Scalable Manufacturing of Alkyl Ether Sulfonyl Fluoride Precursors for Next-Gen Surfactants
The global chemical industry is currently undergoing a paradigm shift driven by stringent environmental regulations regarding per- and polyfluoroalkyl substances (PFAS). Patent CN111499547A introduces a groundbreaking methodology for the preparation of alkyl ether sulfonyl fluoride compounds, which serve as critical precursors for next-generation perfluoroether surfactants. Unlike traditional long-chain perfluoroalkyl compounds that persist in the environment, the compounds synthesized via this novel route possess ether linkages that facilitate biodegradation while maintaining essential surface tension properties. This technical insight report analyzes the robust synthetic pathway detailed in the patent, highlighting its potential to revolutionize the production of eco-friendly specialty chemicals. By leveraging inexpensive commodity feedstocks and avoiding complex catalytic systems, this process offers a viable solution for manufacturers seeking to transition away from restricted substances like PFOS and PFOA without compromising on performance metrics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing fluorinated surfactants often rely on electrochemical fluorination (ECF) of long-chain sulfonyl halides or telomerization processes that result in mixtures of linear and branched isomers. These conventional approaches frequently utilize perfluorooctane sulfonyl fluoride (PFOSF) as a starting material, which has been identified as a persistent organic pollutant with significant bioaccumulation potential. The environmental persistence of C8-based compounds arises from the extreme stability of the carbon-fluorine and carbon-carbon bonds, leading to long-term ecological damage and health risks. Furthermore, the regulatory landscape has severely restricted the use of these legacy compounds, forcing R&D teams to search for alternatives that can match their surface activity without the associated toxicity. Existing short-chain alternatives often suffer from inferior surface tension parameters, limiting their application scope in critical industries such as electroplating, fire suppression, and coating formulations. The inability to balance performance with degradability has remained a significant bottleneck in the development of sustainable fluorinated materials.
The Novel Approach
The methodology disclosed in the patent presents a sophisticated yet practical solution by focusing on alkyl ether sulfonyl fluoride structures with specific carbon chain distributions, such as the (4+4) configuration. This approach strategically incorporates an ether bond into the molecular backbone, which acts as a weak point for environmental degradation while preserving the hydrophobic character necessary for surface activity. The synthesis begins with the ring-opening of tetrahydrofuran, a cheap and abundant cyclic ether, followed by a series of functional group transformations including sulfonation and etherification. 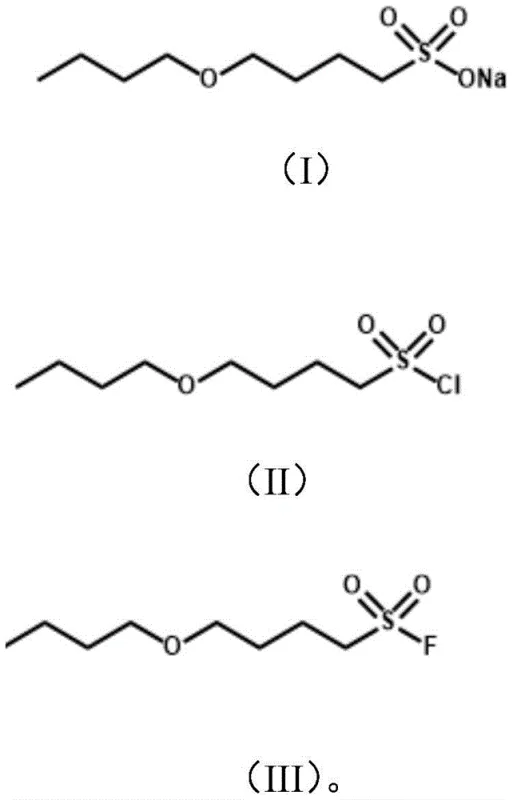 As illustrated in the reaction scheme, the process converts a sodium alkyl ether sulfonate intermediate into the corresponding sulfonyl chloride, which is subsequently fluorinated to yield the target sulfonyl fluoride. This stepwise construction allows for precise control over the carbon chain length and the position of the ether linkage, enabling the fine-tuning of physicochemical properties. The resulting precursors can be electrolytically fluorinated to produce perfluoroether compounds that exhibit excellent surface tension reduction capabilities comparable to legacy C8 compounds but with significantly improved environmental profiles.
As illustrated in the reaction scheme, the process converts a sodium alkyl ether sulfonate intermediate into the corresponding sulfonyl chloride, which is subsequently fluorinated to yield the target sulfonyl fluoride. This stepwise construction allows for precise control over the carbon chain length and the position of the ether linkage, enabling the fine-tuning of physicochemical properties. The resulting precursors can be electrolytically fluorinated to produce perfluoroether compounds that exhibit excellent surface tension reduction capabilities comparable to legacy C8 compounds but with significantly improved environmental profiles.
