Scalable Enzymatic Synthesis of Alpha-Aspartyl Small Peptides for High-Purity Applications
Introduction to Advanced Peptide Synthesis Technology
The pharmaceutical and fine chemical industries constantly seek robust methodologies for synthesizing bioactive peptides with high stereochemical integrity. Patent CN102864196B introduces a groundbreaking enzymatic approach for preparing alpha-aspartyl small peptides, addressing long-standing challenges in regioselectivity and purity. This technology leverages the unique structural properties of dihydroorotic acid to co-protect the alpha-amino and beta-carboxyl groups of aspartic acid, effectively masking reactive sites that typically lead to unwanted by-products. By integrating biocatalysis with classical organic synthesis, this method offers a streamlined pathway to critical intermediates used in high-intensity sweeteners like aspartame and neuroactive peptides such as NAAG. For procurement and R&D leaders, this represents a significant opportunity to enhance product quality while simplifying manufacturing workflows.
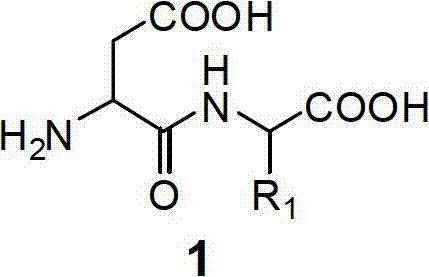
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of alpha-aspartyl peptides is fraught with complications, primarily stemming from the dual reactivity of aspartic acid. Conventional routes, such as the internal anhydride or lactone methods, often struggle with regioselectivity, leading to the formation of beta-linked isomers which are difficult to separate and reduce overall yield. Furthermore, the harsh conditions required for activation and coupling frequently induce racemization at the chiral center, compromising the biological activity and safety profile of the final product. These issues necessitate extensive purification steps, including repeated recrystallization or chromatography, which drastically increase production costs and extend lead times. The reliance on multiple protection and deprotection cycles also generates substantial chemical waste, posing environmental compliance challenges for modern manufacturing facilities.
The Novel Approach
The innovative method described in the patent circumvents these pitfalls by utilizing dihydroorotic acid as a cyclic protecting group that inherently directs reactivity. This strategy ensures that only the alpha-carboxyl group is exposed for peptide bond formation, guaranteeing position specificity without the need for auxiliary protecting groups. The subsequent enzymatic ring-opening step using dihydroorotase occurs under mild physiological conditions, preserving the stereochemical integrity of the molecule. This biomimetic approach not only eliminates the risk of beta-isomer formation but also significantly reduces the number of synthetic operations. By merging the efficiency of enzymatic catalysis with the scalability of chemical processing, this novel route provides a superior alternative for the reliable production of high-purity pharmaceutical intermediates and food additive ingredients.
Mechanistic Insights into Dihydroorotase-Catalyzed Ring Opening
The core of this synthesis lies in the precise action of dihydroorotase (DHOase, EC 3.5.2.3), a cyclic amidohydrolase that facilitates the reversible cyclization of N-carbamoyl-L-aspartic acid to L-dihydroorotic acid. In the context of this patented process, the enzyme operates in the hydrolytic direction, cleaving the lactam ring of the dihydroorotic acid peptide derivative. This reaction is highly pH-dependent; while acidic conditions favor cyclization, shifting the environment to weakly alkaline conditions (pH 7.5-9.5) drives the hydrolysis forward rapidly. The enzyme recognizes the exposed carboxyl group on the peptide chain as a positioning anchor, ensuring that the hydrolysis occurs specifically at the intended bond. This specificity is crucial for maintaining the structural fidelity of the peptide backbone, preventing random cleavage or side reactions that could degrade the product quality.
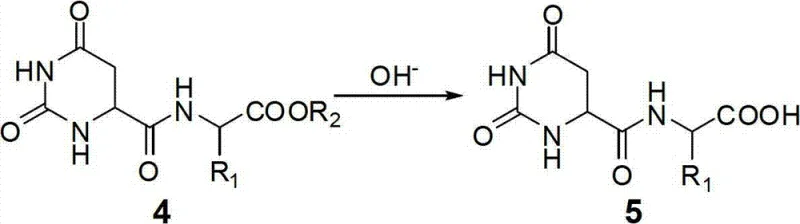
Impurity control is inherently built into this mechanistic design. Because the dihydroorotic acid ring physically blocks the beta-carboxyl group and the alpha-amino group during the initial coupling phase, the formation of branched peptides or diketopiperazines is sterically hindered. The enzymatic step further refines the mixture, as DHOase exhibits high substrate specificity for the dihydroorotate moiety, ignoring potential non-specific by-products generated during the chemical coupling. This dual-layer of selectivity—first steric protection, then enzymatic recognition—results in a crude product profile that is significantly cleaner than those obtained via purely chemical routes. Consequently, downstream purification becomes more efficient, requiring less solvent and fewer processing stages to achieve the stringent purity specifications demanded by regulatory bodies for pharmaceutical and food applications.
