Advanced Manufacturing of Neutral Sodium Fosfomycin via Cation Exchange Technology
Advanced Manufacturing of Neutral Sodium Fosfomycin via Cation Exchange Technology
The pharmaceutical industry continuously seeks robust synthetic routes that enhance both the therapeutic profile and the manufacturability of critical antibiotics. Patent CN1268628C introduces a significant technological advancement in the production of neutral sodium fosfomycin, a vital broad-spectrum antibiotic used for treating serious bacterial infections. This innovation addresses long-standing challenges in pH control and process efficiency by utilizing rotatory fosfomycin dextrorotatory phenethylamine salts as the starting material. Through a sophisticated sequence of sodium hydroxide dissociation, cation exchange resin treatment, and anhydrous alcohol crystallization, the method yields a mixed sodium salt with a pH value closely aligned with the human physiological environment. For global procurement leaders and R&D directors, this patent represents a pivotal shift towards safer, more cost-effective, and environmentally sustainable manufacturing practices for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of sodium fosfomycin has been plagued by significant operational complexities and safety concerns regarding the final product's acidity. Traditional processes, such as those employed by certain legacy manufacturers, typically involve producing a highly alkaline fosfomycin sodium salt with a pH ranging from 9.0 to 10.5. To render this suitable for intravenous administration, manufacturers were forced to undertake a cumbersome post-treatment regimen involving the production of sterile organic acids, followed by their pulverization and precise quantitative mixing with the alkaline salt. This multi-step approach not only increases the risk of contamination due to the handling of multiple sterile components but also introduces substantial variability in the final pH. Furthermore, alternative methods reported in European patents utilized methanol and sodium methylate, which resulted in the irreversible loss of the valuable phenethylamine auxiliary due to complexation with acetone during crystallization, leading to inflated raw material costs and complicated solvent recovery systems.
The Novel Approach
The methodology outlined in patent CN1268628C offers a transformative solution by fundamentally redesigning the salt formation mechanism to intrinsically achieve the desired physiological pH. Instead of relying on external acidification, this novel approach leverages the precise stoichiometry of cation exchange resins to convert the initial disodium salt into a balanced mixture of mono- and di-sodium salts. By reacting the raw material with aqueous sodium hydroxide under controlled thermal conditions, the process efficiently liberates the phenethylamine, which separates naturally as an upper organic layer for immediate recovery and reuse. The subsequent treatment with strong acid cation exchange resins allows for fine-tuning of the sodium ion content, resulting in a final product with a pH between 7.0 and 8.0. This eliminates the need for sterile organic acid production entirely, streamlining the workflow and ensuring a product profile that is inherently safer for clinical application while drastically simplifying the supply chain logistics.
Mechanistic Insights into Cation Exchange and Salt Modulation
The core chemical innovation lies in the strategic manipulation of ionic equilibria to modulate the basicity of the fosfomycin molecule without introducing foreign acidic counterions. The process begins with the dissociation of the rotatory fosfomycin dextrorotatory phenethylamine salt (left salt) using a calculated amount of sodium hydroxide in an aqueous medium. This reaction cleaves the amine bond, releasing the free phenethylamine and generating the disodium salt of fosfomycin in the aqueous phase. The reaction conditions are meticulously maintained between 35°C and 40°C to ensure complete dissociation while preventing thermal degradation of the sensitive epoxypropyl moiety. Following static layering, the upper phenethylamine layer is physically separated, capitalizing on the immiscibility of the organic amine with the aqueous salt solution, which is a critical step for material economy.
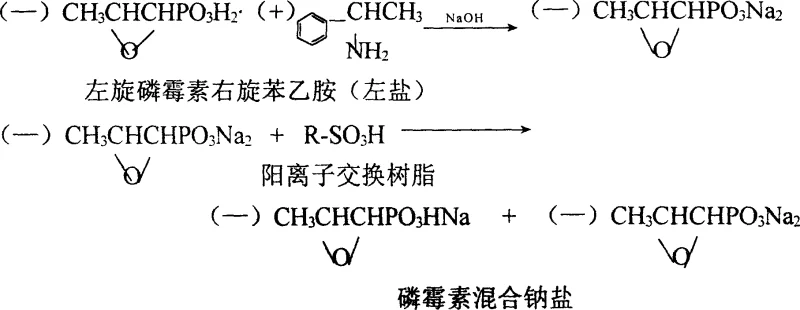
Subsequent to the separation, the aqueous fosfomycin sodium solution undergoes a critical ion exchange process. The solution is passed through or stirred with a strong acid cation exchange resin, such as D001 or 732 type resins. During this phase, hydrogen ions from the resin partially replace sodium ions on the phosphonate groups, effectively converting a portion of the disodium salt into the monosodium form. This partial neutralization is the key to achieving the target pH range of 7.0 to 8.0, which mimics the human blood environment (pH 7.4) far better than the highly alkaline predecessors. The mechanism ensures that the final crystal lattice contains a stable ratio of mono- and di-sodium species, providing enhanced biological availability and reduced sodium load for patients with cardiovascular sensitivities, all while maintaining the structural integrity of the antibiotic.
