beta-amino acid intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this biocatalytic process are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with the necessary fermentation and downstream processing capabilities to handle enzymatic reactions efficiently, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for these critical chiral intermediates. Our technical team is ready to perform a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this enzymatic route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and to discuss detailed route feasibility assessments for your target molecules, ensuring a seamless transition to a more sustainable and cost-effective supply source.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