Mechanistic Insights into Multi-Step Functionalization and Halogen Exchange
The core of this synthetic strategy lies in the efficient construction of the sulfonate backbone followed by a halogen exchange sequence. The initial phase involves the reaction of tetrahydrofuran with acetyl chloride in the presence of zinc powder. This ring-opening reaction is carefully controlled, starting at low temperatures of 0-10°C to manage exothermicity, followed by heating to 60°C and finally 100°C to drive the formation of 4-chlorobutyl acetate. The use of zinc powder serves as a Lewis acid catalyst to facilitate the cleavage of the ether bond, ensuring high conversion rates without the need for expensive transition metals. Subsequently, the chloro-ester undergoes sulfonation with saturated aqueous sodium sulfite at elevated temperatures ranging from 100°C to 110°C. This nucleophilic substitution replaces the chlorine atom with a sulfonate group, yielding sodium 4-hydroxybutanesulfonate. The reaction conditions are optimized to prevent hydrolysis of the ester group while ensuring complete displacement of the halide, a critical balance for maintaining yield integrity.
Following the formation of the hydroxy-sulfonate, the process proceeds to an etherification step where the hydroxyl group is alkylated using a bromoalkane, such as 1-bromobutane, in the presence of sodium hydroxide. This step is conducted at high temperatures, typically around 110°C, to overcome the activation energy barrier for the SN2 reaction, resulting in the formation of the sodium alkyl ether sulfonate. 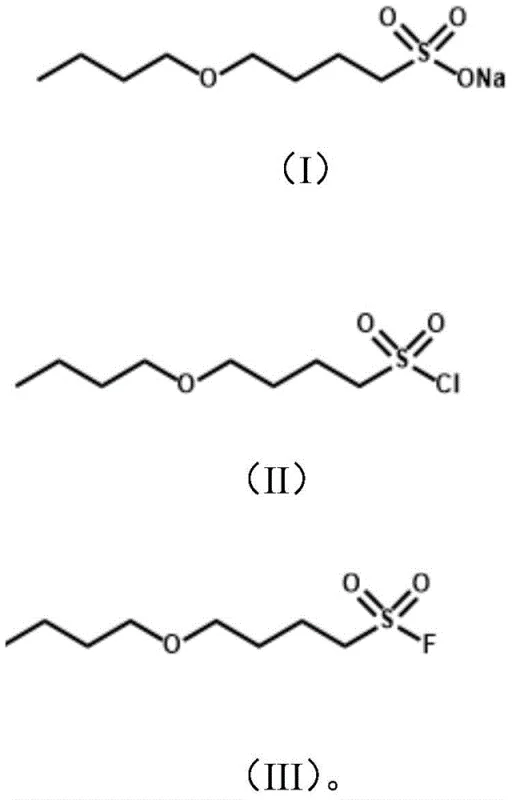 The final transformation involves converting the sulfonate salt into the reactive sulfonyl fluoride. First, the sodium sulfonate is treated with a chlorinating reagent such as phosphorus oxychloride (POCl3) or phosphorus pentachloride (PCl5) in a solvent like dichloromethane or chlorobenzene at 60-80°C. This generates the unstable sulfonyl chloride intermediate in situ. Immediately following this, a nucleophilic fluorination is performed using a fluoride source like potassium fluoride (KF) or sodium fluoride (NaF) in the presence of triethanolamine. The triethanolamine acts as a phase transfer catalyst and acid scavenger, facilitating the displacement of the chloride by the fluoride ion at temperatures between 50°C and 85°C. This two-step halogen exchange is crucial for introducing the sulfur-fluorine bond, which is the defining feature of the SuFEx (Sulfur-Fluoride Exchange) click chemistry motif, rendering the molecule highly versatile for downstream applications.
The final transformation involves converting the sulfonate salt into the reactive sulfonyl fluoride. First, the sodium sulfonate is treated with a chlorinating reagent such as phosphorus oxychloride (POCl3) or phosphorus pentachloride (PCl5) in a solvent like dichloromethane or chlorobenzene at 60-80°C. This generates the unstable sulfonyl chloride intermediate in situ. Immediately following this, a nucleophilic fluorination is performed using a fluoride source like potassium fluoride (KF) or sodium fluoride (NaF) in the presence of triethanolamine. The triethanolamine acts as a phase transfer catalyst and acid scavenger, facilitating the displacement of the chloride by the fluoride ion at temperatures between 50°C and 85°C. This two-step halogen exchange is crucial for introducing the sulfur-fluorine bond, which is the defining feature of the SuFEx (Sulfur-Fluoride Exchange) click chemistry motif, rendering the molecule highly versatile for downstream applications.