How to Synthesize Alpha-Aspartyl Small Peptides Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for implementing this technology at scale. It begins with the activation of dihydroorotic acid using standard peptide coupling reagents, followed by a mild alkaline hydrolysis to prepare the substrate for enzymatic action. The critical enzymatic transformation is conducted at moderate temperatures, ensuring enzyme stability and activity. Finally, a straightforward chemical deprotection yields the target molecule. This sequence balances chemical robustness with biological precision, offering a practical guide for process chemists aiming to adopt this greener methodology. For detailed operational parameters and specific reagent grades, please refer to the standardized synthesis guide below.
- Perform peptide coupling between dihydroorotic acid and amino acid esters using DCC/HOBt or DIC/HOBt in DMF or water to form dihydroorotic acid small peptide esters.
- Hydrolyze the ester bond of the intermediate using dilute alkali solution (1-5% NaOH/KOH) at room temperature to expose the terminal carboxyl group.
- Utilize dihydroorotase (DHOase) enzyme at pH 7.5-9.5 and 35-40°C to catalyze the ring-opening hydrolysis, forming N-carbamylated aspartyl small peptides.
- Remove the carbamoyl group via nitrous acid hydrolysis (NaNO2/HCl) under ice-bath conditions to yield the final alpha-aspartyl small peptide compound.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this enzymatic methodology offers tangible strategic benefits for supply chain optimization and cost management. By eliminating the need for complex multi-step protection strategies, the process reduces the consumption of expensive reagents and solvents, directly impacting the cost of goods sold. The simplified workflow also shortens the overall production cycle time, allowing for faster response to market demand fluctuations. Furthermore, the use of biocatalysts aligns with increasing global pressure for sustainable manufacturing practices, potentially reducing waste disposal costs and enhancing the company's environmental, social, and governance (ESG) profile. These factors combine to create a more resilient and cost-effective supply chain for critical peptide intermediates.
- Cost Reduction in Manufacturing: The elimination of repeated protection and deprotection steps significantly lowers material costs. Traditional methods often require stoichiometric amounts of protecting group reagents and subsequent removal agents, whereas this method uses the dihydroorotic acid ring as a reusable structural motif that is removed in a single enzymatic step. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling, contributing to substantial cost savings in utility expenses. The higher selectivity also means less product is lost to isomeric by-products, improving the overall mass balance and yield efficiency.
- Enhanced Supply Chain Reliability: The reliance on recombinant E. coli strains for enzyme production ensures a stable and scalable source of the biocatalyst. Unlike rare earth metal catalysts or exotic reagents that may face geopolitical supply constraints, fermentation-derived enzymes can be produced locally with consistent quality. This decentralization of supply risk enhances the reliability of the manufacturing process. Moreover, the robustness of the enzyme under various conditions allows for flexibility in processing, reducing the likelihood of batch failures due to minor deviations in reaction parameters.
- Scalability and Environmental Compliance: The process is designed for seamless scale-up from laboratory to commercial production. The enzymatic steps operate in aqueous or mixed solvent systems at near-neutral pH, minimizing the generation of hazardous acidic or basic waste streams. This simplifies wastewater treatment and reduces the environmental footprint of the facility. The ability to produce high-purity products with fewer purification steps also means less solvent waste is generated, aligning with green chemistry principles and facilitating easier regulatory approval for new drug applications or food additive notifications.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for successful implementation. The following questions address common concerns regarding process robustness, enzyme sourcing, and product quality. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this technology for their production lines.
Q: How does this method prevent aspartic acid racemization compared to traditional chemical synthesis?
A: Traditional chemical methods often involve harsh conditions or anhydride intermediates that promote racemization at the alpha-carbon. This patented method utilizes the dihydroorotic acid ring to co-protect both the alpha-amino and beta-carboxyl groups simultaneously. This structural constraint inherently prevents the formation of beta-isomers and minimizes racemization risks during the peptide coupling step, ensuring high optical purity without the need for complex chiral resolution.
Q: What is the source of the dihydroorotase enzyme used in this process?
A: The process employs dihydroorotase (DHOase, EC 3.5.2.3) which can be sourced from Escherichia coli K-12 (CICC20091) or produced via recombinant strains like E. coli Rosetta(DE3) carrying the pET-22b(+)-DHO-his plasmid. The enzyme is robust and operates under mild alkaline conditions (pH 7.5-9.5), making it suitable for large-scale fermentation and downstream processing without requiring extreme temperatures or pressures.
Q: Why is nitrous acid hydrolysis preferred for the final deprotection step?
A: While carbamoyl groups can be removed enzymatically, nitrous acid hydrolysis is selected for this specific small peptide synthesis due to its efficiency and simplicity in an industrial setting. The reaction proceeds rapidly under ice-bath conditions with sodium nitrite and dilute hydrochloric acid, achieving conversion rates close to 90-93%. This chemical step avoids the need for additional specialized enzymes, streamlining the supply chain and reducing overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Aspartyl Peptides Supplier
NINGBO INNO PHARMCHEM stands at the forefront of peptide synthesis innovation, leveraging advanced enzymatic technologies to deliver superior intermediates for the global market. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest standards required for pharmaceutical and food-grade applications. We understand the critical nature of supply continuity and are committed to being a partner you can trust.
We invite you to explore how this patented enzymatic route can optimize your production costs and enhance product quality. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization efforts. Let us collaborate to bring high-quality alpha-aspartyl peptides to the market faster and more efficiently.