How to Synthesize Neutral Sodium Fosfomycin Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process is designed to be scalable, moving seamlessly from laboratory validation to commercial production lines. Operators must carefully monitor the temperature during the initial alkaline dissociation to prevent side reactions, and the residence time on the ion exchange column must be optimized to achieve the exact pH specification without over-acidifying the product. The final crystallization step utilizes anhydrous ethanol as an anti-solvent, which induces the precipitation of the mixed sodium salt in high purity. For a detailed breakdown of the specific operational parameters, reagent ratios, and equipment setups required to replicate this high-efficiency pathway, please refer to the standardized technical guide below.
- Dissociate rotatory fosfomycin dextrorotatory phenethylamine salts using aqueous sodium hydroxide at controlled temperatures to release free amine.
- Perform cation exchange on the resulting saline solution using strong acid cation exchange resin to adjust the sodium ion ratio.
- Crystallize the final mixed sodium salt product by adding the purified solution to anhydrous ethanol followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this resin-exchange technology offers profound benefits for cost reduction in pharmaceutical manufacturing and supply chain resilience. By eliminating the requirement for the separate production, sterilization, and micronization of organic acids, manufacturers can significantly reduce capital expenditure on specialized processing equipment and cleanroom infrastructure. The removal of these unit operations not only shortens the overall production cycle time but also minimizes the potential points of failure where contamination could occur, thereby enhancing the overall reliability of the supply chain. Furthermore, the ability to recover and recycle the dextrorotatory phenethylamine directly from the reaction mixture represents a substantial saving in raw material costs, as this chiral auxiliary is often a high-value component in the synthesis of fosfomycin derivatives.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the downstream processing stages. In traditional methods, the quantitative mixing of sterile acids and bases requires rigorous quality control testing at multiple stages to ensure homogeneity and pH accuracy, which consumes significant labor and analytical resources. By contrast, the new method achieves the target pH intrinsically through the resin exchange mechanism, effectively collapsing multiple processing steps into a single continuous operation. This consolidation leads to drastic reductions in utility consumption, particularly in terms of energy required for drying and milling, and reduces the workforce needed for manual handling of sterile powders, resulting in a leaner and more cost-efficient production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the dependency on multiple specialized raw materials, such as sterile organic acids, which may have long lead times or limited supplier bases. This innovative route mitigates such risks by relying on commodity chemicals like sodium hydroxide and standard ion exchange resins, which are readily available in the global market. Additionally, the internal recycling of the phenethylamine auxiliary reduces the net consumption of this critical chiral building block, insulating the manufacturer from volatility in the pricing or availability of external chiral reagents. This self-sufficiency ensures a more stable and predictable production schedule, allowing suppliers to meet tight delivery windows for high-purity pharmaceutical intermediates with greater confidence.
- Scalability and Environmental Compliance: The environmental footprint of pharmaceutical production is increasingly scrutinized, and this method offers clear advantages in waste management and solvent usage. Unlike the methanol-acetone systems described in prior art, which generate complex mixed solvent wastes that are difficult to separate and recycle, this process utilizes water and ethanol, both of which are easier to recover and dispose of safely. The elimination of acetone also removes the risk of forming difficult-to-remove adducts with the amine, further simplifying waste streams. The process is inherently scalable, as ion exchange technology is well-established in large-scale industrial applications, allowing for seamless expansion from pilot batches to multi-ton annual production capacities without the need for fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this neutral sodium fosfomycin synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of how this technology resolves historical manufacturing bottlenecks. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production facilities or for procurement specialists assessing the quality attributes of the final API.
Q: How does the new resin exchange method improve patient safety compared to traditional alkaline fosfomycin sodium?
A: Traditional methods often yield highly alkaline products (pH 9.0-10.5) requiring complex mixing with sterile organic acids to lower pH. The patented resin exchange method directly produces a mixed mono- and di-sodium salt with a pH of 7.0-8.0, which is much closer to human blood pH (7.4), thereby reducing the risk of injection site irritation and systemic side effects without needing additional acidification steps.
Q: What are the economic advantages of recovering phenethylamine in this synthesis route?
A: Unlike previous methods using methanol and acetone where phenethylamine forms complexes that are difficult to recover, this aqueous NaOH method allows the phenethylamine to separate as a distinct upper organic layer. This physical separation enables direct recycling of the chiral auxiliary, significantly reducing raw material costs and waste disposal burdens associated with lost amines.
Q: Does this process eliminate the need for sterile organic acid production?
A: Yes, a major bottleneck in the prior art was the requirement to produce, pulverize, and quantitatively mix sterile organic acids with the alkaline salt to adjust pH. This new method achieves the target pH range intrinsically through stoichiometric control during the resin exchange step, completely removing the need for separate sterile acid manufacturing and mixing operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Fosfomycin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN1268628C to deliver superior pharmaceutical ingredients to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this resin-exchange technology are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise pH control and residual solvent analysis, guaranteeing that every batch of sodium fosfomycin meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and clinically superior manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your COGS. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply of this essential antibiotic remains uninterrupted, high-quality, and economically competitive in the evolving healthcare landscape.