How to Synthesize 4-Butoxy-1-butylsulfonyl Fluoride Efficiently
The synthesis of 4-butoxy-1-butylsulfonyl fluoride represents a prime example of applying this patented technology to create a specific, high-value intermediate. The process integrates ring-opening chemistry with classical sulfonation and modern fluorination techniques to achieve a robust manufacturing protocol. Operators must strictly adhere to the temperature gradients specified in the patent, particularly during the initial ring-opening and the final fluorination steps, to maximize yield and minimize byproduct formation. The detailed standardized synthesis steps for this specific compound are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale execution.
- React tetrahydrofuran with acetyl chloride and zinc powder to form 4-chlorobutyl acetate.
- Perform sulfonation with sodium sulfite to generate sodium 4-hydroxybutanesulfonate.
- Conduct etherification with bromoalkane and sodium hydroxide to yield the sodium sulfonate intermediate.
- Convert the sulfonate to sulfonyl chloride using phosphorus oxychloride or phosphorus pentachloride.
- Execute nucleophilic fluorination with potassium fluoride or sodium fluoride to obtain the final sulfonyl fluoride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial strategic benefits over traditional fluorination methods. The reliance on commodity chemicals rather than specialized fluorinated building blocks drastically simplifies the sourcing process and mitigates supply risk. Tetrahydrofuran, acetyl chloride, sodium sulfite, and bromoalkanes are produced on a massive global scale, ensuring consistent availability and price stability. This contrasts sharply with processes that require imported perfluorinated telomers or specialized electrochemical cells, which are often subject to volatile market dynamics and geopolitical trade restrictions. By utilizing a chemistry based on abundant hydrocarbon feedstocks, manufacturers can secure a more resilient supply chain that is less susceptible to disruptions in the niche fluorine chemical sector.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of expensive catalysts and the use of straightforward reaction conditions. Traditional methods often require precious metal catalysts or energy-intensive electrochemical setups, whereas this route utilizes zinc powder and common phosphorus halides which are cost-effective and easily handled. Furthermore, the solvents employed, such as dichloromethane or chlorobenzene, are recyclable, allowing for significant recovery and reuse which lowers the overall operational expenditure. The avoidance of complex purification steps, relying instead on standard distillation and extraction, further reduces utility consumption and waste treatment costs, leading to a leaner manufacturing profile.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible production scheduling and inventory management. Since the intermediate sodium alkyl ether sulfonate is a stable solid, it can be stockpiled and converted to the final sulfonyl fluoride on demand, providing agility in responding to market fluctuations. The raw materials do not require cryogenic storage or special handling protocols associated with hazardous fluorine gas or anhydrous hydrogen fluoride, simplifying logistics and warehousing requirements. This ease of handling translates to faster lead times and reduced administrative burdens related to hazardous material transport, ensuring a smoother flow of goods from the production facility to the end customer.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard batch reactor equipment commonly found in fine chemical plants. The reaction parameters, such as temperature and pressure, are within the operating limits of standard glass-lined or stainless steel reactors, removing the need for capital-intensive specialized infrastructure. Additionally, the environmental profile of the process aligns with modern sustainability goals; the degradation products of the final perfluoroether derivatives are less toxic and bioaccumulative, reducing the long-term liability for manufacturers. The ability to produce "greener" alternatives positions companies favorably against tightening global regulations on persistent organic pollutants, future-proofing their product portfolios.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkyl ether sulfonyl fluoride synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of adopting this route for industrial applications.
Q: What are the key advantages of this alkyl ether sulfonyl fluoride synthesis route?
A: The process utilizes cheap, readily available raw materials like tetrahydrofuran and acetyl chloride. It avoids expensive transition metal catalysts and employs standard unit operations like distillation and extraction, making it highly suitable for industrial scale-up.
Q: How does this precursor contribute to environmental safety compared to traditional PFAS?
A: The resulting perfluoroether compounds derived from this precursor contain ether bonds which have lower bond energy than carbon-carbon single bonds. This structural feature facilitates easier degradation in the environment, significantly reducing bioaccumulation and toxicity compared to long-chain perfluoroalkyl substances.
Q: What are the critical reaction conditions for the fluorination step?
A: The nucleophilic fluorination typically requires reacting the sulfonyl chloride intermediate with a fluoride source such as potassium fluoride or sodium fluoride in the presence of a phase transfer catalyst like triethanolamine. The reaction is generally conducted at temperatures between 50°C and 85°C for 1 to 6 hours to ensure high conversion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Ether Sulfonyl Fluoride Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative patent technologies into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to next-generation surfactant precursors is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and corrosive challenges associated with sulfonation and halogen exchange reactions, guaranteeing stringent purity specifications for every batch. With rigorous QC labs and a commitment to process safety, we deliver high-purity alkyl ether sulfonyl fluoride intermediates that meet the exacting standards required for downstream electrolytic fluorination and final product formulation.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis route can enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain optimization initiatives, ensuring you stay ahead in the evolving landscape of specialty fluorinated chemicals.
